Key Points
-
The WHO 2016 classification of tumours of the urinary system highlighted the importance of careful morphological description of histological variants in patients with bladder cancer
-
Histological variants are divided in urothelial and nonurothelial subtypes and are present in almost 25% of bladder cancer samples in contemporary series
-
Histological variants present challenges in diagnosis, prognosis, and prediction of outcomes as their biological potential and clinical characteristics are highly variable
-
The percentage of a histological variant in a sample and its association with urothelial carcinoma should be reported by pathologists
-
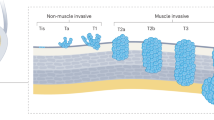
The presence of variant histology at transurethral resection of the bladder should always be evaluated and the management of bladder cancer should be tailored based on the clinical stage and histological variant present
-
The presence of histological variants in a radical cystectomy specimen is generally associated with adverse pathological features; however, no clear difference in survival outcomes compared with urothelial carcinoma has been reported
Abstract
In the past 10 years evidence for the clinical relevance of variant histology in urinary bladder cancer has been increasing. This increase has resulted in new classifications of urothelial cancers by the WHO in 2016, highlighting the importance of an accurate morphological description of pathological specimens for the therapeutic management of patients with bladder cancer. The rising awareness of the importance of an accurate pathological report manifests itself in the increasing prevalence of reporting of variant histology in daily practice. Histological variants can generally be divided into urothelial and nonurothelial. Urothelial variants often have similar features that also have specific morphological phenotypes, whereas nonurothelial variants have independent features. Overall, histological variants follow a more aggressive clinical course than conventional urothelial carcinoma, but conclusive data on their effect on survival are currently lacking. The clinical relevance of variant histology can manifest at three different levels: diagnostic, as identification is challenging and misinterpretation is not uncommon; prognostic, for patient risk stratification and outcome estimation; and therapeutic, as particular variants could be responsive to specific treatment strategies. An accurate morphological description of histological variants is necessary for patient consultation and therapy planning. Moreover, the association of variant histology with specific mutation patterns promises to be helpful in discovering targeted therapeutic approaches based on specific molecular pathways.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout













Similar content being viewed by others
References
Wasco, M. J. et al. Urothelial carcinoma with divergent histologic differentiation (mixed histologic features) predicts the presence of locally advanced bladder cancer when detected at transurethral resection. Urology 70, 69–74 (2007).
Cai, T. et al. Concordance and clinical significance of uncommon variants of bladder urothelial carcinoma in transurethral resection and radical cystectomy specimens. Urology 84, 1141–1146 (2014).
International Agency for Research on Cancer. WHO Classification of Tumours of the Urinary System and Male Genital Organs (eds Moch, H., Humphrey, P. A., Ulbright, T. M. & Reueter, V. E.) (WHO, 2016).
Shah, R. B., Montgomery, J. S., Montie, J. E. & Kunju, L. P. Variant (divergent) histologic differentiation in urothelial carcinoma is under-recognized in community practice: impact of mandatory central pathology review at a large referral hospital. Urol. Oncol. 31, 1650–1655 (2013).
Monn, M. F. et al. Contemporary bladder cancer: variant histology may be a significant driver of disease. Urol. Oncol. 33, 18.e15–18.e20 (2015).
Kaimakliotis, H. Z. et al. Plasmacytoid variant urothelial bladder cancer: is it time to update the treatment paradigm? Urol. Oncol. 32, 833–838 (2014).
Babjuk, M. et al. EAU guidelines on non-muscle-invasive urothelial carcinoma of the bladder: update 2016. Eur. Urol. 71, 447–461 (2017).
Alfred Witjes, J. et al. Updated 2016 EAU guidelines on muscle-invasive and metastatic bladder cancer. Eur. Urol. 71, 462–475 (2016).
Chang, S. S. et al. Treatment of non-metastatic muscle-invasive bladder cancer: AUA/ASCO/ASTRO/SUO guideline. J. Urol. http://dx.doi.org/10.1016/j.juro.2017.04.086 (2017).
Humphrey, P. A., Moch, H., Cubilla, A. L., Ulbright, T. M. & Reuter, V. E. The 2016 WHO classification of tumours of the urinary system and male genital organs-part B: prostate and bladder tumours. Eur. Urol. 70, 106–119 (2016).
