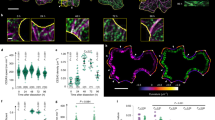
Abstract
The plant actin cytoskeleton is an unstable network of filaments that influences polarized growth through poorly understood mechanisms. Here, we used a combination of live cell imaging and finite element computational modelling of Arabidopsis trichome morphogenesis to determine how the actin and microtubule cytoskeletons cooperate to pattern the cell wall and growth. The actin-related protein (ARP)2/3 complex generates an actin meshwork that operates within a tip-localized, microtubule-depleted zone to modulate cell wall anisotropy locally. The actin meshwork also positions an actin bundle network that organizes organelle flow patterns. This activity is required to maintain cell wall thickness gradients that enable tip-biased diffuse growth. These newly discovered couplings between cytoskeletal patterns and wall textures provide important insights into the cellular mechanism of growth control in plants.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Rodriguez, O. C. et al. Conserved microtubule-actin interactions in cell movement and morphogenesis. Nature Cell Biol. 5, 599–609 (2003).
Smith, L. G. & Oppenheimer, D. G. Spatial control of cell expansion by the plant cytoskeleton. Annu. Rev. Cell Dev. Biol. 21, 271–295 (2005).
Baskin, T. I. Anisotropic expansion of the plant cell wall. Annu. Rev. Cell Dev. Biol. 21, 203–222 (2005).
Fayant, P. et al. Finite element model of polar growth in pollen tubes. Plant Cell 22, 2579–2593 (2010).
Szymanski, D. B. & Cosgrove, D. J. Dynamic coordination of cytoskeletal and cell wall systems during plant cell morphogenesis. Curr. Biol. 19, R800–811 (2009).
Zhang, C., Halsey, L. E. & Szymanski, D. B. The development and geometry of shape change in Arabidopsis thaliana cotyledon pavement cells. BMC Plant Biol. 11, 27 (2011).
Andriankaja, M. et al. Exit from proliferation during leaf development in Arabidopsis thaliana: a not-so-gradual process. Dev. Cell 22, 64–78 (2012).
Walter, A., Silk, W. K. & Schurr, U. Environmental effects on spatial and temporal patterns of leaf and root growth. Annu. Rev. Plant Biol. 60, 279–304 (2009).
Paredez, A. R., Somerville, C. R. & Ehrhardt, D. W. Visualization of cellulose synthase demonstrates functional association with microtubules. Science 312, 1491–1495 (2006).
Fujita, M. et al. The anisotropy1 D604N mutation in the Arabidopsis cellulose synthase1 catalytic domain reduces cell wall crystallinity and the velocity of cellulose synthase complexes. Plant Physiol. 162 (2013).
Mauricio, R. Costs of resistance to natural enemies in field populations of the annual plant Arabidopsis thaliana. Am. Nat. 151, 20–28 (1998).
Tiwari, S. C. & Wilkins, T. A. Cotton (Gossypium hirsutum) seed trichomes expand via diffuse growing mechanism. Can. J. Bot. 73, 746–757 (1995).
Szymanski, D. B., Marks, M. D. & Wick, S. M. Organized F-actin is essential for normal trichome morphogenesis in Arabidopsis. Plant Cell 11, 2331–2347 (1999).
Mathur, J., Spielhofer, P., Kost, B. & Chua, N. The actin cytoskeleton is required to elaborate and maintain spatial patterning during trichome cell morphogenesis in Arabidopsis thaliana. Development 126, 5559–5568 (1999).
Zhang, C. et al. Arabidopsis SCARs function interchangeably to meet actin-related protein 2/3 activation thresholds during morphogenesis. Plant Cell 20, 995–1011 (2008).
Mathur, J. et al. Arabidopsis CROOKED encodes for the smallest subunit of the ARP2/3 complex and controls cell shape by region specific fine F-actin formation. Development 130, 3137–3146 (2003).
Le, J., El-Assal, S. E., Basu, D., Saad, M. E. & Szymanski, D. B. Requirements for Arabidopsis ATARP2 and ATARP3 during epidermal development. Curr. Biol. 13, 1341–1347 (2003).
Yanagisawa, M., Zhang, C. & Szymanski, D. B. ARP2/3-dependent growth in the plant kingdom: SCARs for life. Front. Plant Sci. 4, 166 (2013).
Deeks, M. J., Kaloriti, D., Davies, B., Malho, R. & Hussey, P. J. Arabidopsis NAP1 is essential for ARP2/3-dependent trichome morphogenesis. Curr. Biol. 14, 1410–1414 (2004).
