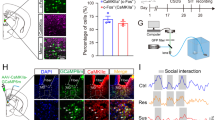
Abstract
Postsynaptic remodeling of glutamatergic synapses on ventral striatum (vSTR) medium spiny neurons (MSNs) is critical for shaping stress responses. However, it is unclear which presynaptic inputs are involved. Susceptible mice exhibited increased synaptic strength at intralaminar thalamus (ILT), but not prefrontal cortex (PFC), inputs to vSTR MSNs following chronic social stress. Modulation of ILT-vSTR versus PFC-vSTR neuronal activity differentially regulated dendritic spine plasticity and social avoidance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Christoffel, D.J., Golden, S.A. & Russo, S.J. Rev. Neurosci. 22, 535–549 (2011).
Russo, S.J. & Nestler, E.J. Nat. Rev. Neurosci. 14, 609–625 (2013).
Golden, S.A. et al. Nat. Med. 19, 337–344 (2013).
Christoffel, D.J. et al. J. Neurosci. 31, 314–321 (2011).
Britt, J.P. et al. Neuron 76, 790–803 (2012).
Stuber, G.D. et al. Nature 475, 377–380 (2011).
Covington, H.E. III et al. J. Neurosci. 30, 16082–16090 (2010).
Bubser, M. & Deutch, A.Y. Synapse 32, 13–22 (1999).
Browning, J.R., Jansen, H.T. & Sorg, B.A. Drug Alcohol Depend. 134, 387–390 (2014).
Chen, B.T. et al. Nature 496, 359–362 (2013).
Penzo, M.A. et al. Nature 519, 455–459 (2015).
Fremeau, R.T. Jr. et al. Neuron 31, 247–260 (2001).
Berton, O. et al. Science 311, 864–868 (2006).
Krishnan, V. et al. Cell 131, 391–404 (2007).
Golden, S.A., Covington, H.E. III, Berton, O. & Russo, S.J. Nat. Protoc. 6, 1183–1191 (2011).
Auer, S. et al. Nat. Methods 7, 229–236 (2010).
Kim, S.-Y.Y. et al. Nature 496, 219–223 (2013).
Vialou, V. et al. J. Neurosci. 34, 3878–3887 (2014).
Tye, K.M. et al. Nature 493, 537–541 (2013).
Chaudhury, D. et al. Nature 493, 532–536 (2013).
Burger, C. et al. Mol. Ther. 10, 302–317 (2004).
Sparta, D.R. et al. Nat. Protoc. 7, 12–23 (2012).
Radley, J.J. et al. Cereb. Cortex 16, 313–320 (2006).
Rodriguez, A., Ehlenberger, D.B., Dickstein, D.L., Hof, P.R. & Wearne, S.L. PLoS One 3, e1997 (2008).
Acknowledgements
This research was supported by US National Institute of Mental Health grants R01 MH090264 (S.J.R.), R01 MH092306 (M.-H.H.), P50 MH096890 (S.J.R. and M.-H.H.), T32 GM089626 (D.J.C.), T32 MH087004 (M.P. and M.H.), F31 MH105217 (M.P.), F32 MH096464 (A.K.F.), P01 DA008227 (R.C.M.) and F30 MH100835 (M.H.).
Author information
Authors and Affiliations
Contributions
D.J.C., S.A.G. and S.J.R. designed the studies, interpreted the results and wrote the paper. D.J.C., S.A.G., J.J.W., M.H., A.K.F., H.A., L.A.K., N.R., M.P., G.A.B.-D. and G.E.H. performed stereotactic injections, optogenetic experiments and electrophysiology. K.G.G., S.A.G. and M.L.S. performed in vivo ChR2 validation. D.J.C., A.D., J.L.A. and M.S. performed immunohistochemistry. J.L.A. and I.I.-T. generated viruses. K.D., J.J.W., A.K.F., R.C.M. and M.-H.H. provided optogenetics and electrophysiology training. All of the authors edited the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 vSTR VGLUT2 levels correlate with susceptibility to social defeat stress
(a) Confocal images of VGLUT2 puncta in the vSTR of control, resilient and susceptible mice; scale bar = 10 μm. (b) Quantification of VGLUT2 puncta density (control/resilient/susceptible, n = 4/4/5 mice, one-way ANOVA, * p = 0.020, F (2,10) = 5.826). (c) Linear regression analysis revealing a significant correlation of VGLUT2 puncta density with social interaction ratio (r2 = 0.519, p = 0.005; curved lines represent the 95% confidence interval). (d) Confocal images of VGLUT1 puncta in the vSTR of control, resilient and susceptible mice; scale bar = 10 μm. (e) Quantification of VGLUT1 puncta density (control/resilient/susceptible, n = 4/3/5 mice, one-way ANOVA, p = 0.533, F (2,9) = 0.7205). (f) Linear regression analysis revealing no correlation of VGLUT1 puncta density to social interaction ratio (r2 = 0.005, p = 0.303; curved lines represent the 95% confidence interval). Confocal image of VGLUT1 puncta (red) and EYFP containing terminals (green) in the vSTR from the (g) ILT and (i) PFC; scale bar = 10 μm. Colocalization mask is shown to the right of each image. Confocal image of VGLUT2 puncta (red) and EYFP containing terminals (green) in the vSTR from the (h) ILT and (j) PFC; scale bar = 10 μm.
