Abstract
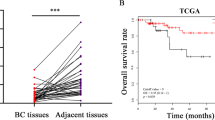
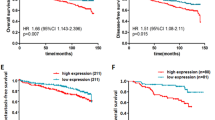
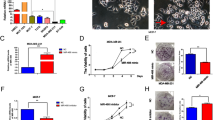
A micro-RNA, miR-155, is overexpressed in many types of cancer cells, including breast cancer, and its role(s) in tumor metastasis has been studied on a very limited basis. Tumor metastasis is a multi-step process with the last step in the process being formation of macroscopic tumor in organs distant from the primary tumor site. This step is the least studied. Here, we report that stable expression of miR-155 in 4T1 breast tumor cells reduces significantly the aggressiveness of tumor cell dissemination as a result of preventing epithelial-to-mesenchymal transition (EMT) of tumor cells in vivo. Further, miR-155 directly suppresses the expression of the transcription factor TCF4, which is an important regulator of EMT. However, when tumor cells are injected directly into the bloodstream, miR-155 remarkably promotes macroscopic tumor formation in the lung. Analysis of gene expression profiling identified a group of genes that are associated with promoting macroscopic tumor formation in the lung. Importantly, most of these genes are overexpressed in epithelial cells. Our findings provide new insight into how miR-155 modulates the development of tumor metastasis. This study suggests that the location of tumor cells overexpressing miR-155 is a critical factor: in mammary fat pads miR-155 prevents tumor dissemination; whereas in the lung miR-155 apparently maintains the epithelial phenotype of tumor cells that is critical for macroscopic tumor formation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
Accession codes
References
Acloque H, Adams MS, Fishwick K, Bronner-Fraser M, Nieto MA . (2009). Epithelial-mesenchymal transitions: the importance of changing cell state in development and disease. J Clin Invest 119: 1438–1449.
Aokage K, Ishii G, Ohtaki Y, Yamaguchi Y, Hishida T, Yoshida J et al. (2011). Dynamic molecular changes associated with epithelial-mesenchymal transition and subsequent mesenchymal-epithelial transition in the early phase of metastatic tumor formation. Int J Cancer 128: 1585–1595.
Bukholm IK, Nesland JM, Borresen-Dale AL . (2000). Re-expression of E-cadherin, alpha-catenin and beta-catenin, but not of gamma-catenin, in metastatic tissue from breast cancer patients [seecomments]. J Pathol 190: 15–19.
Chen CZ, Li L, Lodish HF, Bartel DP . (2004). MicroRNAs modulate hematopoietic lineage differentiation. Science 303: 83–86.
Cheng CW, Wu PE, Yu JC, Huang CS, Yue CT, Wu CW et al. (2001). Mechanisms of inactivation of E-cadherin in breast carcinoma: modification of the two-hit hypothesis of tumor suppressor gene. Oncogene 20: 3814–3823.
Chung KH, Hart CC, Al-Bassam S, Avery A, Taylor J, Patel PD et al. (2006). Polycistronic RNA polymerase II expression vectors for RNA interference based on BIC/miR-155. Nucleic Acids Res 34: e53.
Costinean S, Zanesi N, Pekarsky Y, Tili E, Volinia S, Heerema N et al. (2006). Pre-B cell proliferation and lymphoblastic leukemia/high-grade lymphoma in E(mu)-miR155 transgenic mice. Proc Natl Acad Sci USA 103: 7024–7029.
Eis PS, Tam W, Sun L, Chadburn A, Li Z, Gomez MF et al. (2005). Accumulation of miR-155 and BIC RNA in human B cell lymphomas. Proc Natl Acad Sci USA 102: 3627–3632.
Gallardo E, Navarro A, Vinolas N, Marrades RM, Diaz T, Gel B et al. (2009). miR-34a as a prognostic marker of relapse in surgically resected non-small-cell lung cancer. Carcinogenesis 30: 1903–1909.
Gibbons DL, Lin W, Creighton CJ, Rizvi ZH, Gregory PA, Goodall GJ et al. (2009). Contextual extracellular cues promote tumor cell EMT and metastasis by regulating miR-200 family expression. Genes Dev 23: 2140–2151.
Grizzle WE, Mountz JD, Yang PA, Xu X, Sun S, Van Zant GE et al. (2002). BXD recombinant inbred mice represent a novel T cell-mediated immune response tumor model. Int J Cancer 101: 270–279.
Han H-J, Russo J, Kohwi Y, Kohwi-Shigematsu T . (2008). SATB1 reprogrammes gene expression to promote breast tumour growth and metastasis. Nature 452: 187–193.
