Abstract
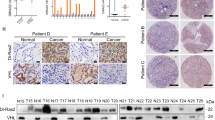
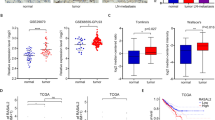
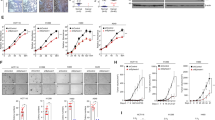
The small GTPase Ral is known to be highly activated in several human cancers, such as bladder, colon and pancreas cancers. It is reported that activated Ral is involved in cell proliferation, migration and metastasis of bladder cancer. This protein is activated by Ral guanine nucleotide exchange factors (RalGEFs) and inactivated by Ral GTPase-activating proteins (RalGAPs), the latter of which consist of heterodimers containing a catalytic α1 or α2 subunit and a common β subunit. In Ras-driven cancers, such as pancreas and colon cancers, constitutively active Ras mutant activates Ral through interaction with RalGEFs, which contain the Ras association domain. However, little is known with regard to the mechanism that governs aberrant activation of Ral in bladder cancer, in which Ras mutations are relatively infrequent. Here, we show that Ral was highly activated in invasive bladder cancer cells due to reduced expression of RalGAPα2, the dominant catalytic subunit in bladder, rather than increased expression of RalGEFs. Exogenous expression of wild-type RalGAPα2 in KU7 bladder cancer cells with invasive phenotype, but not mutant RalGAPα2-N1742K lacking RalGAP activity, resulted in attenuated cell migration in vitro and lung metastasis in vivo. Furthermore, genetic ablation of Ralgapa2 promoted tumor invasion in a chemically-induced murine bladder cancer model. Importantly, immunohistochemical analysis of human bladder cancer specimens revealed that lower expression of RalGAPα2 was associated with advanced clinical stage and poor survival of patients. Collectively, these results are highly indicative that attenuated expression of RalGAPα2 leads to disease progression of bladder cancer through enhancement of Ral activity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Cordon-Cardo C . Molecular alterations associated with bladder cancer initiation and progression. Scand J Urol Nephrol 2008; 218: 154–165.
Knowles MA . Molecular subtypes of bladder cancer: Jekyll and Hyde or chalk and cheese? Carcinogenesis 2006; 27: 361–373.
Wu XR . Urothelial tumorigenesis: a tale of divergent pathways. Nat Rev 2005; 5: 713–725.
Smith SC, Oxford G, Baras AS, Owens C, Havaleshko D, Brautigan DL et al. Expression of ral GTPases, their effectors, and activators in human bladder cancer. Clin Cancer Res 2007; 13: 3803–3813.
Martin TD, Samuel JC, Routh ED, Der CJ, Yeh JJ . Activation and involvement of Ral GTPases in colorectal cancer. Cancer Res 2011; 71: 206–215.
Lim KH, O'Hayer K, Adam SJ, Kendall SD, Campbell PM, Der CJ et al. Divergent roles for RalA and RalB in malignant growth of human pancreatic carcinoma cells. Curr Biol 2006; 16: 2385–2394.
Oxford G, Owens CR, Titus BJ, Foreman TL, Herlevsen MC, Smith SC et al. RalA and RalB: antagonistic relatives in cancer cell migration. Cancer Res 2005; 65: 7111–7120.
Wang H, Owens C, Chandra N, Conaway MR, Brautigan DL, Theodorescu D . Phosphorylation of RalB is important for bladder cancer cell growth and metastasis. Cancer Res 2010; 70: 8760–8769.
Feig LA . Ral-GTPases: approaching their 15 min of fame. Trends cell biol 2003; 13: 419–425.
Bodemann BO, White MA . Ral GTPases and cancer: linchpin support of the tumorigenic platform. Nat Rev 2008; 8: 133–140.
Chien Y, White MA . RAL GTPases are linchpin modulators of human tumour-cell proliferation and survival. EMBO Reports 2003; 4: 800–806.
Shao H, Andres DA . A novel RalGEF-like protein, RGL3, as a candidate effector for rit and Ras. J Biol Chem 2000; 275: 26914–26924.
Shirakawa R, Fukai S, Kawato M, Higashi T, Kondo H, Ikeda T et al. Tuberous sclerosis tumor suppressor complex-like complexes act as GTPase-activating proteins for Ral GTPases. J Biol Chem 2009; 284: 21580–21588.
Downward J . Targeting RAS signalling pathways in cancer therapy. Nat Rev 2003; 3: 11–22.
Blaveri E, Simko JP, Korkola JE, Brewer JL, Baehner F, Mehta K et al. Bladder cancer outcome and subtype classification by gene expression. Clin Cancer Res 2005; 11: 4044–4055.