Supplementary Figure 2 CSDS does not alter the AMPAR/NMDAR ratio at all inputs to vSTR
Quantification of electrically evoked EPSCs at -70 mV and +40 mV show that chronic social defeat significantly does not regulate the AMPAR/NMDAR ratio at all inputs to MSNs (control/resilient/susceptible, n = 10/6/8cells, one-way ANOVA, p = 0.371, F(2,21) = 0.36)
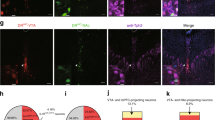
Supplementary Figure 3 Immunohistochemical validation of tToxins.
(a) Representative images of toxin infection in area of recordings (left-right: merge, green - ω-conotoxin, red - ω-agatoxin, scale bar = 5 μm). (b) Confocal images of coronal ILT sections stained for Cas-3. Left, EYFP control virus (green) in ILT with no Cas-3; Middle, tToxin (red) in ILT with no Cas-3; Right, Dentate Gyrus shown from the same coronal section with tToxin expression as a positive control for Cas-3 staining, positive cell shown in green. DAPI (blue) stain present in all images scale = 100 μm.
Supplementary Figure 4 Effects of ILT-vSTR silencing with tToxins on CSDS induced adaptations.
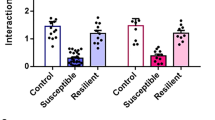
(a) Linear regression analysis revealing a significant correlation of stubby spine density with social interaction ratio (r2 = 0.49, p < 0.0243; curved lines represent the 95% confidence interval). (b) Time spent in corner zones in mice expressing either DIO-EYFP or -tToxins in ILT-vSTR circuit (EYFP/tToxin, n = 14/15 mice, 2-way ANOVA, * p = 0.045, F (1, 54) = 4.195 (interaction)). (c) Total distance traveled during social interaction in mice expressing either DIO-EYFP or -tToxins in ILT-vSTR circuit (EYFP/tToxin, n = 14/15 mice, 2-way ANOVA, p = 0.315, F (1, 54) = 1.027 (interaction)).
Supplementary Figure 5 Effects of PFC-vSTR silencing with tToxins on CSDS-induced adaptations.
(a) Linear regression analysis revealing no correlation of stubby spine density with social interaction ratio (r2 = 0.01, p = 0.862; curved lines represent the 95% confidence interval; right). (b) Time spent in corner zones in mice expressing either EYFP or tToxin in ILT-vSTR circuit (EYFP/tToxin, n = 7/6 mice, 2-way ANOVA, t p = 0.07, F (1, 22) = 4.392 (interaction)). (c) Total distance traveled during social interaction in mice expressing either EYFP or tToxin in ILT-vSTR circuit (EYFP/tToxin, n = 7/6 mice, 2-way ANOVA, p = 0.705, F (1, 22) = 0.1468 (interaction)).
Supplementary Figure 6 Quantification of spine densities
3-D reconstructions of MSN dendrites representative of animals expressing EYFP or tToxin after social defeat stress in (a) ILT-vSTR or (b) PFC-vSTR neurons (white arrows-stubby spines, yellow arrows-thin spines, orange arrows–mushroom spines). (c-h) Bar graph representations of mean number of other spine types (EYFP/tToxin, n= 5 mice per group, unpaired t-test, c – p =.112, t = 1.785 df = 8, d – p = 0.796, t=0.2658 df = 8, e – p =.157, t = 1.542 df = 8; EYFP/tToxin, n = 4 mice per group, f – p =.551, t = 0.6498 df = 6, g – p = 0.493, t = 0.7528 df = 6, h – p = 0.666, t = 0.4658 df = 6); scale bar = 5μm.
Supplementary Figure 7 Effects of ILT-vSTR and PFC-vSTR silencing with NpHR on social defeat–induced behaviors.
