Abstract
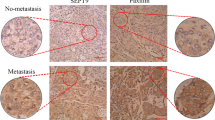
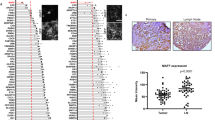
Hypoxia is a common feature of locally advanced breast cancers that is associated with increased metastasis and poorer survival. Stabilisation of hypoxia-inducible factor-1α (HIF1α) in tumours causes transcriptional changes in numerous genes that function at distinct stages of the metastatic cascade. We demonstrate that expression of RIOK3 (RIght Open reading frame kinase 3) was increased during hypoxic exposure in an HIF1α-dependent manner. RIOK3 was localised to distinct cytoplasmic aggregates in normoxic cells and underwent redistribution to the leading edge of the cell in hypoxia with a corresponding change in the organisation of the actin cytoskeleton. Depletion of RIOK3 expression caused MDA-MB-231 to become elongated and this morphological change was due to a loss of protraction at the trailing edge of the cell. This phenotypic change resulted in reduced cell migration in two-dimensional cultures and inhibition of cell invasion through three-dimensional extracellular matrix. Proteomic analysis identified interactions of RIOK3 with actin and several actin-binding factors including tropomyosins (TPM3 and TPM4) and tropomodulin 3. Depletion of RIOK3 in cells resulted in fewer and less organised actin filaments. Analysis of these filaments showed reduced association of TPM3, particularly during hypoxia, suggesting that RIOK3 regulates actin filament specialisation. RIOK3 depletion reduced the dissemination of MDA-MB-231 cells in both a zebrafish model of systemic metastasis and a mouse model of pulmonary metastasis. These findings demonstrate that RIOK3 is necessary for maintaining actin cytoskeletal organisation required for migration and invasion, biological processes that are necessary for hypoxia-driven metastasis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Elias AD . Triple-negative breast cancer: a short review. Am J Clin Oncol 2010; 33: 637–645.
Vaupel P, Hockel M, Mayer A . Detection and characterization of tumor hypoxia using pO2 histography. Antioxid Redox Signal 2007; 9: 1221–1235.
Rundqvist H, Johnson RS . Hypoxia and metastasis in breast cancer. Curr Top Microbiol Immunol 2010; 345: 121–139.
Dales JP, Garcia S, Meunier-Carpentier S, Andrac-Meyer L, Haddad O, Lavaut MN et al. Overexpression of hypoxia-inducible factor HIF-1alpha predicts early relapse in breast cancer: retrospective study in a series of 745 patients. Int J Cancer J Int Cancer 2005; 116: 734–739.
Yamamoto Y, Ibusuki M, Okumura Y, Kawasoe T, Kai K, Iyama K et al. Hypoxia-inducible factor 1alpha is closely linked to an aggressive phenotype in breast cancer. Breast Cancer Res Treat 2008; 110: 465–475.
Erler JT, Bennewith KL, Nicolau M, Dornhofer N, Kong C, Le QT et al. Lysyl oxidase is essential for hypoxia-induced metastasis. Nature 2006; 440: 1222–1226.
Gilkes DM, Chaturvedi P, Bajpai S, Wong CC, Wei H, Pitcairn S et al. Collagen prolyl hydroxylases are essential for breast cancer metastasis. Cancer Res 2013; 73: 3285–3296.
Munoz-Najar UM, Neurath KM, Vumbaca F, Claffey KP . Hypoxia stimulates breast carcinoma cell invasion through MT1-MMP and MMP-2 activation. Oncogene 2006; 25: 2379–2392.
Zhang H, Wong CC, Wei H, Gilkes DM, Korangath P, Chaturvedi P et al. HIF-1-dependent expression of angiopoietin-like 4 and L1CAM mediates vascular metastasis of hypoxic breast cancer cells to the lungs. Oncogene 2012; 31: 1757–1770.
