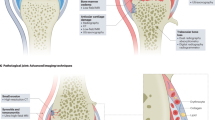
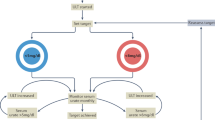
Abstract
The clinical course of gout is initially characterized by acute self-limited joint inflammation, but long-standing disease is often associated with chronic inflammation followed by the development of erosive joint damage, which can result in long-term functional impairment. Preventing joint damage is now a major focus of therapeutic intervention in gout. New light has been shed on the mechanisms leading to cartilage and bone damage in patients with this disease. Here, we discuss basic science studies focusing on the cellular immunology of bone and cartilage in gout and the effects of monosodium urate crystals on signaling pathways, cytokine release and the function of osteoclasts, osteoblasts and chondrocytes. We then explore the use of advanced imaging modalities (including MRI, ultrasonography, CT and dual-energy CT) to investigate pathology in gout, as they provide new ways to visualize joint tissues. These modalities vary in their ability to detect the various pathological features of gout and have different clinical applications. Imaging provides information about the inflammatory nature of the joint lesion, position and size of tophaceous deposits, and extent of bone and cartilage damage. Imaging is also increasingly being used to monitor the progression of joint damage and regression of tophi with effective urate-lowering therapy.
Key Points
-
Bone erosion in patients with gout is likely mediated by activated osteoclasts
-
Osteoblast viability and function are inhibited by contact with monosodium urate crystals
-
Chondrocytes might contribute to cartilage damage in patients with gout
-
Advanced imaging techniques, including MRI, CT, ultrasound and dual-energy CT, allow improved detection of joint damage and tophi
-
Tophus volume can be measured using these modalities and can be used to monitor urate-lowering therapy
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Edwards, N. L. et al. Work productivity loss due to flares in patients with chronic gout refractory to conventional therapy. J. Med. Econ. 14, 10–15 (2011).
Singh, J. A. et al. Patient-reported outcomes in chronic gout: a report from OMERACT 10. J. Rheumatol. 38, 1452–1457 (2011).
Dalbeth, N. et al. Tophaceous joint disease strongly predicts hand function in patients with gout. Rheumatology (Oxford) 46, 1804–1807 (2007).
Rome, K. et al. Functional and biomechanical characteristics of foot disease in chronic gout: a case–control study. Clin. Biomech. 26, 90–94 (2011).
Levin, M. H., Lichtenstein, L. & Scott, H. W. Pathologic changes in gout; survey of eleven necropsied cases. Am. J. Pathol. 32, 871–895 (1956).
Guerra, J. & Resnick, D. Arthritides affecting the foot: radiographic–pathological correlation. Foot Ankle 2, 325–331 (1982).
Sokoloff, L. The pathology of gout. Metabolism 6, 230–243 (1957).
Palmer, D. G. et al. Comparison of phenotype expression by mononuclear phagocytes within subcutaneous gouty tophi and rheumatoid nodules. Rheumatol. Int. 7, 187–193 (1987).
Schweyer, S., Hemmerlein, B., Radzun, H. J. & Fayyazi, A. Continuous recruitment, co-expression of tumour necrosis factor-α and matrix metalloproteinases, and apoptosis of macrophages in gout tophi. Virchows Arch. 437, 534–539 (2000).
Martinon, F., Pétrilli, V., Mayor, A., Tardivel, A. & Tschopp, J. Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature 440, 237–241 (2006).
Chen, C.-J. et al. MyD88-dependent IL-1 receptor signaling is essential for gouty inflammation stimulated by monosodium urate crystals. J. Clin. Invest. 116, 2262–2271 (2006).
Becker, M. A. et al. The urate-lowering efficacy and safety of febuxostat in the treatment of the hyperuricemia of gout: the CONFIRMS trial. Arthritis Res. Ther. 12, R63 (2010).
Schlesinger, N. et al. Canakinumab reduces the risk of acute gouty arthritis flares during initiation of allopurinol treatment: results of a double-blind, randomised study. Ann. Rheum. Dis. 70, 1264–1271 (2011).
Schlesinger, N. & Thiele, R. G. The pathogenesis of bone erosions in gouty arthritis. Ann. Rheum. Dis. 69, 1907–1912 (2010).
Dalbeth, N. et al. Cellular characterization of the gouty tophus: a quantitative analysis. Arthritis Rheum. 62, 1549–1556 (2010).
Alwan, W. H., Dieppe, P. A., Elson, C. J. & Bradfield, J. W. Hydroxyapatite and urate crystal induced cytokine release by macrophages. Ann. Rheum. Dis. 48, 476–482 (1989).
