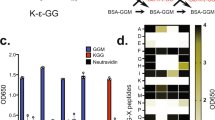
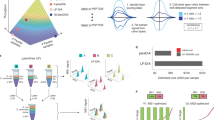
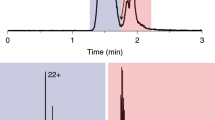
Abstract
Ubiquitination can have profound effects on the stability and function of cellular proteins. Mass spectrometry (MS) can be used to map the specific amino acid residues that are conjugated to ubiquitin in a target protein. However, the purification required for proteomic analysis can be challenging. In this paper, we describe a bimolecular affinity purification scheme for the isolation of a specific ubiquitinated protein in which affinity moieties are fused to ubiquitin and to a target protein of interest. After ubiquitin conjugation in vivo, the protein target acquires two affinity tags, allowing the specific purification of its ubiquitin-modified forms. To prevent deubiquitination after lysis or the copurification of interacting cofactors, this procedure is performed after protein denaturation using polyhistidine and biotinylation tags. Using this procedure, the ubiquitinated forms of a given protein can be efficiently purified in large amounts of sufficient purity for MS analysis and for mapping of ubiquitin acceptor sites.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Weissman, A.M. Themes and variations on ubiquitylation. Nat. Rev. Mol. Cell Biol. 2, 169–178 (2001).
Wilkinson, C.R. New tricks for ubiquitin and friends. Trends Cell Biol. 12, 545–546 (2002).
Deshaies, R.J. & Joazeiro, C.A. RING domain E3 ubiquitin ligases. Annu. Rev. Biochem. 78, 399–434 (2009).
Rotin, D. & Kumar, S. Physiological functions of the HECT family of ubiquitin ligases. Nat. Rev. Mol. Cell Biol. 10, 398–409 (2009).
Bloom, J., Amador, V., Bartolini, F., DeMartino, G. & Pagano, M. Proteasome-mediated degradation of p21 via N-terminal ubiquitinylation. Cell 115, 71–82 (2003).
Ben-Saadon, R. et al. The tumor suppressor protein p16(INK4a) and the human papillomavirus oncoprotein-58 E7 are naturally occurring lysine-less proteins that are degraded by the ubiquitin system. Direct evidence for ubiquitination at the N-terminal residue. J. Biol. Chem. 279, 41414–41421 (2004).
Breitschopf, K., Bengal, E., Ziv, T., Admon, A. & Ciechanover, A. A novel site for ubiquitination: the N-terminal residue, and not internal lysines of MyoD, is essential for conjugation and degradation of the protein. EMBO J. 17, 5964–5973 (1998).
Peng, J. et al. A proteomics approach to understanding protein ubiquitination. Nat. Biotechnol. 21, 921–926 (2003).
Tokunaga, F. et al. Involvement of linear polyubiquitylation of NEMO in NF-κB activation. Nat. Cell Biol. 11, 123–132 (2009).
Finley, D. Recognition and processing of ubiquitin-protein conjugates by the proteasome. Annu. Rev. Biochem. 78, 477–513 (2009).
Aguilar, R.C. & Wendland, B. Ubiquitin: not just for proteasomes anymore. Curr. Opin. Cell Biol. 15, 184–190 (2003).
Rahighi, S. et al. Specific recognition of linear ubiquitin chains by NEMO is important for NF-κB activation. Cell 136, 1098–1109 (2009).
Rodriguez, M.S., Desterro, J.M., Lain, S., Lane, D.P. & Hay, R.T. Multiple C-terminal lysine residues target p53 for ubiquitin-proteasome-mediated degradation. Mol. Cell Biol. 20, 8458–8467 (2000).
Ea, C.K., Deng, L., Xia, Z.P., Pineda, G. & Chen, Z.J. Activation of IKK by TNFα requires site-specific ubiquitination of RIP1 and polyubiquitin binding by NEMO. Mol. Cell 22, 245–257 (2006).
Lee, J.S., Hong, U.S., Lee, T.H., Yoon, S.K. & Yoon, J.B. Mass spectrometric analysis of tumor necrosis factor receptor-associated factor 1 ubiquitination mediated by cellular inhibitor of apoptosis 2. Proteomics 4, 3376–3382 (2004).
Witze, E.S., Old, W.M., Resing, K.A. & Ahn, N.G. Mapping protein post-translational modifications with mass spectrometry. Nat. Methods 4, 798–806 (2007).
Denis, N.J., Vasilescu, J., Lambert, J.P., Smith, J.C. & Figeys, D. Tryptic digestion of ubiquitin standards reveals an improved strategy for identifying ubiquitinated proteins by mass spectrometry. Proteomics 7, 868–874 (2007).
Seyfried, N.T. et al. Systematic approach for validating the ubiquitinated proteome. Anal. Chem. 80, 4161–4169 (2008).
Nielsen, M.L. et al. Iodoacetamide-induced artifact mimics ubiquitination in mass spectrometry. Nat. Methods 5, 459–460 (2008).