Imai T, Horiuchi A, Wang C, Oka K, Ohira S, Nikaido T et al. Hypoxia attenuates the expression of E-cadherin via up-regulation of SNAIL in ovarian carcinoma cells. Am J Pathol 2003; 163: 1437–1447.
Pennacchietti S, Michieli P, Galluzzo M, Mazzone M, Giordano S, Comoglio PM . Hypoxia promotes invasive growth by transcriptional activation of the met protooncogene. Cancer Cell 2003; 3: 347–361.
Ponti A, Machacek M, Gupton SL, Waterman-Storer CM, Danuser G . Two distinct actin networks drive the protrusion of migrating cells. Science 2004; 305: 1782–1786.
Insall RH, Machesky LM . Actin dynamics at the leading edge: from simple machinery to complex networks. Dev Cell 2009; 17: 310–322.
Gupton SL, Anderson KL, Kole TP, Fischer RS, Ponti A, Hitchcock-DeGregori SE et al. Cell migration without a lamellipodium: translation of actin dynamics into cell movement mediated by tropomyosin. J Cell Biol 2005; 168: 619–631.
Weber KL, Fischer RS, Fowler VM . Tmod3 regulates polarized epithelial cell morphology. J Cell Sci 2007; 120: 3625–3632.
Tojkander S, Gateva G, Schevzov G, Hotulainen P, Naumanen P, Martin C et al. A molecular pathway for myosin II recruitment to stress fibers. Curr Biol 2011; 21: 539–550.
Baumas K, Soudet J, Caizergues-Ferrer M, Faubladier M, Henry Y, Mougin A . Human RioK3 is a novel component of cytoplasmic pre-40S pre-ribosomal particles. RNA Biol 2012; 9: 162–174.
Widmann B, Wandrey F, Badertscher L, Wyler E, Pfannstiel J, Zemp I et al. The kinase activity of human Rio1 is required for final steps of cytoplasmic maturation of 40S subunits. Mol Biol Cell 2012; 23: 22–35.
Zemp I, Wild T, O'Donohue MF, Wandrey F, Widmann B, Gleizes PE et al. Distinct cytoplasmic maturation steps of 40S ribosomal subunit precursors require hRio2. J Cell Biol 2009; 185: 1167–1180.
Kimmelman AC, Hezel AF, Aguirre AJ, Zheng H, Paik JH, Ying H et al. Genomic alterations link Rho family of GTPases to the highly invasive phenotype of pancreas cancer. Proc Natl Acad Sci USA 2008; 105: 19372–19377.
Rzymski T, Milani M, Pike L, Buffa F, Mellor HR, Winchester L et al. Regulation of autophagy by ATF4 in response to severe hypoxia. Oncogene 2010; 29: 4424–4435.
Curtis C, Shah SP, Chin SF, Turashvili G, Rueda OM, Dunning MJ et al. The genomic and transcriptomic architecture of 2,000 breast tumours reveals novel subgroups. Nature 2012; 486: 346–352.
TCGA. Comprehensive molecular portraits of human breast tumours. Nature 2012; 490: 61–70.
Buffa FM, Harris AL, West CM, Miller CJ . Large meta-analysis of multiple cancers reveals a common, compact and highly prognostic hypoxia metagene. Br J Cancer 2010; 102: 428–435.
Carroll VA, Ashcroft M . Role of hypoxia-inducible factor (HIF)-1alpha versus HIF-2alpha in the regulation of HIF target genes in response to hypoxia, insulin-like growth factor-I, or loss of von Hippel-Lindau function: implications for targeting the HIF pathway. Cancer Res 2006; 66: 6264–6270.
Hu CJ, Wang LY, Chodosh LA, Keith B, Simon MC . Differential roles of hypoxia-inducible factor 1alpha (HIF-1alpha) and HIF-2alpha in hypoxic gene regulation. Mol Cell Biol 2003; 23: 9361–9374.
