Abstract
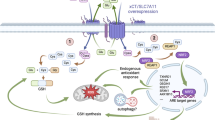
The epithelial–mesenchymal transition (EMT) is activated in cancer cells by ZEB1, a member of the zinc finger/homeodomain family of transcriptional repressors. The mucin 1 (MUC1) heterodimeric protein is aberrantly overexpressed in human carcinoma cells. The present studies in breast cancer cells demonstrate that the oncogenic MUC1-C subunit induces expression of ZEB1 by a NF-κB (nuclear factor kappa B) p65-dependent mechanism. MUC1-C occupies the ZEB1 promoter with NF-κB p65 and thereby promotes ZEB1 transcription. In turn, ZEB1 associates with MUC1-C and the ZEB1/MUC1-C complex contributes to the transcriptional suppression of miR-200c, an inducer of epithelial differentiation. The co-ordinate upregulation of ZEB1 and suppression of miR-200c has been linked to the induction of EMT. In concert with the effects of MUC1-C on ZEB1 and miR-200c, we show that MUC1-C induces EMT and cellular invasion by a ZEB1-mediated mechanism. These findings indicate that (i) MUC1-C activates ZEB1 and suppresses miR-200c with the induction of EMT and (ii) targeting MUC1-C could be an effective approach for the treatment of breast and possibly other types of cancers that develop EMT properties.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Polyak K, Weinberg RA . Transitions between epithelial and mesenchymal states: acquisition of malignant and stem cell traits. Nat Rev Cancer 2009; 9: 265–273.
Thiery JP, Acloque H, Huang RY, Nieto MA . Epithelial-mesenchymal transitions in development and disease. Cell 2009; 139: 871–890.
Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan A, Zhou AY et al. The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell 2008; 133: 704–715.
Morel AP, Lievre M, Thomas C, Hinkal G, Ansieau S, Puisieux A . Generation of breast cancer stem cells through epithelial-mesenchymal transition. PLoS ONe 2008; 3: e2888.
Wellner U, Schubert J, Burk UC, Schmalhofer O, Zhu F, Sonntag A et al. The EMT-activator ZEB1 promotes tumorigenicity by repressing stemness-inhibiting microRNAs. Nat Cell Biol 2009; 11: 1487–1495.
Brabletz S, Brabletz T . The ZEB/miR-200 feedback loop–a motor of cellular plasticity in development and cancer? EMBO Rep 2010; 11: 670–677.
Spaderna S, Schmalhofer O, Wahlbuhl M, Dimmler A, Bauer K, Sultan A et al. The transcriptional repressor ZEB1 promotes metastasis and loss of cell polarity in cancer. Cancer Res 2008; 68: 537–544.
Spaderna S, Schmalhofer O, Hlubek F, Berx G, Eger A, Merkel S et al. A transient, EMT-linked loss of basement membranes indicates metastasis and poor survival in colorectal cancer. Gastroenterology 2006; 131: 830–840.
Aigner K, Dampier B, Descovich L, Mikula M, Sultan A, Schreiber M et al. The transcription factor ZEB1 (deltaEF1) promotes tumour cell dedifferentiation by repressing master regulators of epithelial polarity. Oncogene 2007; 26: 6979–6988.
Vandewalle C, Van Roy F, Berx G . The role of the ZEB family of transcription factors in development and disease. Cell Mol Life Sci 2009; 66: 773–787.
Burk U, Schubert J, Wellner U, Schmalhofer O, Vincan E, Spaderna S et al. A reciprocal repression between ZEB1 and members of the miR-200 family promotes EMT and invasion in cancer cells. EMBO Rep 2008; 9: 582–589.
Kufe D . Mucins in cancer: function, prognosis and therapy. Nat Rev Canc 2009; 9: 874–885.
Kufe D . MUC1-C oncoprotein as a target in breast cancer: activation of signaling pathways and therapeutic approaches. Oncogene 2012; 32: 1073–1081.
Horn G, Gaziel A, Wreschner DH, Smorodinsky NI, Ehrlich M . ERK and PI3K regulate different aspects of the epithelial to mesenchymal transition of mammary tumor cells induced by truncated MUC1. Exp Cell Res 2009; 315: 1490–1504.
Roy LD, Sahraei M, Subramani DB, Besmer D, Nath S, Tinder TL et al. MUC1 enhances invasiveness of pancreatic cancer cells by inducing epithelial to mesenchymal transition. Oncogene 2011; 30: 1449–1459.
Rajabi H, Ahmad R, Jin C, Joshi M, Guha M, Alam M et al. MUC1-C oncoprotein confers androgen-independent growth of human prostate cancer cells. Prostate 2012; 72: 1659–1668.
Ahmad R, Raina D, Joshi MD, Kawano T, Kharbanda S, Kufe D . MUC1-C oncoprotein functions as a direct activator of the NF-κB p65 transcription factor. Cancer Res 2009; 69: 7013–7021.
Ren J, Agata N, Chen D, Li Y, Yu W-H, Huang L et al. Human MUC1 carcinoma-associated protein confers resistance to genotoxic anti-cancer agents. Cancer Cell 2004; 5: 163–175.
Chua HL, Bhat-Nakshatri P, Clare SE, Morimiya A, Badve S, Nakshatri H . NF-kappaB represses E-cadherin expression and enhances epithelial to mesenchymal transition of mammary epithelial cells: potential involvement of ZEB-1 and ZEB-2. Oncogene 2007; 26: 711–724.