Zhang, X., Dyachok, J., Krishnakumar, S., Smith, L. G. & Oppenheimer, D. G. IRREGULAR TRICHOME BRANCH1 in Arabidopsis encodes a plant homolog of the actin-related protein2/3 complex activator Scar/WAVE that regulates actin and microtubule organization. Plant Cell 17, 2314–2326 (2005).
Sambade, A., Findlay, K., Schaffner, A. R., Lloyd, C. W. & Buschmann, H. Actin-dependent and -independent functions of cortical microtubules in the differentiation of Arabidopsis leaf trichomes. Plant Cell 26 (2014).
Schwab, B. et al. Regulation of cell expansion by the DISTORTED genes in Arabidopsis thaliana: actin controls the spatial organization of microtubules. Mol. Genet. Genome 269, 350–360 (2003).
Beilstein, M. & Szymanski, D. in The Plant Cytoskeleton in Cell Differentiation and Development (ed. Hussey, P. ) 265–289 (Blackwell, 2004).
Gasser, T. C., Ogden, R. W. & Holzapfel, G. A. Hyperelastic modelling of arterial layers with distributed collagen fibre orientations. J. R. Soc. Interface 3, 15–35 (2006).
Huang, R., Becker, A. A. & Jones, I. A. Modelling cell wall growth using a fibre-reinforced hyperelastic-viscoplastic constitutive law. J. Mech. Phys. Solids 60, 750–783 (2012).
McKenna, S. T. et al. Exocytosis precedes and predicts the increase in growth in oscillating pollen tubes. Plant Cell 21, 3026–3040 (2009).
Rounds, C. M., Lubeck, E., Hepler, P. K. & Winship, L. J. Propidium iodide competes with Ca(2+) to label pectin in pollen tubes and Arabidopsis root hairs. Plant Physiol. 157, 175–187 (2011).
Timoshenko, S. Strength of materials (New York, 1930).
Basu, D., Le, J., Zakharova, T., Mallery, E. L. & Szymanski, D. B. A SPIKE1 signaling complex controls actin-dependent cell morphogenesis through the heteromeric WAVE and ARP2/3 complexes. Proc. Natl Acad. Sci. USA 105, 4044–4049 (2008).
Basu, D., El-Assal, S. E., Le, J., Mallery, E. L. & Szymanski, D. B. Interchangeable functions of Arabidopsis PIROGI and the human WAVE complex subunit SRA1 during leaf epidermal development. Development 131, 4345–4355 (2004).
Basu, D. et al. DISTORTED3/SCAR2 is a putative Arabidopsis WAVE complex subunit that activates the Arp2/3 complex and is required for epidermal morphogenesis. Plant Cell 17, 502–524 (2005).
Kotchoni, S. O. et al. The association of the Arabidopsis actin-related protein (ARP) 2/3 complex with cell membranes is linked to its assembly status, but not its activation. Plant Physiol. 151, 2095–2109 (2009).
El-Assal, S. E., Le, J., Basu, D., Mallery, E. L. & Szymanski, D. B. DISTORTED2 encodes an ARPC2 subunit of the putative Arabidopsis ARP2/3 complex. Plant J. 38, 526–538 (2004).
Sampathkumar, A. et al. Patterning and lifetime of plasma membrane-localized cellulose synthase is dependent on actin organization in Arabidopsis interphase cells. Plant Physiol. 162, 675–688 (2013).
Lu, L., Lee, Y.-R. J., Pan, R., Maloof, J. N. & Liu, B. An internal motor kinesin is associated with the Golgi apparatus and plays a role in trichome morphogenesis in Arabidopsis. Mol. Biol. Cell 16, 811–823 (2005).
Oda, Y. & Fukuda, H. Initiation of cell wall pattern by a Rho- and microtubule-driven symmetry breaking. Science 337, 1333–1336 (2012).
Pollard, T. D. & Borisy, G. G. Cellular motility driven by assembly and disassembly of actin filaments. Cell 112, 453–465 (2003).
McCollum, D., Feoktistova, A., Morphew, M., Balasubramanian, M. & Gould, K. L. The Schizosaccharomyces pombe actin-related protein, Arp3, is a component of the cortical actin cytoskeleton and interacts with profilin. EMBO J. 15, 6438–6446 (1996).
Winter, D., Podtelejnikov, A. V., Mann, M. & Li, R. The complex containing actin-related proteins Arp2 and Arp3 is required for the motility and integrity of yeast actin patches. Curr. Biol. 7, 519–529 (1997).
