Abstract
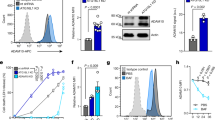
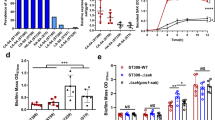
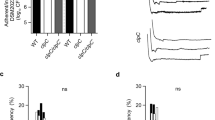
Staphylococcus aureus is a major cause of human disease, responsible for half a million infections and approximately 20,000 deaths per year in the United States alone1,2. This pathogen secretes α-hemolysin, a pore-forming cytotoxin that contributes to the pathogenesis of pneumonia3,4,5. α-hemolysin injures epithelial cells in vitro by interacting with its receptor, the zinc-dependent metalloprotease ADAM10 (ref. 6). We show here that mice harboring a conditional disruption of the Adam10 gene in lung epithelium are resistant to lethal pneumonia. Investigation of the molecular mechanism of toxin-receptor function revealed that α-hemolysin upregulates ADAM10 metalloprotease activity in alveolar epithelial cells, resulting in cleavage of the adherens junction protein E-cadherin. Cleavage is associated with disruption of epithelial barrier function, contributing to the pathogenesis of lethal acute lung injury. A metalloprotease inhibitor of ADAM10 prevents E-cadherin cleavage in response to Hla; similarly, toxin-dependent E-cadherin proteolysis and barrier disruption is attenuated in ADAM10-knockout mice. Together, these data attest to the function of ADAM10 as the cellular receptor for α-hemolysin. The observation that α-hemolysin can usurp the metalloprotease activity of its receptor reveals a previously unknown mechanism of pore-forming cytotoxin action in which pathologic insults are not solely the result of irreversible membrane injury and defines ADAM10 inhibition as a strategy to attenuate α-hemolysin-induced disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Klevens, R.M. et al. Invasive methicillin-resistant Staphylococcus aureus infections in the United States. J. Am. Med. Assoc. 298, 1763–1771 (2007).
Otto, M. Basis of virulence in community-associated methicillin-resistant Staphylococcus aureus. Annu. Rev. Microbiol. 64, 143–162 (2010).
Bubeck Wardenburg, J., Patel, R.J. & Schneewind, O. Surface proteins and exotoxins are required for the pathogenesis of Staphylococcus aureus pneumonia. Infect. Immun. 75, 1040–1044 (2007).
Bubeck Wardenburg, J., Bae, T., Otto, M., Deleo, F.R. & Schneewind, O. Poring over pores: α-hemolysin and Panton-Valentine leukocidin in Staphylococcus aureus pneumonia. Nat. Med. 13, 1405–1406 (2007).
Bubeck Wardenburg, J. & Schneewind, O. Vaccine protection against Staphylococcus aureus pneumonia. J. Exp. Med. 205, 287–294 (2008).
Wilke, G.A. & Bubeck Wardenburg, J. Role of a disintegrin and metalloprotease 10 in Staphylococcus aureus α-hemolysin–mediated cellular injury. Proc. Natl. Acad. Sci. USA 107, 13473–13478 (2010).
Gonzalez, M.R., Bischofberger, M., Pernot, L., van der Goot, F.G. & Freche, B. Bacterial pore-forming toxins: the (w)hole story? Cell. Mol. Life Sci. 65, 493–507 (2008).
Tomita, T. & Kamio, Y. Molecular biology of the pore-forming cytolysins from Staphylococcus aureus, α- and γa-hemolysins and leukocidin. Biosci. Biotechnol. Biochem. 61, 565–572 (1997).
O'Callaghan, R.J. et al. Specific roles of α-toxin and β-toxin during Staphylococcus aureus corneal infection. Infect. Immun. 65, 1571–1578 (1997).
Kennedy, A.D. et al. Targeting of α-hemolysin by active or passive immunization decreases severity of USA300 skin infection in a mouse model. J. Infect. Dis. 202, 1050–1058 (2010).