(a) Time spent in corner zones in mice expressing either EYFP or NpHR in ILT-vSTR circuit (EYFP/NpHr, n = 16/17 mice, paired t-test, t = 0.07, t = 1.908 df = 16). (b) Total distance traveled during social interaction in mice expressing either EYFP or NpHR in ILT-vSTR circuit (EYFP/NpHr, n = 16/17 mice, 2-way ANOVA, p = 0.187, F (1, 62) = 1.777(interaction)). (c) Time spent in corner zones in mice expressing either EYFP or NpHR in PFC-vSTR circuit (n = 11 mice/group, unpaired t-test, *p = 0.048, t = 2.102 df = 20). (d) Total distance traveled during social interaction in mice expressing either EYFP or NpHR in PFC-vSTR circuit (n = 11 mice/group, 2-way ANOVA, p = 0.40, F (1,40) = 0.7162 (interaction)).
Supplementary Figure 8 Effects of ILT- and PFC-vSTR activation with ChR2 on CSDS-induced behaviors.
(a) Schematic, immunohistochemistry and in vitro validation of stimulation parameters of ILT neurons; scale bar = 200 μm. (b) Schematic, immunohistochemistry and, in vitro validation of stimulation parameters of PFC neurons circuit; scale bar = 200 μm (1 cell per trace). Time spent in corner zones in mice expressing either DIO-EYFP or ChR2 in (c) ILT-vSTR stimulation (EYFP/ChR2, n = 14/18 mice, 2-way ANOVA, *p = 0.045, F (1, 60) = 6.893 (interaction)) and (c) PFC-vSTR stimulation (EYFP/Chr2, n = 6 mice per group, 2-way ANOVA, p = 0.518, F (1, 20) = 0.4324 (interaction)) with blue light. Total distance traveled during social interaction in mice expressing either DIO-EYFP or ChR2 in (c) ILT-vSTR (EYFP/ChR2, n = 14/18 mice, 2-way ANOVA, p = 0.325, F (1, 60) = 0.9815 (interaction)) and (d) PFC-vSTR with blue light stimulation (n = 6 mice per group, 2-way ANOVA, p = 0.472, F (1, 20) = 0.5385 (interaction)).
Supplementary Figure 9 Effects of ILT-vSTR ChR2 terminal stimulation on social defeat–induced behaviors.
(a) Quantification of time spent in the interaction zone and social interaction ratio after a subthreshold defeat and stimulation of ChR2-expressing ILT terminals during the SI test (EYFP/Chr2, n = 16/14 mice, unpaired t-test (Time spent) * p = 0.004 (EYFP; target absent vs target present), t = 3.087 df = 27, (SI ratio) tp = 0.11, t = 1.672 df = 27). (b) Time spent in corner zones in mice expressing either DIO-EYFP or ChR2 and stimulation of ILT terminals during the SI test (unpaired t-test, * p = 0.032, t = 2.372 df = 14 (interaction)). (c) Total distance traveled during social interaction in mice expressing either DIO-EYFP or ChR2 and stimulation of ILT terminals during the SI test (2-way ANOVA, p = 0.361, F (1, 34) = 0.8549 (interaction)).
Supplementary Figure 10 Effects of high frequency PFC-vSTR ChR2 terminal stimulation on social defeat–induced behaviors.
(a) Quantification of time spent in the interaction zone and social interaction ratio in previously susceptible animals with 100 Hz stimulation of ChR2-expressing PFC terminals during the SI test (EYFP/ChR2, n = 9/10 mice, two-ANOVA (Time spent) * p = 0.046, F (1, 34) = 4.294 (interaction), unpaired t-test (SI ratio) * p = 0.040, t = 2.214 df = 17). (b) Time spent in corner zones in mice expressing either EYFP or ChR2 and stimulation of PFC terminals during the SI test (EYFP/ChR2, n= 9/10 mice, 2-way ANOVA, p = 0.64, F (1, 34) = 0.2221 (interaction)). (c) Total distance traveled during social interaction in mice expressing either EYFP or ChR2 and stimulation of PFC terminals during the SI test (EYFP/ChR2, n = 9/10 mice, 2-way ANOVA, p = 0.813, F (1, 34) = 0.05669 (interaction)).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–10 (PDF 1920 kb)
Rights and permissions
About this article
Cite this article
Christoffel, D., Golden, S., Walsh, J. et al. Excitatory transmission at thalamo-striatal synapses mediates susceptibility to social stress. Nat Neurosci 18, 962–964 (2015). https://doi.org/10.1038/nn.4034
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.4034
This article is cited by
-
Neural circuits regulating prosocial behaviors
Neuropsychopharmacology (2023)
-
A Systematic Review of Direct Outputs from the Cerebellum to the Brainstem and Diencephalon in Mammals
The Cerebellum (2022)
-
Behavioral and accumbens synaptic plasticity induced by cues associated with restraint stress
Neuropsychopharmacology (2021)
-
Kappa opioid receptor modulation of excitatory drive onto nucleus accumbens fast-spiking interneurons
Neuropsychopharmacology (2021)
-
Input-specific modulation of murine nucleus accumbens differentially regulates hedonic feeding
Nature Communications (2021)