Evangelista, M., Pruyne, D., Amberg, D. C., Boone, C. & Bretscher, A. Formins direct Arp2/3-independent actin filament assembly to polarize cell growth in yeast. Nature Cell Biol. 4, 260–269 (2002).
Le, J., Mallery, E. L., Zhang, C., Brankle, S. & Szymanski, D. B. Arabidopsis BRICK1/HSPC300 is an essential WAVE-complex subunit that selectively stabilizes the Arp2/3 activator SCAR2. Curr. Biol. 16, 895–901 (2006).
Djakovic, S., Dyachok, J., Burke, M., Frank, M. J. & Smith, L. G. BRICK1/HSPC300 functions with SCAR and the ARP2/3 complex to regulate epidermal cell shape in Arabidopsis. Development 133, 1091–1100 (2006).
Dyachok, J. et al. Plasma membrane-associated SCAR complex subunits promote cortical F-actin accumulation and normal growth characteristics in Arabidopsis roots. Mol. Plant 1, 990–1006 (2008).
Zhang, C., Mallery, E. & Szymanski, D. B. ARP2/3 localization in Arabidopsis leaf pavement cells: a diversity of intracellular pools and cytoskeletal interactions. Front. Plant Biol. 4, 1–16 (2013).
El-Assal, S. E., Le, J., Basu, D., Mallery, E. L. & Szymanski, D. B. Arabidopsis GNARLED encodes a NAP125 homologue that positively regulates ARP2/3. Curr. Biol. 14, 1405–1409 (2004).
Ojangu, E. L. et al. Myosins XI-K, XI-1, and XI-2 are required for development of pavement cells, trichomes, and stigmatic papillae in Arabidopsis. BMC Plant Biol. 12, 81 (2012).
Fendrych, M. et al. The Arabidopsis exocyst complex is involved in cytokinesis and cell plate maturation. Plant Cell 22, 3053–3065 (2010).
Boudaoud, A. An introduction to the mechanics of morphogenesis for plant biologists. Trends Plant Sci. 15, 353–360 (2010).
Szymanski, D. B. The kinematics and mechanics of leaf expansion: new pieces to the Arabidopsis puzzle. Curr. Opin. Plant Biol. 22C, 141–148 (2014).
Butterworth, K. M., Adams, D. C., Horner, H. T. & Wendel, J. F. Initiation and early development of fiber in wild and cultivated cotton. Int. J. Plant Sci. 170, 561–574 (2009).
Forouzesh, E., Goel, A., Mackenzie, S. A. & Turner, J. A. In vivo extraction of Arabidopsis cell turgor pressure using nanoindentation in conjunction with finite element modeling. Plant J. 73, 509–520 (2013).
Acknowledgements
We thank David Umulis for helpful discussions. Thanks to Adam Fessenden for reflected light time-lapse imaging. We also thank Chia-Ping Huang and the Purdue Life Science Microscopy Facility for their expert assistance. This research was supported by NSF Grant IOS Grant No. 1249652 to D.B.S and J.A.T. and NSF MCB Grant No. 1121893 to D.B.S.
Author information
Authors and Affiliations
Contributions
All authors participated in the experimental design, data collection, data analysis and manuscript preparation. D.S. conceived the project. J.T. and A.D. developed the finite element models. S.B. did the reflected light and TEM analyses. E.M. created the ARP2/3–GFP live-cell probe. M.Y. conducted all other wet laboratory experiments.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Yanagisawa, M., Desyatova, A., Belteton, S. et al. Patterning mechanisms of cytoskeletal and cell wall systems during leaf trichome morphogenesis. Nature Plants 1, 15014 (2015). https://doi.org/10.1038/nplants.2015.14
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nplants.2015.14
This article is cited by
-
Probing stress-regulated ordering of the plant cortical microtubule array via a computational approach
BMC Plant Biology (2023)
-
ARP2/3 complex associates with peroxisomes to participate in pexophagy in plants
Nature Plants (2023)
-
A comprehensive overview of cotton genomics, biotechnology and molecular biological studies
Science China Life Sciences (2023)
-
CRISPR-Cas9 Arabidopsis mutants of genes for ARPC1 and ARPC3 subunits of ARP2/3 complex reveal differential roles of complex subunits
Scientific Reports (2022)
-
TOR signaling is the potential core of conserved regulation of trichome development in plant
Acta Physiologiae Plantarum (2022)