Brosnahan, A.J., Mantz, M.J., Squier, C.A., Peterson, M.L. & Schlievert, P.M. Cytolysins augment superantigen penetration of stratified mucosa. J. Immunol. 182, 2364–2373 (2009).
Song, L. et al. Structure of staphylococcal α-hemolysin, a heptameric transmembrane pore. Science 274, 1859–1866 (1996).
Hartmann, D. et al. The disintegrin/metalloprotease ADAM 10 is essential for Notch signalling but not for α-secretase activity in fibroblasts. Hum. Mol. Genet. 11, 2615–2624 (2002).
Perl, A.K., Wert, S.E., Nagy, A., Lobe, C.G. & Whitsett, J.A. Early restriction of peripheral and proximal cell lineages during formation of the lung. Proc. Natl. Acad. Sci. USA 99, 10482–10487 (2002).
Tian, L. et al. ADAM10 is essential for proteolytic activation of Notch during thymocyte development. Int. Immunol. 20, 1181–1187 (2008).
Ragle, B.E. & Bubeck Wardenburg, J. Anti–α-hemolysin monoclonal antibodies mediate protection against Staphylococcus aureus pneumonia. Infect. Immun. 77, 2712–2718 (2009).
Reiss, K. & Saftig, P. The “a disintegrin and metalloprotease” (ADAM) family of sheddases: physiological and cellular functions. Semin. Cell Dev. Biol. 20, 126–137 (2009).
Seals, D.F. & Courtneidge, S.A. The ADAMs family of metalloproteases: multidomain proteins with multiple functions. Genes Dev. 17, 7–30 (2003).
Menzies, B.E. & Kernodle, D.S. Site-directed mutagenesis of the α-toxin gene of Staphylococcus aureus: role of histidines in toxin activity in vitro and in a murine model. Infect. Immun. 62, 1843–1847 (1994).
Kim, M. et al. Bacterial interactions with the host epithelium. Cell Host Microbe 8, 20–35 (2010).
Maretzky, T. et al. ADAM10 mediates E-cadherin shedding and regulates epithelial cell-cell adhesion, migration, and beta-catenin translocation. Proc. Natl. Acad. Sci. USA 102, 9182–9187 (2005).
Murphy, G. Regulation of the proteolytic disintegrin metalloproteinases, the 'Sheddases'. Semin. Cell Dev. Biol. 20, 138–145 (2009).
Rubins, J.B. et al. Dual function of pneumolysin in the early pathogenesis of murine pneumococcal pneumonia. J. Clin. Invest. 95, 142–150 (1995).
Marriott, H.M. & Dockrell, D.H. Streptococcus pneumoniae: the role of apoptosis in host defense and pathogenesis. Int. J. Biochem. Cell Biol. 38, 1848–1854 (2006).
Steinhusen, U. et al. Cleavage and shedding of E-cadherin after induction of apoptosis. J. Biol. Chem. 276, 4972–4980 (2001).
Matthay, M.A. & Zemans, R.L. The Acute respiratory distress syndrome: pathogenesis and treatment. Annu. Rev. Pathol. 6, 147–163 (2011).
Gómez, M.I., Seaghdha, M.O. & Prince, A.S. Staphylococcus aureus protein A activates TACE through EGFR-dependent signaling. EMBO J. 26, 701–709 (2007).
Martin, F.J., Parker, D., Harfenist, B.S., Soong, G. & Prince, A. Participation of CD11c+ leukocytes in methicillin-resistant Staphylococcus aureus clearance from the lung. Infect. Immun. 79, 1898–1904 (2011).
Ludwig, A. et al. Metalloproteinase inhibitors for the disintegrin-like metalloproteinases ADAM10 and ADAM17 that differentially block constitutive and phorbol ester-inducible shedding of cell surface molecules. Comb. Chem. High Throughput Screen. 8, 161–171 (2005).