Huang, F., Kirkpatrick, D., Jiang, X., Gygi, S. & Sorkin, A. Differential regulation of EGF receptor internalization and degradation by multiubiquitination within the kinase domain. Mol. Cell 21, 737–748 (2006).
Shin, H. et al. Identification of ubiquitination sites on the X-linked inhibitor of apoptosis protein. Biochem. J. 373, 965–971 (2003).
Maine, G.N., Gluck, N., Zaidi, I.W. & Burstein, E. Bimolecular affinity purification (BAP): tandem affinity purification using two protein baits. Cold Spring Harb. Protoc. doi10.1101/pdb.prot5318 (2009).
Mao, X. et al. GCN5 is a required cofactor for a ubiquitin ligase that targets NF-κB/RelA. Genes Dev. 23, 849–861 (2009).
Brady, G.F. et al. Regulation of the copper chaperone CCS by XIAP-mediated ubiquitination. Mol. Cell Biol. 30, 1923–1936 (2010).
Mayor, T. & Deshaies, R.J. Two-step affinity purification of multiubiquitylated proteins from Saccharomyces cerevisiae. Methods Enzymol. 399, 385–392 (2005).
Tagwerker, C. et al. A tandem-affinity tag for two-step purification under fully denaturing conditions: application in ubiquitin profiling and protein complex identification combined with in vivo cross-linking. Mol. Cell Proteomics 5, 366–378 (2006).
Ota, K., Kito, K., Iemura, S., Natsume, T. & Ito, T. A parallel affinity purification method for selective isolation of polyubiquitinated proteins. Proteomics 8, 3004–3007 (2008).
Hershko, A. & Rose, I.A. Ubiquitin-aldehyde: a general inhibitor of ubiquitin-recycling processes. Proc. Natl. Acad. Sci. USA 84, 1829–1833 (1987).
Hjerpe, R. et al. Efficient protection and isolation of ubiquitylated proteins using tandem ubiquitin-binding entities. EMBO Rep. 10, 1250–1258 (2009).
Basso, M. et al. Insoluble mutant SOD1 is partly oligoubiquitinated in amyotrophic lateral sclerosis mice. J. Biol. Chem. 281, 33325–33335 (2006).
Habelhah, H. et al. Ubiquitination and translocation of TRAF2 is required for activation of JNK but not of p38 or NF-κB. EMBO J. 23, 322–332 (2004).
Monia, B.P. et al. Gene synthesis, expression, and processing of human ubiquitin carboxyl extension proteins. J. Biol. Chem. 264, 4093–4103 (1989).
Hunter, A.M. et al. A novel ubiquitin fusion system bypasses the mitochondria and generates biologically active Smac/DIABLO. J. Biol. Chem. 278, 7494–7499 (2003).
Xu, M., Skaug, B., Zeng, W. & Chen, Z.J. A ubiquitin replacement strategy in human cells reveals distinct mechanisms of IKK activation by TNFα and IL-1β. Mol. Cell 36, 302–314 (2009).
Caron, C., Boyault, C. & Khochbin, S. Regulatory cross-talk between lysine acetylation and ubiquitination: role in the control of protein stability. Bioessays 27, 408–415 (2005).
Schenborn, E.T. & Goiffon, V. Calcium phosphate transfection of mammalian cultured cells. Methods Mol. Biol. 130, 135–145 (2000).
Narindrasorasak, S., Kulkarni, P., Deschamps, P., She, Y.M. & Sarkar, B. Characterization and copper binding properties of human COMMD1 (MURR1). Biochemistry Mosc. 46, 3116–3128 (2007).
Xu, P. et al. Quantitative proteomics reveals the function of unconventional ubiquitin chains in proteasomal degradation. Cell 137, 133–145 (2009).
Acknowledgements
This work was supported by an R01 DK073639 grant and the University of Texas DOCS Award to E.B., and by an R01 DE19249 grant to K.S.J.-E.J. We thank J. Strahler and M. Hurley at the Michigan Proteome Consortium for their initial assistance in sample analysis.
Author information
Authors and Affiliations
Contributions
E.B. conceived and supervised the project. The specific tandem affinity purification strategy used here was devised by G.N.M. and E.B. The initial experimental conditions were determined by G.N.M., and subsequent repeats of the purification scheme, including some of the data shown here for COMMD1, were performed by I.W.Z. and H.L. V.B. and K.S.J.-E.J. contributed to the mass spectrometry analysis of COMMD1.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Maine, G., Li, H., Zaidi, I. et al. A bimolecular affinity purification method under denaturing conditions for rapid isolation of a ubiquitinated protein for mass spectrometry analysis. Nat Protoc 5, 1447–1459 (2010). https://doi.org/10.1038/nprot.2010.109
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2010.109
This article is cited by
-
IL-17-driven intestinal fibrosis is inhibited by Itch-mediated ubiquitination of HIC-5
Mucosal Immunology (2018)
-
Ubiquitination involved enzymes and cancer
Medical Oncology (2014)
-
Regulation of NF-κB activity by competition between RelA acetylation and ubiquitination
Oncogene (2012)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.