Raval RR, Lau KW, Tran MG, Sowter HM, Mandriota SJ, Li JL et al. Contrasting properties of hypoxia-inducible factor 1 (HIF-1) and HIF-2 in von Hippel-Lindau-associated renal cell carcinoma. Mol Cell Biol 2005; 25: 5675–5686.
Agus DB, Alexander JF, Arap W, Ashili S, Aslan JE, Austin RH et al. A physical sciences network characterization of non-tumorigenic and metastatic cells. Scientific Rep 2013; 3: 1449.
LaRonde-LeBlanc N, Wlodawer A . A family portrait of the RIO kinases. J Biol Chem 2005; 280: 37297–37300.
Koch A, Hauf S . Strategies for the identification of kinase substrates using analog-sensitive kinases. Eur J Cell Biol 2010; 89: 184–193.
Rouhi P, Jensen LD, Cao Z, Hosaka K, Lanne T, Wahlberg E et al. Hypoxia-induced metastasis model in embryonic zebrafish. Nat Protoc 2010; 5: 1911–1918.
Lammermann T, Bader BL, Monkley SJ, Worbs T, Wedlich-Soldner R, Hirsch K et al. Rapid leukocyte migration by integrin-independent flowing and squeezing. Nature 2008; 453: 51–55.
Fournier MF, Sauser R, Ambrosi D, Meister JJ, Verkhovsky AB . Force transmission in migrating cells. J Cell Biol 2010; 188: 287–297.
Cramer LP . Mechanism of cell rear retraction in migrating cells. Curr Opin Cell Biol 2013; 25: 591–599.
Poincloux R, Collin O, Lizarraga F, Romao M, Debray M, Piel M et al. Contractility of the cell rear drives invasion of breast tumor cells in 3D Matrigel. Proc Natl Acad Sci USA 2011; 108: 1943–1948.
Semenza GL . Hypoxia-inducible factors in physiology and medicine. Cell 2012; 148: 399–408.
Weidemann A, Breyer J, Rehm M, Eckardt KU, Daniel C, Cicha I et al. HIF-1alpha activation results in actin cytoskeleton reorganization and modulation of Rac-1 signaling in endothelial cells. Cell Commun Signal 2013; 11: 80.
Sims D, Mendes-Pereira AM, Frankum J, Burgess D, Cerone MA, Lombardelli C et al. High-throughput RNA interference screening using pooled shRNA libraries and next generation sequencing. Genome Biol 2011; 12: R104.
Meijering E, Dzyubachyk O, Smal I . Methods for cell and particle tracking. Methods Enzymol 2012; 504: 183–200.
Mackeen MM, Kramer HB, Chang KH, Coleman ML, Hopkinson RJ, Schofield CJ et al. Small-molecule-based inhibition of histone demethylation in cells assessed by quantitative mass spectrometry. J Proteome Res 2010; 9: 4082–4092.
Alcoser SY, Kimmel DJ, Borgel SD, Carter JP, Dougherty KM, Hollingshead MG . Real-time PCR-based assay to quantify the relative amount of human and mouse tissue present in tumor xenografts. BMC Biotechnol 2011; 11: 124.
Acknowledgements
European Commission Metoxia Grant No. 222741 (D Singleton, P Rouhi, Y Cao, A Harris), Cancer Research UK (A Harris, C Zois, S Haider, J Li), Breast Cancer Research Foundation (A Harris) and Oxford NIHR Biomedical Research Centre (B Kessler). This study makes use of data generated by the Molecular Taxonomy of Breast Cancer International Consortium, which was funded by Cancer Research UK and the British Columbia Cancer Agency Branch.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Singleton, D., Rouhi, P., Zois, C. et al. Hypoxic regulation of RIOK3 is a major mechanism for cancer cell invasion and metastasis. Oncogene 34, 4713–4722 (2015). https://doi.org/10.1038/onc.2014.396
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2014.396