Rice, K. R. et al. Lymph node metastases in patients with urothelial carcinoma variants: influence of the specific variant on nodal histology. Urol. Oncol. 33, 20.e23–20.e29 (2015).
Abol-Enein, H., Kava, B. R. & Carmack, A. J. K. Nonurothelial cancer of the bladder. Urology 69, 93–104 (2007).
Felix, A. S. et al. The changing patterns of bladder cancer in Egypt over the past 26 years. Cancer Causes Control 19, 421–429 (2008).
Guo, C. C. et al. Squamous cell carcinoma of the urinary bladder: a clinicopathologic and immunohistochemical study of 16 cases. Hum. Pathol. 40, 1448–1452 (2009).
Lopez-Beltran, A. & Cheng, L. Histologic variants of urothelial carcinoma: differential diagnosis and clinical implications. Hum. Pathol. 37, 1371–1388 (2006).
Ehdaie, B. et al. Comparative outcomes of pure squamous cell carcinoma and urothelial carcinoma with squamous differentiation in patients treated with radical cystectomy. J. Urol. 187, 74–79 (2012).
Lagwinski, N. et al. Squamous cell carcinoma of the bladder: a clinicopathologic analysis of 45 cases. Am. J. Surg. Pathol. 31, 1777–1787 (2007).
Rambau, P., Chalya, P. L. & Jackson, K. Schistosomiasis and urinary bladder cancer in North Western Tanzania: a retrospective review of 185 patients. Infecti. Agents Cancer 8, 19 (2013).
Salem, H. K. & Mahfouz, S. Changing patterns (age, incidence, and pathologic types) of schistosoma-associated bladder cancer in Egypt in the past decade. Urology 79, 379–383 (2012).
Mouli, S., Casalino, D. D. & Nikolaidis, P. Imaging features of common and uncommon bladder neoplasms. Radiol. Clin. North Amer. 50, 301–316 (2012).
Gaisa, N. T. et al. Different immunohistochemical and ultrastructural phenotypes of squamous differentiation in bladder cancer. Virchows Archiv: Int. J. Pathol. 458, 301–312 (2011).
el-Mekresh, M. M., el-Baz, M. A., Abol-Enein, H. & Ghoneim, M. A. Primary adenocarcinoma of the urinary bladder: a report of 185 cases. Br. J. Urol. 82, 206–212 (1998).
Zaghloul, M. S. et al. Long-term results of primary adenocarcinoma of the urinary bladder: a report on 192 patients. Urol. Oncol. 24, 13–20 (2006).
Grignon, D. J., Ro, J. Y., Ayala, A. G., Johnson, D. E. & Ordóñez, N. G. Primary adenocarcinoma of the urinary bladder. A clinicopathologic analysis of 72 cases. Cancer 67, 2165–2172 (1991).
Grignon, D. J., Ro, J. Y., Ayala, A. G. & Johnson, D. E. Primary signet-ring cell carcinoma of the urinary bladder. Am. J. Clin. Pathol. 95, 13–20 (1991).
Amin, M. B. Histological variants of urothelial carcinoma: diagnostic, therapeutic and prognostic implications. Mod. Pathol. 22 (Suppl. 2), S96–S118 (2009).
Kim, S. P. et al. The impact of squamous and glandular differentiation on survival after radical cystectomy for urothelial carcinoma. J. Urol. 188, 405–409 (2012).
Purnell, S., Sidana, A., Maruf, M., Grant, C. & Agarwal, P. K. Genitourinary paraganglioma: demographic, pathologic, and clinical characteristics in the surveillance, epidemiology, and end results database. Urol. Oncol. 35, 457.e9–457.e14 (2017).
Sved, P., Gomez, P., Manoharan, M., Civantos, F. & Soloway, M. S. Small cell carcinoma of the bladder. BJU Int. 94, 12–17 (2004).
Ploeg, M. et al. Clinical epidemiology of nonurothelial bladder cancer: analysis of the Netherlands Cancer Registry. J. Urol. 183, 915–920 (2010).
Koay, E. J., Teh, B. S., Paulino, A. C. & Butler, E. B. A. Surveillance, epidemiology, and end results analysis of small cell carcinoma of the bladder: epidemiology, prognostic variables, and treatment trends. Cancer 117, 5325–5333 (2011).