Hu X, Janssen WE, Moscinski LC, Bryington M, Dangsupa A, Rezai-Zadeh N et al. An IkappaBalpha inhibitor causes leukemia cell death through a p38 MAP kinase-dependent, NF-kappaB-independent mechanism. Cancer Res 2001; 61: 6290–6296.
Rajabi H, Ahmad R, Jin C, Kosugi M, Alam M, Joshi M et al. MUC1-C oncoprotein induces TCF7L2 activation and promotes cyclin D1 expression in human breast cancer cells. J Biol Chem 2012; 287: 10703–10713.
Leng Y, Cao C, Ren J, Huang L, Chen D, Ito M et al. Nuclear import of the MUC1-C oncoprotein is mediated by nucleoporin Nup62. J Biol Chem 2007; 282: 19321–19330.
Raina D, Ahmad R, Rajabi H, Panchamoorthy G, Kharbanda S, Kufe D . Targeting cysteine-mediated dimerization of the MUC1-C oncoprotein in human cancer cells. Int J Oncol 2012; 40: 1643–1649.
Massague J . TGFbeta in Cancer. Cell 2008; 134: 215–230.
Wang Z, Banerjee S, Li Y, Rahman KM, Zhang Y, Sarkar FH . Down-regulation of notch-1 inhibits invasion by inactivation of nuclear factor-kappaB, vascular endothelial growth factor, and matrix metalloproteinase-9 in pancreatic cancer cells. Cancer Res 2006; 66: 2778–2784.
Sanchez-Tillo E, de Barrios O, Siles L, Cuatrecasas M, Castells A, Postigo A . beta-catenin/TCF4 complex induces the epithelial-to-mesenchymal transition (EMT)-activator ZEB1 to regulate tumor invasiveness. Proc Natl Acad Sci USA 2011; 108: 19204–19209.
Huang L, Chen D, Liu D, Yin L, Kharbanda S, Kufe D . MUC1 oncoprotein blocks GSK3β-mediated phosphorylation and degradation of β-catenin. Cancer Res 2005; 65: 10413–10422.
Bracken CP, Gregory PA, Kolesnikoff N, Bert AG, Wang J, Shannon MF et al. A double-negative feedback loop between ZEB1-SIP1 and the microRNA-200 family regulates epithelial-mesenchymal transition. Cancer Res 2008; 68: 7846–7854.
Shimono Y, Zabala M, Cho RW, Lobo N, Dalerba P, Qian D et al. Downregulation of miRNA-200c links breast cancer stem cells with normal stem cells. Cell 2009; 138: 592–603.
Ocana OH, Corcoles R, Fabra A, Moreno-Bueno G, Acloque H, Vega S et al. Metastatic colonization requires the repression of the epithelial-mesenchymal transition inducer prrx1. Cancer Cell 2012; 22: 709–724.
Eger A, Aigner K, Sonderegger S, Dampier B, Oehler S, Schreiber M et al. DeltaEF1 is a transcriptional repressor of E-cadherin and regulates epithelial plasticity in breast cancer cells. Oncogene 2005; 24: 2375–2385.
Jin C, Rajabi H, Pitroda S, Kharbanda A, Li A, Weichselbaum R et al. Cooperative interaction between the MUC1 oncoprotein and the Rab31 GTPase in human breast cancer cells. PLoS ONE 2012; 7: e39432.
Ahmad R, Rajabi H, Kosugi M, Joshi M, Alam M, Vasir B et al. MUC1-C oncoprotein promotes STAT3 activation in an auto-inductive regulatory loop. Science Signaling 2011; 4: ra9.
Wang Q, Carroll JS, Brown M . Spatial and temporal recruitment of androgen receptor and its coactivators involves chromosomal looping and polymerase tracking. Mol Cell 2005; 19: 631–642.
Panchamoorthy G, Rehan H, Kharbanda A, Ahmad R, Kufe D . A monoclonal antibody against the oncogenic mucin 1 cytoplasmic domain. Hybridoma 2011; 30: 531–535.
Rajabi H, Jin C, Ahmad R, McClary C, Kufe D . Mucin 1 oncoprotein expression is suppressed by the miR-125b oncomir. Genes & Cancer 2010; 1: 62–65.
Mehrotra S, Languino LR, Raskett CM, Mercurio AM, Dohi T, Altieri DC . IAP regulation of metastasis. Cancer Cell 2010; 17: 53–64.
Acknowledgements
Research reported in this publication was supported by the National Cancer Institute of the National Institutes of Health under award numbers CA97098 and CA166480.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Dr D Kufe is a founder of Genus Oncology and holds equity in the company. The remaining authors disclosed no potential conflicts of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Rajabi, H., Alam, M., Takahashi, H. et al. MUC1-C oncoprotein activates the ZEB1/miR-200c regulatory loop and epithelial–mesenchymal transition. Oncogene 33, 1680–1689 (2014). https://doi.org/10.1038/onc.2013.114
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2013.114
Keywords
This article is cited by
-
MUC1-C regulates NEAT1 lncRNA expression and paraspeckle formation in cancer progression
Oncogene (2024)
-
MUC1-C intersects chronic inflammation with epigenetic reprogramming by regulating the set1a compass complex in cancer progression
Communications Biology (2023)
-
Impact of AMPK on cervical carcinoma progression and metastasis
Cell Death & Disease (2023)
-
The role of the ZEB1–neuroinflammation axis in CNS disorders
Journal of Neuroinflammation (2022)
-
Programmed death-ligand 1 signaling and expression are reversible by lycopene via PI3K/AKT and Raf/MEK/ERK pathways in tongue squamous cell carcinoma
Genes & Nutrition (2022)