Sanchez-Carbayo M, Socci ND, Lozano J, Saint F, Cordon-Cardo C . Defining molecular profiles of poor outcome in patients with invasive bladder cancer using oligonucleotide microarrays. J Clin Oncol 2006; 24: 778–789.
Vigil D, Martin TD, Williams F, Yeh JJ, Campbell SL, Der CJ . Aberrant overexpression of the Rgl2 Ral small GTPase-specific guanine nucleotide exchange factor promotes pancreatic cancer growth through Ral-dependent and Ral-independent mechanisms. J Biol Chem 2010; 285: 34729–34740.
Lim KH, Baines AT, Fiordalisi JJ, Shipitsin M, Feig LA, Cox AD et al. Activation of RalA is critical for Ras-induced tumorigenesis of human cells. Cancer Cell 2005; 7: 533–545.
Chien Y, Kim S, Bumeister R, Loo YM, Kwon SW, Johnson CL et al. RalB GTPase-mediated activation of the IkappaB family kinase TBK1 couples innate immune signaling to tumor cell survival. Cell 2006; 127: 157–170.
Smith JJ, Deane NG, Wu F, Merchant NB, Zhang B, Jiang A et al. Experimentally derived metastasis gene expression profile predicts recurrence and death in patients with colon cancer. Gastroenterology 2009; 138: 958–968.
Zou TT, Selaru FM, Xu Y, Shustova V, Yin J, Mori Y et al. Application of cDNA microarrays to generate a molecular taxonomy capable of distinguishing between colon cancer and normal colon. Oncogene 2002; 21: 4855–4862.
Pei H, Li L, Fridley BL, Jenkins GD, Kalari KR, Lingle W et al. FKBP51 affects cancer cell response to chemotherapy by negatively regulating Akt. Cancer Cell 2009; 16: 259–266.
Badea L, Herlea V, Dima SO, Dumitrascu T, Popescu I . Combined gene expression analysis of whole-tissue and microdissected pancreatic ductal adenocarcinoma identifies genes specifically overexpressed in tumor epithelia. Hepato-Gastroenterology 2008; 55: 2016–2027.
Richardson AL, Wang ZC, De Nicolo A, Lu X, Brown M, Miron A et al. X chromosomal abnormalities in basal-like human breast cancer. Cancer Cell 2006; 9: 121–132.
Finak G, Bertos N, Pepin F, Sadekova S, Souleimanova M, Zhao H et al. Stromal gene expression predicts clinical outcome in breast cancer. Nat Med 2008; 14: 518–527.
Korkola JE, Houldsworth J, Chadalavada RS, Olshen AB, Dobrzynski D, Reuter VE et al. Down-regulation of stem cell genes, including those in a 200-kb gene cluster at 12p13.31, is associated with in vivo differentiation of human male germ cell tumors. Cancer Res 2006; 66: 820–827.
Gaspar C, Cardoso J, Franken P, Molenaar L, Morreau H, Moslein G et al. Cross-species comparison of human and mouse intestinal polyps reveals conserved mechanisms in adenomatous polyposis coli (APC)-driven tumorigenesis. Am J Pathol 2008; 172: 1363–1380.
Su LJ, Chang CW, Wu YC, Chen KC, Lin CJ, Liang SC et al. Selection of DDX5 as a novel internal control for Q-RT-PCR from microarray data using a block bootstrap re-sampling scheme. BMC Genomics 2007; 8: 140.
Iacobuzio-Donahue CA, Maitra A, Olsen M, Lowe AW, van Heek NT, Rosty C et al. Exploration of global gene expression patterns in pancreatic adenocarcinoma using cDNA microarrays. Am J Pathol 2003; 162: 1151–1162.
Wallace TA, Prueitt RL, Yi M, Howe TM, Gillespie JW, Yfantis HG et al. Tumor immunobiological differences in prostate cancer between African-American and European-American men. Cancer Res 2008; 68: 927–936.
Masters JR, Hepburn PJ, Walker L, Highman WJ, Trejdosiewicz LK, Povey S et al. Tissue culture model of transitional cell carcinoma: characterization of twenty-two human urothelial cell lines. Cancer Res 1986; 46: 3630–3636.
Nishiyama H, Takahashi T, Kakehi Y, Habuchi T, Knowles MA . Homozygous deletion at the 9q32-33 candidate tumor suppressor locus in primary human bladder cancer. Genes Chromosomes Cancer 1999; 26: 171–175.
Williams SV, Sibley KD, Davies AM, Nishiyama H, Hornigold N, Coulter J et al. Molecular genetic analysis of chromosome 9 candidate tumor-suppressor loci in bladder cancer cell lines. Genes Chromosomes Cancer 2002; 34: 86–96.