Abrahams, N. A., Moran, C., Reyes, A. O., Siefker-Radtke, A. & Ayala, A. G. Small cell carcinoma of the bladder: a contemporary clinicopathological study of 51 cases. Histopathology 46, 57–63 (2005).
Abbas, F., Civantos, F., Benedetto, P. & Soloway, M. S. Small cell carcinoma of the bladder and prostate. URL 46, 617–630 (1995).
Choong, N. W. W., Quevedo, J. F. & Kaur, J. S. Small cell carcinoma of the urinary bladder. The Mayo Clinic experience. Cancer 103, 1172–1178 (2005).
Mukesh, M., Cook, N., Hollingdale, A. E., Ainsworth, N. L. & Russell, S. G. Small cell carcinoma of the urinary bladder: a 15-year retrospective review of treatment and survival in the Anglian Cancer Network. BJU Int. 103, 747–752 (2009).
Trias, I. et al. Small cell carcinoma of the urinary bladder. Presentation of 23 cases and review of 134 published cases. Eur. Urol. 39, 85–90 (2001).
Cheng, L. et al. Small cell carcinoma of the urinary bladder: a clinicopathologic analysis of 64 patients. Cancer 101, 957–962 (2004).
Quek, M. L. et al. Radical cystectomy for primary neuroendocrine tumors of the bladder: the university of southern california experience. J. Urol. 174, 93–96 (2005).
Siefker-Radtke, A. O. et al. Evidence supporting preoperative chemotherapy for small cell carcinoma of the bladder: a retrospective review of the M. D. Anderson cancer experience. J. Urol. 172, 481–484 (2004).
Compérat, E. et al. Micropapillary urothelial carcinoma of the urinary bladder: a clinicopathological analysis of 72 cases. Pathology 42, 650–654 (2010).
Kamat, A. M. et al. Micropapillary bladder cancer: a review of the University of Texas M. D. Anderson Cancer Center experience with 100 consecutive patients. Cancer 110, 62–67 (2007).
Amin, M. B. et al. Micropapillary variant of transitional cell carcinoma of the urinary bladder. Histologic pattern resembling ovarian papillary serous carcinoma. Am. J. Surg. Pathol. 18, 1224–1232 (1994).
Sangoi, A. R. et al. Interobserver reproducibility in the diagnosis of invasive micropapillary carcinoma of the urinary tract among urologic pathologists. Am. J. Surg. Pathol. 34, 1367–1376 (2010).
Guo, C. C. et al. Gene expression profile of the clinically aggressive micropapillary variant of bladder cancer. Eur. Urol. 70, 611–620 (2016).
Watts, K. E. & Hansel, D. E. Emerging concepts in micropapillary urothelial carcinoma. Adv. Anatom. Pathol. 17, 182–186 (2010).
Summerhayes, I. C. et al. Clinical and pathological characteristics of micropapillary transitional cell carcinoma: a highly aggressive variant. J. Urol. 163, 748–751 (2000).
Kamat, A. M. et al. The case for early cystectomy in the treatment of nonmuscle invasive micropapillary bladder carcinoma. J. Urol. 175, 881–885 (2006).
Sui, W. et al. Micropapillary bladder cancer: insights from the National Cancer Database. Bladder Cancer 2, 415–423 (2016).
Spaliviero, M. et al. Clinical outcome of patients with T1 micropapillary urothelial carcinoma of the bladder. J. Urol. 192, 702–707 (2014).
Willis, D. L. et al. Micropapillary bladder cancer: current treatment patterns and review of the literature. Urol. Oncol. 32, 826–832 (2014).
Wright, J. L. et al. Differences in survival among patients with sarcomatoid carcinoma, carcinosarcoma and urothelial carcinoma of the bladder. J. Urol. 178, 2302–2306 (2007).
Cheng, L. et al. Sarcomatoid carcinoma of the urinary bladder: the final common pathway of urothelial carcinoma dedifferentiation. Am. J. Surg. Pathol. 35, e34–e46 (2011).
Jones, E. C. & Young, R. H. Myxoid and sclerosing sarcomatoid transitional cell carcinoma of the urinary bladder: a clinicopathologic and immunohistochemical study of 25 cases. Mod. Pathol. 10, 908–916 (1997).
Gronau, S. et al. Immunohistomorphologic and molecular cytogenetic analysis of a carcinosarcoma of the urinary bladder. Virchows Arch. 440, 436–440 (2002).