Pouliot, M., James, M. J., McColl, S. R., Naccache, P. H. & Cleland, L. G. Monosodium urate microcrystals induce cyclooxygenase-2 in human monocytes. Blood 91, 1769–1776 (1998).
Olmsted-Davis, E. Cox-2 RANKs high in osteoclastogenesis. Blood 106, 1144–1145 (2005).
Di Giovine, F. S., Malawista, S. E., Nuki, G. & Duff, G. W. Interleukin 1 (IL 1) as a mediator of crystal arthritis. Stimulation of T cell and synovial fibroblast mitogenesis by urate crystal-induced IL 1. J. Immunol. 138, 3213–3218 (1987).
Zwerina, J. et al. TNF-induced structural joint damage is mediated by IL-1. Proc. Natl Acad. Sci. USA 104, 11742–11747 (2007).
Kim, J. H. et al. The mechanism of osteoclast differentiation induced by IL-1. J. Immunol. 183, 1862–1870 (2009).
Kusano, K. et al. Regulation of matrix metalloproteinases (MMP-2, -3, -9, and -13) by interleukin-1 and interleukin-6 in mouse calvaria: association of MMP induction with bone resorption. Endocrinology 139, 1338–1345 (1998).
Goldring, S. R. Bone and joint destruction in rheumatoid arthritis: what is really happening? J. Rheumatol. Suppl. 65, 44–48 (2002).
Dempster, D. W. in Primer on the Metabolic Bone Diseases and Disorders of Mineral Metabolism (eds Favus, M. J. & Bikle, D. D.) 7–11 (The American Society for Bone and Mineral Research, Washington, DC, 2006).
Diarra, D. et al. Dickkopf-1 is a master regulator of joint remodeling. Nat. Med. 13, 156–163 (2007).
Bromley, M. & Woolley, D. E. Chondroclasts and osteoclasts at subchondral sites of erosion in the rheumatoid joint. Arthritis Rheum. 27, 968–975 (1984).
Dalbeth, N. et al. Circulating mediators of bone remodeling in psoriatic arthritis: implications for disordered osteoclastogenesis and bone erosion. Arthritis Res. Ther. 12, R164 (2010).
Boyce, B. F. & Xing, L. Biology of RANK, RANKL, and osteoprotegerin. Arthritis Res. Ther. 9 (Suppl. 1), S1 (2007).
Dalbeth, N. et al. Enhanced osteoclastogenesis in patients with tophaceous gout: urate crystals promote osteoclast development through interactions with stromal cells. Arthritis Rheum 58, 1854–1865 (2008).
Ritchlin, C. T., Haas-Smith, S. A., Li, P., Hicks, D. G. & Schwarz, E. M. Mechanisms of TNF-α- and RANKL-mediated osteoclastogenesis and bone resorption in psoriatic arthritis. J. Clin. Invest. 111, 821–831 (2003).
Choe, J.-Y., Lee, G. H. & Kim, S.-K. Radiographic bone damage in chronic gout is negatively associated with the inflammatory cytokines soluble interleukin 6 receptor and osteoprotegerin. J. Rheumatol. 38, 485–491 (2011).
Schett, G., Hayer, S., Zwerina, J., Redlich, K. & Smolen, J. S. Mechanisms of Disease: the link between RANKL and arthritic bone disease. Nat. Clin. Pract. Rheumatol. 1, 47–54 (2005).
Barthelemy, C. R., Nakayama, D. A., Carrera, G. F., Lightfoot, R. W. Jr & Wortmann, R. L. Gouty arthritis: a prospective radiographic evaluation of sixty patients. Skeletal Radiol. 11, 1–8 (1984).
McQueen, F. M. et al. High-grade MRI bone oedema is common within the surgical field in rheumatoid arthritis patients undergoing joint replacement and is associated with osteitis in subchondral bone. Ann. Rheum. Dis. 66, 1581–1587 (2007).
Poh, Y., Dalbeth, N., Doyle, A. & McQueen, F. M. Magnetic resonance imaging bone edema is not a major feature of gout unless there is concomitant osteomyelitis: 10-year findings from a high-prevalence population. J. Rheumatol. 38, 2475–2481 (2011).
McQueen, F. & Naredo, E. The 'disconnect' between synovitis and erosion in rheumatoid arthritis: a result of treatment or intrinsic to the disease process itself? Ann. Rheum. Dis. 70, 241–244 (2011).
McQueen, F. M. A vital clue to deciphering bone pathology: MRI bone oedema in rheumatoid arthritis and osteoarthritis. Ann. Rheum. Dis. 66, 1549–1552 (2007).