Shibayama T, Tachibana M, Deguchi N, Jitsukawa S, Tazaki H . SCID mice: a suitable model for experimental studies of urologic malignancies. J Urol 1991; 146: 1136–1137.
Hadaschik BA, Adomat H, Fazli L, Fradet Y, Andersen RJ, Gleave ME et al. Intravesical chemotherapy of high-grade bladder cancer with HTI-286, a synthetic analogue of the marine sponge product hemiasterlin. Clin Cancer Res 2008; 14: 1510–1518.
Konety BR, Lavelle JP, Pirtskalaishvili G, Dhir R, Meyers SA, Nguyen TS et al. Effects of vitamin D (calcitriol) on transitional cell carcinoma of the bladder in vitro and in vivo. J Urol 2001; 165: 253–258.
Hurst RE, Kyker KD, Bonner RB, Bowditch RD, Hemstreet III GP . Matrix-dependent plasticity of the malignant phenotype of bladder cancer cells. Anticancer Res 2003; 23: 3119–3128.
Hsieh JL, Wu CL, Lai MD, Lee CH, Tsai CS, Shiau AL . Gene therapy for bladder cancer using E1B-55 kD-deleted adenovirus in combination with adenoviral vector encoding plasminogen kringles 1-5. Br J Cancer 2003; 88: 1492–1499.
Adhim Z, Matsuoka T, Bito T, Shigemura K, Lee KM, Kawabata M et al. In vitro and in vivo inhibitory effect of three Cox-2 inhibitors and epithelial-to-mesenchymal transition in human bladder cancer cell lines. Br J Cancer 2011; 105: 393–402.
Matsui Y, Watanabe J, Ding S, Nishizawa K, Kajita Y, Ichioka K et al. Dicoumarol enhances doxorubicin-induced cytotoxicity in p53 wild-type urothelial cancer cells through p38 activation. BJU International 2010; 105: 558–564.
Kawato M, Shirakawa R, Kondo H, Higashi T, Ikeda T, Okawa K et al. Regulation of platelet dense granule secretion by the Ral GTPase-exocyst pathway. J Biol Chem 2008; 283: 166–174.
Ding S, Nishizawa K, Kobayashi T, Oishi S, Lv J, Fujii N et al. A potent chemotherapeutic strategy for bladder cancer: (S)-methoxy-trityl-L-cystein, a novel Eg5 inhibitor. J urol 2010; 184: 1175–1181.
Copeland NG, Jenkins NA, Court DL . Recombineering: a powerful new tool for mouse functional genomics. Nat Rev Genet 2001; 2: 769–779.
Kobayashi T, Inoue T, Shimizu Y, Terada N, Maeno A, Kajita Y et al. Activation of Rac1 is closely related to androgen-independent cell proliferation of prostate cancer cells both in vitro and in vivo. Mol endocrinol 2010; 24: 722–734.
Acknowledgements
We would like to thank Aaron Mathew Coutts for proofreading of the manuscript. We appreciate all members of the Cancer Research Course for Integrated Research Training in Kyoto University Graduate School of Medicine for their helpful advice and discussion. This study was supported by a Grant-in-Aid from the Ministry of Education, Culture, Sports, Science and Technology, Japan (No. 22591766 to HN and OO, 22890015, 22501009, 20013201 and 23113702 to R Shirakawa and HH) as well as grants from Uehara Memorial Foundation (to T Kobayashi and OO), Takeda Science Foundation, the Suzuken Memorial Foundation, Daiichi-Sankyo Foundation of Life-Science, Kurokawa Cancer Research Foundation (to R Shirakawa), and Novartis Foundation for the Promotion of Science (to HH).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Saito, R., Shirakawa, R., Nishiyama, H. et al. Downregulation of Ral GTPase-activating protein promotes tumor invasion and metastasis of bladder cancer. Oncogene 32, 894–902 (2013). https://doi.org/10.1038/onc.2012.101
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.101
Keywords
This article is cited by
-
Regulation of Ras p21 and RalA GTPases activity by quinine in mammary epithelial cells
Molecular and Cellular Biochemistry (2023)
-
Identification of single-nucleotide variants associated with susceptibility to Salmonella in pigs using a genome-wide association approach
BMC Veterinary Research (2020)
-
κB-Ras and Ral GTPases regulate acinar to ductal metaplasia during pancreatic adenocarcinoma development and pancreatitis
Nature Communications (2020)
-
Faithful preclinical mouse models for better translation to bedside in the field of immuno-oncology
International Journal of Clinical Oncology (2020)
-
The interaction between MALAT1 target, miR-143-3p, and RALGAPA2 is affected by functional SNP rs3827693 in breast cancer
Human Cell (2020)