Lopez-Beltran, A., Luque, R. J., Mazzucchelli, R., Scarpelli, M. & Montironi, R. Changes produced in the urothelium by traditional and newer therapeutic procedures for bladder cancer. J. Clin. Pathol. 55, 641–647 (2002).
Mukhopadhyay, S., Shrimpton, A. E., Jones, L. A., Nsouli, I. S. & Abraham, N. Z. Carcinosarcoma of the urinary bladder following cyclophosphamide therapy: evidence for monoclonal origin and chromosome 9p allelic loss. Arch. Pathol. Lab. Med. 128, e8–e11 (2004).
Stamatiou, K. et al. Sarcomatoid carcinoma of the urinary bladder: a clinicopathological study of 4 cases and a review of the literature. Kor. J. Urol. 51, 724–728 (2010).
Wang, J., Wang, F. W., Lagrange, C. A., Hemstreet Iii, G. P. & Kessinger, A. Clinical features of sarcomatoid carcinoma (carcinosarcoma) of the urinary bladder: analysis of 221 cases. Sarcoma 2010, 454792 (2010).
Malla, M., Wang, J. F., Trepeta, R., Feng, A. & Wang, J. Sarcomatoid carcinoma of the urinary bladder. Clin. Genitourinary Cancer 14, 366–372 (2016).
Moschini, M. et al. Incidence and effect of variant histology on oncological outcomes in patients with bladder cancer treated with radical cystectomy. Urol. Oncol. 35, 335–341 (2017).
Lopez-Beltran, A., Requena, M. J., Montironi, R., Blanca, A. & Cheng, L. Plasmacytoid urothelial carcinoma of the bladder. Hum. Pathol. 40, 1023–1028 (2009).
Dayyani, F. et al. Plasmacytoid urothelial carcinoma, a chemosensitive cancer with poor prognosis, and peritoneal carcinomatosis. J. Urol. 189, 1656–1661 (2013).
Kaimakliotis, H. Z. et al. Plasmacytoid bladder cancer: variant histology with aggressive behavior and a new mode of invasion along fascial planes. Urology 83, 1112–1116 (2014).
Ricardo-Gonzalez, R. R. et al. Plasmacytoid carcinoma of the bladder: a urothelial carcinoma variant with a predilection for intraperitoneal spread. J. Urol. 187, 852–855 (2012).
Keck, B. et al. Plasmacytoid variant of bladder cancer defines patients with poor prognosis if treated with cystectomy and adjuvant cisplatin-based chemotherapy. BMC Cancer 13, 71 (2013).
Amin, M. B. et al. Lymphoepithelioma-like carcinoma of the urinary bladder. Am. J. Surg. Pathol. 18, 466–473 (1994).
Fadare, O., Renshaw, I. L. & Rubin, C. Pleomorphic lymphoepithelioma-like carcinoma of the urinary bladder. Int. J. Clin. Exp. Pathol. 2, 194–199 (2009).
Lopez-Beltran, A. et al. Lymphoepithelioma-like carcinoma of the urinary bladder: a clinicopathologic study of 13 cases. Virchows Archiv: Int. J. Pathol. 438, 552–557 (2001).
Williamson, S. R. et al. Lymphoepithelioma-like carcinoma of the urinary bladder: clinicopathologic, immunohistochemical, and molecular features. Am. J. Surg. Pathol. 35, 474–483 (2011).
Pantelides, N. et al. Lymphoepithelioma-like carcinoma of the urinary bladder: a case report and review of systemic treatment options. Urol. Ann. 4, 45 (2012).
Talbert, M. L. & Young, R. H. Carcinomas of the urinary bladder with deceptively benign-appearing foci. A report of three cases. Am. J. Surg. Pathol. 13, 374–381 (1989).
Young, R. H. & Oliva, E. Transitional cell carcinomas of the urinary bladder that may be underdiagnosed. A report of four invasive cases exemplifying the homology between neoplastic and non-neoplastic transitional cell lesions. Am. J. Surg. Pathol. 20, 1448–1454 (1996).
Beltran, A. L. et al. Clinicopathological characteristics and outcome of nested carcinoma of the urinary bladder. Virchows Arch. 465, 199–205 (2014).