Chhana, A. et al. Monosodium urate monohydrate crystals inhibit osteoblast viability and function: implications for development of bone erosion in gout. Ann. Rheum. Dis. 70, 1684–1691 (2011).
Bouchard, L., de Médicis, R., Lussier, A., Naccache, P. H. & Poubelle, P. E. Inflammatory microcrystals alter the functional phenotype of human osteoblast-like cells in vitro: synergism with IL-1 to overexpress cyclooxygenase-2. J. Immunol. 168, 5310–5317 (2002).
Allaeys, I. et al. Osteoblast retraction induced by adherent neutrophils promotes osteoclast bone resorption: implication for altered bone remodeling in chronic gout. Lab. Invest. 91, 905–920 (2011).
Goldring, M. B. Update on the biology of the chondrocyte and new approaches to treating cartilage diseases. Best Pract. Res. Clin. Rheumatol. 20, 1003–1025 (2006).
Goldring, M. B. Human chondrocyte cultures as models of cartilage-specific gene regulation. Methods Mol. Med. 2, 217–232 (1996).
Roddy, E., Zhang, W. & Doherty, M. Are joints affected by gout also affected by osteoarthritis? Ann. Rheum. Dis. 66, 1374–1377 (2007).
Muehleman, C. et al. Association between crystals and cartilage degeneration in the ankle. J. Rheumatol. 35, 1108–1117 (2008).
Denoble, A. E. et al. Uric acid is a danger signal of increasing risk for osteoarthritis through inflammasome activation. Proc. Natl. Acad. Sci. USA 108, 2088–2093 (2011).
Roddy, E. Revisiting the pathogenesis of podagra: why does gout target the foot? J. Foot Ankle Res. 4, 13 (2011).
Katz, W. A. & Schubert, M. The interaction of monosodium urate with connective tissue components. J. Clin. Invest. 49, 1783–1789 (1970).
Laurent, T. C. Solubility of sodium urate in the presence of chondroitin-4-sulphate. Nature 202, 1334 (1964).
Burt, H. M. & Dutt, Y. C. Growth of monosodium urate monohydrate crystals: effect of cartilage and synovial fluid components on in vitro growth rates. Ann. Rheum. Dis. 45, 858–864 (1986).
Shi, Y. Caught red-handed: uric acid is an agent of inflammation. J. Clin. Invest. 120, 1809–1811 (2010).
Barksby, H. E. et al. Matrix metalloproteinase 10 promotion of collagenolysis via procollagenase activation: implications for cartilage degradation in arthritis. Arthritis Rheum. 54, 3244–3253 (2006).
Dreier, R., Grässel, S., Fuchs, S., Schaumburger, J. & Bruckner, P. Pro-MMP-9 is a specific macrophage product and is activated by osteoarthritic chondrocytes via MMP-3 or a MT1-MMP/MMP-13 cascade. Exp. Cell Res. 297, 303–312 (2004).
Goldring, M. B. & Marcu, K. B. Cartilage homeostasis in health and rheumatic diseases. Arthritis Res. Ther. 11, 224 (2009).
Villalobo, A. Nitric oxide and cell proliferation. FEBS J. 273, 2329–2344 (2006).
Liu-Bryan, R., Pritzker, K., Firestein, G. S. & Terkeltaub, R. TLR2 signaling in chondrocytes drives calcium pyrophosphate dihydrate and monosodium urate crystal-induced nitric oxide generation. J. Immunol. 174, 5016–5023 (2005).
Liu, R., Lioté, F., Rose, D. M., Merz, D. & Terkeltaub, R. Proline-rich tyrosine kinase 2 and Src kinase signaling transduce monosodium urate crystal-induced nitric oxide production and matrix metalloproteinase 3 expression in chondrocytes. Arthritis Rheum. 50, 247–258 (2004).
Popa-Nita, O. & Naccache, P. H. Crystal-induced neutrophil activation. Immunol. Cell Biol. 88, 32–40 (2010).
Lee, H.-S., Lee, C.-H., Tsai, H.-C. & Salter, D. M. Inhibition of cyclooxygenase 2 expression by diallyl sulfide on joint inflammation induced by urate crystal and IL-1β. Osteoarthritis Cartilage 17, 91–99 (2009).
Buckley, T. J. Radiologic features of gout. Am. Fam. Physician 54, 1232–1238 (1996).
Schlesinger, N. Diagnosis of gout: clinical, laboratory, and radiologic findings. Am. J. Manag. Care 11, 443–450 (2005).