Hugo H, Ackland ML, Blick T, Lawrence MG, Clements JA, Williams ED et al. (2007). Epithelial--mesenchymal and mesenchymal--epithelial transitions in carcinoma progression. J Cell Physiol 213: 374–383.
Iorio MV, Ferracin M, Liu C-G, Veronese A, Spizzo R, Sabbioni S et al. (2005). MicroRNA gene expression deregulation in human breast cancer. Cancer Res 65: 7065–7070.
Iorns E, Hnatyszyn HJ, Seo P, Clarke J, Ward T, Lippman M . (2010). The role of SATB1 in breast cancer pathogenesis. J Natl Cancer Inst 102: 1284–1296.
Jiang S, Zhang H-W, Lu M-H, He X-H, Li Y, Gu H et al. (2010). MicroRNA-155 functions as an OncomiR in breast cancer by targeting the suppressor of cytokine signaling 1 gene. Cancer Res 70: 3119–3127.
Johnson MH . (2009). From mouse egg to mouse embryo: polarities, axes, and tissues. Annu Rev Cell Dev Biol 25: 483–512.
Katoh M . (2005). Epithelial-mesenchymal transition in gastric cancer (Review). Int J Oncol 27: 1677–1683.
Kong W, He L, Coppola M, Guo J, Esposito NN, Coppola D et al. (2010). MicroRNA-155 regulates cell survival, growth, and chemosensitivity by targeting FOXO3a in breast cancer. J Biol Chem 285: 17869–17879.
Kong W, Yang H, He L, Zhao JJ, Coppola D, Dalton WS et al. (2008). MicroRNA-155 is regulated by the transforming growth factor beta/Smad pathway and contributes to epithelial cell plasticity by targeting RhoA. Mol Cell Biol 28: 6773–6784.
Li J, Huang H, Sun L, Yang M, Pan C, Chen W et al. (2009). MiR-21 indicates poor prognosis in tongue squamous cell carcinomas as an apoptosis inhibitor. Clin Cancer Res 15: 3998–4008.
Logullo AF, Nonogaki S, Pasini FS, Osorio CA, Soares FA, Brentani MM . (2010). Concomitant expression of epithelial-mesenchymal transition biomarkers in breast ductal carcinoma: association with progression. Oncol Rep 23: 313–320.
Ma L, Young J, Prabhala H, Pan E, Mestdagh P, Muth D et al. (2010). miR-9, a MYC/MYCN-activated microRNA, regulates E-cadherin and cancer metastasis. Nat Cell Biol 12: 247–256.
Nakaya Y, Kuroda S, Katagiri YT, Kaibuchi K, Takahashi Y . (2004). Mesenchymal-epithelial transition during somitic segmentation is regulated by differential roles of Cdc42 and Rac1. Dev Cell 7: 425–438.
Nguyen DX, Bos PD, Massague J . (2009). Metastasis: from dissemination to organ-specific colonization. Nat Rev Cancer 9: 274–284.
Park S-M, Gaur AB, Lengyel E, Peter ME . (2008). The miR-200 family determines the epithelial phenotype of cancer cells by targeting the E-cadherin repressors ZEB1 and ZEB2. Genes Dev 22: 894–907.
Peinado H, Olmeda D, Cano A . (2007). Snail, Zeb and bHLH factors in tumour progression: an alliance against the epithelial phenotype? Nat Rev Cancer 7: 415–428.
Polyak K, Weinberg RA . (2009). Transitions between epithelial and mesenchymal states: acquisition of malignant and stem cell traits. Nat Rev Cancer 9: 265–273.
Pulaski BA, Ostrand-Rosenberg S . (2001). Mouse 4T1 breast tumor model. Current Protocols in Immunology, Vol. 4, Ch. 20. John Wiley: New York, p. 20.2.1.
Ryu JK, Hong SM, Karikari CA, Hruban RH, Goggins MG, Maitra A . (2010). Aberrant MicroRNA-155 expression is an early event in the multistep progression of pancreatic adenocarcinoma. Pancreatology 10: 66–73.
Sobrado VR, Moreno-Bueno G, Cubillo E, Holt LJ, Nieto MA, Portillo F et al. (2009). The class I bHLH factors E2-2A and E2-2B regulate EMT. J Cell Sci 122: 1014–1024.
Soltermann A, Tischler V, Arbogast S, Braun J, Probst-Hensch N, Weder W et al. (2008). Prognostic significance of epithelial-mesenchymal and mesenchymal-epithelial transition protein expression in non-small cell lung cancer. Clin Cancer Res 14: 7430–7437.