Cox, R. & Epstein, J. I. Large nested variant of urothelial carcinoma: 23 cases mimicking von Brunn nests and inverted growth pattern of noninvasive papillary urothelial carcinoma. Am. J. Surg. Pathol. 35, 1337–1342 (2011).
Holmang, S. & Johansson, S. L. The nested variant of transitional cell carcinoma—a rare neoplasm with poor prognosis. Scand. J. Urol. Nephrol. 35, 102–105 (2001).
Wasco, M. J., Daignault, S., Bradley, D. & Shah, R. B. Nested variant of urothelial carcinoma: a clinicopathologic and immunohistochemical study of 30 pure and mixed cases. Hum. Pathol. 41, 163–171 (2010).
Linder, B. J. et al. Outcomes following radical cystectomy for nested variant of urothelial carcinoma: a matched cohort analysis. J. Urol. 189, 1670–1675 (2013).
Young, R. H. & Zukerberg, L. R. Microcystic transitional cell carcinomas of the urinary bladder. A report of four cases. Am. J. Clin. Pathol. 96, 635–639 (1991).
Lopez-Beltran, A., Montironi, R. & Cheng, L. Microcystic urothelial carcinoma: morphology, immunohistochemistry and clinical behaviour. Histopathology 64, 872–879 (2014).
Mai, K. T., Hakim, S. W., Ball, C. G., Flood, T. A. & Belanger, E. C. Nested and microcystic variants of urothelial carcinoma displaying immunohistochemical features of basal-like urothelial cells: an immunohistochemical and histopathogenetic study. Pathol. Int. 64, 375–381 (2014).
Lopez-Beltran, A., Blanca, A., Montironi, R., Cheng, L. & Regueiro, J. C. Pleomorphic giant cell carcinoma of the urinary bladder. Hum. Pathol. 40, 1461–1466 (2009).
Moch, H., Cubilla, A. L., Humphrey, P. A., Reuter, V. E. & Ulbright, T. M. The 2016 WHO classification of tumours of the urinary system and male genital organs-part A: renal, penile, and testicular tumours. Eur. Urol. 70, 93–105 (2016).
Lopez-Beltran, A. et al. Urothelial carcinoma of the bladder, lipid cell variant: clinicopathologic findings and LOH analysis. Am. J. Surg. Pathol. 34, 371–376 (2010).
Kojima, Y. et al. A case of urothelial carcinoma, lipid cell variant. Pathol. Int. 63, 183–187 (2013).
Yamashita, R., Yamaguchi, R., Yuen, K., Niwakawa, M. & Tobisu, K. Urothelial carcinoma (clear cell variant) diagnosed with useful immunohistochemistry stain. Int. J. Urol. 13, 1448–1450 (2006).
Rotellini, M., Fondi, C., Paglierani, M., Stomaci, N. & Raspollini, M. R. Clear cell carcinoma of the bladder in a patient with a earlier clear cell renal cell carcinoma: a case report with morphologic, immunohistochemical, and cytogenetical analysis. Appl. Immunohistochem. Mol. Morphol. 18, 396–399 (2010).
Young, R. H. & Scully, R. E. Clear cell adenocarcinoma of the bladder and urethra. A report of three cases and review of the literature. Am. J. Surg. Pathol. 9, 816–826 (1985).
Venyo, A. K.-G. Primary clear cell carcinoma of the urinary bladder. Int. Scholarly Res. Notices 2014, 1–15 (2014).
Adeniran, A. J. & Tamboli, P. Clear cell adenocarcinoma of the urinary bladder: a short review. Arch. Pathol. Lab. Med. 133, 987–991 (2009).
Moschini, M. et al. Pure but not mixed histologic variants are associated with poor survival at radical cystectomy in bladder cancer patients. Clin. Genitourin. Cancer http://dx.doi.org/10.1016/j.clgc.2016.12.006 (2016).
Manunta, A., Vincendeau, S., Kiriakou, G., Lobel, B. & Guillé, F. Non-transitional cell bladder carcinomas. BJU Int. 95, 497–502 (2005).
Izard, J. P. et al. Outcomes of squamous histology in bladder cancer: a population-based study. Urol. Oncol. 33, 425.e7–425.e13 (2015).
Kantor, A. F., Hartge, P., Hoover, R. N. & Fraumeni, J. F. Epidemiological characteristics of squamous cell carcinoma and adenocarcinoma of the bladder. Cancer Res. 48, 3853–3855 (1988).