Resnick, D. & Broderick, T. W. Intraosseous calcifications in tophaceous gout. AJR Am. J. Roentgenol. 137, 1157–1161 (1981).
Dalbeth, N., Clark, B., McQueen, F., Doyle, A. & Taylor, W. Validation of a radiographic damage index in chronic gout. Arthritis Rheum. 57, 1067–1073 (2007).
Rettenbacher, T. et al. Diagnostic imaging of gout: comparison of high-resolution US versus conventional X-ray. Eur. Radiol. 18, 621–630 (2008).
Wright, S. A. et al. High-resolution ultrasonography of the first metatarsal phalangeal joint in gout: a controlled study. Ann. Rheum. Dis. 66, 859–864 (2007).
Carter, J. D. et al. An analysis of MRI and ultrasound imaging in patients with gout who have normal plain radiographs. Rheumatology (Oxford) 48, 1442–1446 (2009).
Resnick, D. Crystal-induced arthropathy. Gout and pseudogout. JAMA 242, 2440–2442 (1979).
Peterfy, C. Structural characterization of rheumatoid arthritis by MRI: applications in clinical research and in clinical practice. J. Musculoskelet. Neuronal Interact. 4, 360–361 (2004).
Ko, K.-H., Hsu, Y.-C., Lee, H.-S., Lee, C.-H. & Huang, G.-S. Tophaceous gout of the knee: revisiting MRI patterns in 30 patients. J. Clin. Rheumatol. 16, 209–214 (2010).
Gentili, A. The advanced imaging of gouty tophi. Curr. Rheumatol. Rep. 8, 231–235 (2006).
Ryu, K. et al. Characteristic appearance of large subcutaneous gouty tophi in magnetic resonance imaging. Mod. Rheumatol. 15, 290–293 (2005).
Popp, J. D., Bidgood, W. D. Jr & Edwards, N. L. Magnetic resonance imaging of tophaceous gout in the hands and wrists. Semin. Arthritis Rheum. 25, 282–289 (1996).
Schumacher, H. R. Jr. et al. Magnetic resonance imaging in the quantitative assessment of gouty tophi. Int. J. Clin. Pract. 60, 408–414 (2006).
Perez-Ruiz, F. & Naredo, E. Imaging modalities and monitoring measures of gout. Curr. Opin. Rheumatol. 19, 128–133 (2007).
Hodgson, R. et al. Dynamic contrast enhanced MRI of bone marrow oedema in rheumatoid arthritis. Ann. Rheum. Dis. 67, 270–272 (2008).
Dalbeth, N. et al. Cellular characterisation of magnetic resonance imaging bone oedema in rheumatoid arthritis; implications for pathogenesis of erosive disease. Ann. Rheum. Dis. 68, 279–282 (2009).
McQueen, F. M. et al. Bone edema scored on magnetic resonance imaging scans of the dominant carpus at presentation predicts radiographic joint damage of the hands and feet six years later in patients with rheumatoid arthritis. Arthritis Rheum. 48, 1814–1827 (2003).
Benton, N. et al. MRI of the wrist in early rheumatoid arthritis can be used to predict functional outcome at 6 years. Ann. Rheum. Dis. 63, 555–561 (2004).
Paparo, F. et al. Imaging of tophi with an extremity-dedicated MRI system. Clin. Exp. Rheumatol. 29, 519–526 (2011).
Yu, J. S., Chung, C., Recht, M., Dailiana, T. & Jurdi, R. MR imaging of tophaceous gout. AJR Am. J. Roentgenol. 168, 523–527 (1997).
Conaghan, P. Is MRI useful in osteoarthritis? Best Pract. Res. Clin. Rheumatol. 20, 57–68 (2006).
Thiele, R. G. & Schlesinger, N. Diagnosis of gout by ultrasound. Rheumatology (Oxford) 46, 1116–1121 (2007).
Schueller-Weidekamm, C., Schueller, G., Aringer, M., Weber, M. & Kainberger, F. Impact of sonography in gouty arthritis: comparison with conventional radiography, clinical examination, and laboratory findings. Eur. J. Radiol. 62, 437–443 (2007).
Carter, J. D. et al. An analysis of MRI and ultrasound imaging in patients with gout who have normal plain radiographs. Rheumatology (Oxford) 48, 1442–1446 (2009).
McQueen, F. et al. Pitfalls in scoring MR images of rheumatoid arthritis wrist and metacarpophalangeal joints. Ann. Rheum. Dis. 64 (Suppl. 1), i48–i55 (2005).
Perez-Ruiz, F., Dalbeth, N., Urresola, A., de Miguel, E. & Schlesinger, N. Imaging of gout: findings and utility. Arthritis Res. Ther. 11, 232 (2009).