Stewart TJ, Abrams SI . (2007). Altered immune function during long-term host-tumor interactions can be modulated to retard autochthonous neoplastic growth. J Immunol 179: 2851–2859.
Thiery JP, Acloque H, Huang RYJ, Nieto MA . (2009). Epithelial-mesenchymal transitions in development and disease. Cell 139: 871–890.
Thompson EW, Williams ED . (2008). EMT and MET in carcinoma--clinical observations, regulatory pathways and new models. Clin Exp Metastasis 25: 591–592.
Tsuji T, Ibaragi S, Hu G-f . (2009a). Epithelial-Mesenchymal Transition and Cell Cooperativity in Metastasis. Cancer Res 69: 7135–7139.
Tsuji T, Ibaragi S, Hu GF . (2009b). Epithelial-mesenchymal transition and cell cooperativity in metastasis. Cancer Res 69: 7135–7139.
Tsuji T, Ibaragi S, Shima K, Hu MG, Katsurano M, Sasaki A et al. (2008). Epithelial-mesenchymal transition induced by growth suppressor p12CDK2-AP1 promotes tumor cell local invasion but suppresses distant colony growth. Cancer Res 68: 10377–10386.
Tusher VG, Tibshirani R, Chu G . (2001). Significance analysis of microarrays applied to the ionizing radiation response. Proc Natl Acad Sci USA 98: 5116–5121.
Wang B, Majumder S, Nuovo G, Kutay H, Volinia S, Patel T et al. (2009). Role of microRNA-155 at early stages of hepatocarcinogenesis induced by choline-deficient and amino acid-defined diet in C57BL/6 mice. Hepatology 50: 1152–1161.
Wells A, Yates C, Shepard CR . (2008). E-cadherin as an indicator of mesenchymal to epithelial reverting transitions during the metastatic seeding of disseminated carcinomas. Clin Exp Metastasis 25: 621–628.
Winter J, Jung S, Keller S, Gregory RI, Diederichs S . (2009). Many roads to maturity: microRNA biogenesis pathways and their regulation. Nat Cell Biol 11: 228–234.
Yanaihara N, Caplen N, Bowman E, Seike M, Kumamoto K, Yi M et al. (2006). Unique microRNA molecular profiles in lung cancer diagnosis and prognosis. Cancer Cell 9: 189–198.
Yang J, Mani SA, Donaher JL, Ramaswamy S, Itzykson RA, Come C et al. (2004). Twist, a Master Regulator of Morphogenesis, Plays an Essential Role in Tumor Metastasis. Cell 117: 927–939.
Zhao S, Fernald RD . (2005). Comprehensive algorithm for quantitative real-time polymerase chain reaction. J Comput Biol 12: 1047–1064.
Acknowledgements
We thank Dr David L Turner for providing plasmid UAS-luc-miR-155as, Dr David P Bartel for MDH1-PGK-GFP-miR-223 plasmid and Dr Jeffrey Schlom for the AT-3 breast tumor cell line. This work was supported by grants from the National Institutes of Health (NIH) (RO1CA137037, R01AT004294, RO1CA107181 and RO1CA116092), the Louisville Veterans Administration Medical Center (VAMC) Merit Review Grants (H-G Z) and a grant from the Susan G Komen Breast Cancer Foundation. We thank Dr Jerald Ainsworth for editorial assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
These authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Xiang, X., Zhuang, X., Ju, S. et al. miR-155 promotes macroscopic tumor formation yet inhibits tumor dissemination from mammary fat pads to the lung by preventing EMT. Oncogene 30, 3440–3453 (2011). https://doi.org/10.1038/onc.2011.54
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.54
Keywords
This article is cited by
-
Evaluation of paclitaxel-loaded polymeric nanoparticles in 3D tumor model: impact of tumor stroma on penetration and efficacy
Drug Delivery and Translational Research (2023)
-
Microarray profile analysis identifies ETS1 as potential biomarker regulated by miR-23b and modulates TCF4 in gastric cancer
World Journal of Surgical Oncology (2021)
-
MicroRNAs and Long Non-coding RNAs in Genetic Diseases
Molecular Diagnosis & Therapy (2019)
-
Relationship between epithelial-to-mesenchymal transition and the inflammatory microenvironment of hepatocellular carcinoma
Journal of Experimental & Clinical Cancer Research (2018)
-
miR-155 in cancer drug resistance and as target for miRNA-based therapeutics
Cancer and Metastasis Reviews (2018)