Scosyrev, E., Yao, J. & Messing, E. Urothelial carcinoma versus squamous cell carcinoma of bladder: is survival different with stage adjustment? Urology 73, 822–827 (2009).
Abdollah, F. et al. Survival after radical cystectomy of non-bilharzial squamous cell carcinoma versus urothelial carcinoma: a competing-risks analysis. BJU Int. 109, 564–569 (2012).
Williamson, S. R., Lopez-Beltran, A., Montironi, R. & Cheng, L. Glandular lesions of the urinary bladder:clinical significance and differential diagnosis. Histopathology 58, 811–834 (2011).
Galsky, M. D. et al. Prospective trial of ifosfamide, paclitaxel, and cisplatin in patients with advanced non-transitional cell carcinoma of the urothelial tract. Urology 69, 255–259 (2007).
Lughezzani, G. et al. Adenocarcinoma versus urothelial carcinoma of the urinary bladder: comparison between pathologic stage at radical cystectomy and cancer-specific mortality. Urology 75, 376–381 (2010).
Mills, S. E. et al. Small cell undifferentiated carcinoma of the urinary bladder. A light-microscopic, immunocytochemical, and ultrastructural study of 12 cases. Am. J. Surg. Pathol. 11, 606–617 (1987).
Soria, F. et al. HER2 overexpression is associated with worse outcomes in patients with upper tract urothelial carcinoma (UTUC). World J. Urol. 35, 251–259 (2016).
Ross, J. S. et al. A high frequency of activating extracellular domain ERBB2 (HER2) mutation in micropapillary urothelial carcinoma. Clin. Cancer Res. 20, 68–75 (2014).
Ching, C. B. et al. HER2 gene amplification occurs frequently in the micropapillary variant of urothelial carcinoma: analysis by dual-color in situ hybridization. Mod. Pathol 24, 1111–1119 (2011).
Kim, B. et al. HER2 protein overexpression and gene amplification in plasmacytoid urothelial carcinoma of the urinary bladder. Dis. Markers 2016, 8463731–8463736 (2016).
Schneider, S. A. et al. Outcome of patients with micropapillary urothelial carcinoma following radical cystectomy: ERBB2 (HER2) amplification identifies patients with poor outcome. Mod. Pathol. 27, 758–764 (2014).
de Martino, M. et al. Impact of ERBB2 mutations on in vitro sensitivity of bladder cancer to lapatinib. Cancer Biol. Ther. 15, 1239–1247 (2014).
Wülfing, C. et al. A single-arm, multicenter, open-label phase 2 study of lapatinib as the second-line treatment of patients with locally advanced or metastatic transitional cell carcinoma. Cancer 115, 2881–2890 (2009).
Lim, M. G., Adsay, N. V., Grignon, D. J. & Osunkoya, A. O. E-Cadherin expression in plasmacytoid, signet ring cell and micropapillary variants of urothelial carcinoma: comparison with usual-type high-grade urothelial carcinoma. Mod. Pathol. 24, 241–247 (2011).
Al-Ahmadie, H. A. et al. Frequent somatic CDH1 loss-of-function mutations in plasmacytoid variant bladder cancer. Nat. Genet. 48, 356–358 (2016).
Fong, A. et al. Expression of caveolin-1 and caveolin-2 in urothelial carcinoma of the urinary bladder correlates with tumor grade and squamous differentiation. Am. J. Clin. Pathol. 120, 93–100 (2003).
Babjuk, M. et al. EAU guidelines non-muscle-invasive urothelial carcinoma bladder: update 2013. Eur. Urol. 64, 639–653 (2013).
Abd El-Latif, A., Watts, K. E., Elson, P., Fergany, A. & Hansel, D. E. The sensitivity of initial transurethral resection or biopsy of bladder tumor(s) for detecting bladder cancer variants on radical cystectomy. J. Urol. 189, 1263–1267 (2013).
Weizer, A. Z. et al. Multiple adverse histological features increase the odds of under staging T1 bladder cancer. J. Urol. 182, 59–65 (2009).
Ge, P., Wang, Z.-C., Yu, X., Lin, J. & He, Q. Sensitivity of initial biopsy or transurethral resection of bladder tumor(s) for detecting histological variants on radical cystectomy. BMC Urol. 15, 46 (2015).