Slot, O. & Terslev, L. Ultrasonographic signs of gout in symmetric polyarthritis. Arthritis Rheum. 62, 3487 (2010).
Howard, R. G. et al. Reproducibility of musculoskeletal ultrasound for determining monosodium urate deposition: concordance between readers. Arthritis Care Res. (Hoboken) 63, 1456–1462 (2011).
Filippucci, E., Riveros, M. G., Georgescu, D., Salaffi, F. & Grassi, W. Hyaline cartilage involvement in patients with gout and calcium pyrophosphate deposition disease. An ultrasound study. Osteoarthritis Cartilage 17, 178–181 (2009).
Pineda, C. et al. Joint and tendon subclinical involvement suggestive of gouty arthritis in asymptomatic hyperuricemia: an ultrasound controlled study. Arthritis Res. Ther. 13, R4 (2011).
De Miguel E. et al. Diagnosis of gout in patients with asymptomatic hyperuricaemia: a pilot ultrasound study. Ann. Rheum. Dis. 71, 157–158 (2012).
Thiele, R. G. & Schlesinger, N. Ultrasonography shows disappearance of monosodium urate crystal deposition on hyaline cartilage after sustained normouricemia is achieved. Rheumatol. Int. 30, 495–503 (2010).
de Ávila Fernandes, E. et al. Ultrasound features of tophi in chronic tophaceous gout. Skeletal Radiol. 40, 309–315 (2011).
Perez-Ruiz, F., Martin, I. & Canteli, B. Ultrasonographic measurement of tophi as an outcome measure for chronic gout. J. Rheumatol. 34, 1888–1893 (2007).
Boers, M., Brooks, P., Strand, C. V. & Tugwell, P. The OMERACT filter for Outcome Measures in Rheumatology. J. Rheumatol. 25, 198–199 (1998).
Dalbeth, N. et al. Methods of tophus assessment in clinical trials of chronic gout: a systematic literature review and pictorial reference guide. Ann. Rheum. Dis. 70, 597–604 (2011).
Gerster, J., Landry, M., Dufresne, L. & Meuwly, J. Y. Imaging of tophaceous gout: computed tomography provides specific images compared with magnetic resonance imaging and ultrasonography. Ann. Rheum. Dis. 61, 52–54 (2002).
Dalbeth, N. et al. Computed tomography measurement of tophus volume: comparison with physical measurement. Arthritis Rheum. 57, 461–465 (2007).
Dalbeth, N. et al. Mechanisms of bone erosion in gout: a quantitative analysis using plain radiography and computed tomography. Ann. Rheum. Dis. 68, 1290–1295 (2009).
Desai, M. A., Peterson, J. J., Garner, H. W. & Kransdorf, M. J. Clinical utility of dual-energy CT for evaluation of tophaceous gout. Radiographics 31, 1365–1375 (2011).
Choi, H. K. et al. Dual energy computed tomography in tophaceous gout. Ann. Rheum. Dis. 68, 1609–1612 (2009).
Nicolaou, S. et al. Dual-energy CT as a potential new diagnostic tool in the management of gout in the acute setting. AJR Am. J. Roentgenol. 194, 1072–1078 (2010).
McQueen, F. M., Doyle, A. & Dalbeth, N. Imaging in gout: what can we learn from MRI, CT, DECT and US? Arthritis Res. Ther. 13, 246 (2011).
Author information
Authors and Affiliations
Contributions
All authors contributed equally to researching data for the article, discussing the content, writing and reviewing the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
McQueen, F., Chhana, A. & Dalbeth, N. Mechanisms of joint damage in gout: evidence from cellular and imaging studies. Nat Rev Rheumatol 8, 173–181 (2012). https://doi.org/10.1038/nrrheum.2011.207
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2011.207
This article is cited by
-
Factors secreted by monosodium urate crystal-stimulated macrophages promote a proinflammatory state in osteoblasts: a potential indirect mechanism of bone erosion in gout
Arthritis Research & Therapy (2022)
-
Association of serum uric acid with clinical and radiological severity of knee osteoarthritis in non-gouty patients
Egyptian Rheumatology and Rehabilitation (2021)
-
Laser treatment of synovial inflammatory process in experimentally induced microcrystalline arthritis in Wistar rats
Lasers in Medical Science (2021)
-
Effects of low-level laser therapy on the organization of articular cartilage in an experimental microcrystalline arthritis model
Lasers in Medical Science (2019)
-
Oridonin is a covalent NLRP3 inhibitor with strong anti-inflammasome activity
Nature Communications (2018)