Burger, M. et al. Photodynamic diagnosis of non-muscle-invasive bladder cancer with hexaminolevulinate cystoscopy: a meta-analysis of detection and recurrence based on raw data. Eur. Urol. 64, 846–854 (2013).
Naito, S. et al. The clinical research office of the endourological society (CROES) multicentre randomised trial of narrow band imaging-assisted transurethral resection of bladder tumour (TURBT) versus conventional white light imaging-assisted TURBT in primary non-muscle-invasive bladder cancer patients: trial protocol and 1-year results. Eur. Urol. 70, 506–515 (2016).
Shapur, N. K. et al. Is radical cystectomy mandatory in every patient with variant histology of bladder cancer. Rare Tumors 3, e22 (2011).
Gofrit, O. N. et al. The response of variant histology bladder cancer to intravesical immunotherapy compared to conventional cancer. Front. Oncol. 6, 43 (2016).
Willis, D. L. et al. Clinical outcomes of cT1 micropapillary bladder cancer. J. Urol. 193, 1129–1134 (2015).
Ghoneim, I. A. et al. Neoadjuvant systemic therapy or early cystectomy? Single-center analysis of outcomes after therapy for patients with clinically localized micropapillary urothelial carcinoma of the bladder. Urology 77, 867–870 (2011).
Vourganti, S. et al. Low grade micropapillary urothelial carcinoma, does it exist? - Analysis of management and outcomes from the Surveillance, Epidemiology and End Results (SEER) Database. J. Cancer 4, 336–342 (2013).
Miller, J. S. & Epstein, J. I. Noninvasive urothelial carcinoma of the bladder with glandular differentiation: report of 24 cases. Am. J. Surg. Pathol. 33, 1241–1248 (2009).
Chan, T. Y. & Epstein, J. I. In situ adenocarcinoma of the bladder. Am. J. Surg. Pathol. 25, 892–899 (2001).
Seisen, T., Compérat, E., Léon, P. & Rouprêt, M. Impact of histological variants on the outcomes of nonmuscle invasive bladder cancer after transurethral resection. Curr. Opin. Urol. 24, 524–531 (2014).
Bansal, A., Kumar, N. & Sharma, S. C. Sarcomatoid variant of urothelial carcinoma of the urinary bladder. J. Cancer Res. Ther. 9, 571–573 (2016).
Fatima, N. et al. Sarcomatoid urothelial carcinoma of the bladder: a contemporary clinicopathologic analysis of 37 cases. Can. J. Urol. 22, 7783–7787 (2015).
Raspollini, M. R. et al. Plasmacytoid urothelial carcinoma of the urinary bladder: clinicopathologic, immunohistochemical, ultrastructural, and molecular analysis of a case series. Hum. Pathol. 42, 1149–1158 (2011).
Xylinas, E. et al. Impact of histological variants on oncological outcomes of patients with urothelial carcinoma of the bladder treated with radical cystectomy. Eur. J. Cancer. 49, 1889–1897 (2013).
Fairey, A. S. et al. Impact of micropapillary urothelial carcinoma variant histology on survival after radical cystectomy. Urol. Oncol. 32, 110–116 (2014).
Monn, M. F. et al. The changing reality of urothelial bladder cancer: should non-squamous variant histology be managed as a distinct clinical entity? BJU Int. 116, 236–240 (2015).
Ismaili, N. A rare bladder cancer - small cell carcinoma: review and update. Orphanet J. Rare Diseases 6, 75 (2011).
Grossman, H. B. et al. Neoadjuvant chemotherapy plus cystectomy compared with cystectomy alone for locally advanced bladder cancer. N. Engl. J. Med. 349, 859–866 (2003).
International Collaboration of Trialists. International phase III trial assessing neoadjuvant cisplatin, methotrexate, and vinblastine chemotherapy for muscle-invasive bladder cancer: long-term results of the BA06 30894 trial. J. Clin. Oncol. 29, 2171–2177 (2011).
Pokuri, V. K. et al. Predictors of complete pathologic response (pT0) to neoadjuvant chemotherapy in muscle-invasive bladder carcinoma. Clin. Genitourinary Cancer 14, e59–e65 (2016).
Culp, S. H. et al. Refining patient selection for neoadjuvant chemotherapy before radical cystectomy. J. Urol. 191, 40–47 (2014).
Moschini, M. et al. Validation of preoperative risk grouping of the selection of patients most likely to benefit from neoadjuvant chemotherapy before radical cystectomy. Clin. Genitourinary Cancer 15, e267–e273 (2016).
Black, P. C., Brown, G. A. & Dinney, C. P. N. The impact of variant histology on the outcome of bladder cancer treated with curative intent. Urol. Oncol. 27, 3–7 (2009).
Meeks, J. J. et al. Pathological response to neoadjuvant chemotherapy for muscle-invasive micropapillary bladder cancer. BJU Int. 111, E325–E330 (2013).
Kassouf, W. et al. Outcome and patterns of recurrence of nonbilharzial pure squamous cell carcinoma of the bladder: a contemporary review of the university of Texas M. D. Anderson cancer center experience. Cancer 110, 764–769 (2007).
Gad el Mawla, N. et al. A randomized pilot study of high-dose epirubicin as neoadjuvant chemotherapy in the treatment of cancer of the bilharzial bladder. Ann. Oncol. 2, 137–140 (1991).
Scosyrev, E. et al. Do mixed histological features affect survival benefit from neoadjuvant platinum-based combination chemotherapy in patients with locally advanced bladder cancer? A secondary analysis of Southwest Oncology Group-Directed Intergroup Study (S8710). BJU Int. 108, 693–699 (2011).
Lynch, S. P. et al. Neoadjuvant chemotherapy in small cell urothelial cancer improves pathologic downstaging and long-term outcomes: results from a retrospective study at the MD Anderson Cancer Center. Eur. Urol. 64, 307–313 (2013).
Siefker-Radtke, A. O. et al. Phase II clinical trial of neoadjuvant alternating doublet chemotherapy with ifosfamide/doxorubicin and etoposide/cisplatin in small-cell urothelial cancer. J. Clin. Oncol. 27, 2592–2597 (2009).
Bex, A., de Vries, R., Pos, F., Kerst, M. & Horenblas, S. Long-term survival after sequential chemoradiation for limited disease small cell carcinoma of the bladder. World J. Urol. 27, 101–106 (2009).
Schreiber, D. et al. Characterization and outcomes of small cell carcinoma of the bladder using the surveillance, epidemiology, and end results database. Am. J. Clin. Oncol. 36, 126–131 (2013).
Masson-Lecomte, A. et al. Oncological outcomes of advanced muscle-invasive bladder cancer with a micropapillary variant after radical cystectomy and adjuvant platinum-based chemotherapy. World J. Urol. 33, 1087–1093 (2015).
Sui, W. et al. Contemporary treatment patterns and outcomes of sarcomatoid bladder cancer. World J. Urol. 35, 1055–1061 (2016).
Willis, D. L., Porten, S. P. & Kamat, A. M. Should histologic variants alter definitive treatment of bladder cancer? Curr. Opin. Urol. 23, 435–443 (2013).
Author information
Authors and Affiliations
Contributions
M.M., D.D., S.K. Y.I. and E.C. researched data for the article. S.F.S., M.M., D.D'A. and F. S. made substantial contributions to discussions of content. S.F.S., M.M. and D.D'A. wrote the manuscript. M.M., D.D'A., S.K., F.S., E.C. and S.F.S. reviewed and edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
PowerPoint slides
Rights and permissions
About this article
Cite this article
Moschini, M., D'Andrea, D., Korn, S. et al. Characteristics and clinical significance of histological variants of bladder cancer. Nat Rev Urol 14, 651–668 (2017). https://doi.org/10.1038/nrurol.2017.125
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrurol.2017.125
This article is cited by
-
Determining the clinicopathological significance of the VI-RADS ≧4 group: a retrospective study
BMC Urology (2024)
-
CT and MRI features of sarcomatoid urothelial carcinoma of the bladder and its differential diagnosis with conventional urothelial carcinoma
Cancer Imaging (2024)
-
One Size Fits Some: Approaching Rare Malignancies of the Urinary Tract
Current Treatment Options in Oncology (2024)
-
Predicting individual outcomes after radical cystectomy in urothelial variants with Cancer of the Bladder Risk Assessment (COBRA) score
World Journal of Urology (2024)
-
The prognostic significance of histologic variant on survival outcomes in patients with metastatic urothelial carcinoma receiving immune checkpoint inhibitor therapy
BMC Cancer (2023)