Dijkstra, A. et al. Expression of ADAMs (“a disintegrin and metalloprotease”) in the human lung. Virchows Arch. 454, 441–449 (2009).
Wilke, G.A. & Bubeck Wardenburg, J. Role of a disintegrin and metalloprotease 10 in Staphylococcus aureus α-hemolysin–mediated cellular injury. Proc. Natl. Acad. Sci. USA 107, 13473–13478 (2010).
Bubeck Wardenburg, J. & Schneewind, O. Vaccine protection against Staphylococcus aureus pneumonia. J. Exp. Med. 205, 287–294 (2008).
Illek, B. et al. Cl transport in complemented CF bronchial epithelial cells correlates with CFTR mRNA expression levels. Cell. Physiol. Biochem. 22, 57–68 (2008).
Hoettecke, N., Ludwig, A., Foro, S. & Schmidt, B. Improved synthesis of ADAM10 inhibitor GI254023X. Neurodegener. Dis. 7, 232–238 (2010).
Bubeck Wardenburg, J., Patel, R.J. & Schneewind, O. Surface proteins and exotoxins are required for the pathogenesis of Staphylococcus aureus pneumonia. Infect. Immun. 75, 1040–1044 (2007).
Bubeck Wardenburg, J., Bae, T., Otto, M., Deleo, F.R. & Schneewind, O. Poring over pores: α-hemolysin and Panton-Valentine leukocidin in Staphylococcus aureus pneumonia. Nat. Med. 13, 1405–1406 (2007).
Acknowledgements
We thank T. Golovkina for assistance with mouse breeding strategies, A. Crofts for pilot studies of E-cadherin cleavage in vitro, D. Gruenert (University of California San Francisco) for 16HBE14o- cells, R. Tweten (The University of Oklahoma Health Sciences Center) for the provision of recombinant PLY, J. Whitsett (Cincinnati Children's Medical Center) for SP-C-rtTA-(tetO)7CMV-Cre mice, O. Schneewind for discussions and comments on the manuscript, C. Labno for microscopy support, T. Li for immunohistochemistry support, and the Integrated Microscopy and Immunohistochemistry Facilities at the University of Chicago. This work was supported by the Departments of Pediatrics and Microbiology at the University of Chicago. M.E.P. was partially supported by US National Institutes of Health grant T32 GM007183. The authors acknowledge membership in and support from the Region V 'Great Lakes' Regional Center for Excellence (US National Institutes of Health award 2-U54-AI-057153).
Author information
Authors and Affiliations
Contributions
I.I. performed mouse infection modeling, in vivo E-cadherin cleavage studies and ECIS studies. N.I. performed mouse breeding and genetic analysis and assisted with infection modeling. G.A.W. performed siRNA transfections, analyzed ADAM10 expression on 16HBE14o- cells and performed cellular assays of metalloprotease activity. M.E.P. examined the effects of GI254023X on toxin binding and performed ECIS experiments. K.M.F. generated the HlaPPL mutant. Y.W. performed ECIS experiments. J.B.W. performed cellular assays of metalloprotease activity, E-cadherin cleavage and immunofluorescence microscopy and wrote the paper. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 and Supplementary Methods (PDF 2009 kb)
Rights and permissions
About this article
Cite this article
Inoshima, I., Inoshima, N., Wilke, G. et al. A Staphylococcus aureus pore-forming toxin subverts the activity of ADAM10 to cause lethal infection in mice. Nat Med 17, 1310–1314 (2011). https://doi.org/10.1038/nm.2451
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2451
This article is cited by
-
Cleavage of cell junction proteins as a host invasion strategy in leptospirosis
Applied Microbiology and Biotechnology (2024)
-
Toxin expression during Staphylococcus aureus infection imprints host immunity to inhibit vaccine efficacy
npj Vaccines (2023)
-
Impact of enteric bacterial infections at and beyond the epithelial barrier
Nature Reviews Microbiology (2023)
-
ADAM10-a “multitasker” in sepsis: focus on its posttranslational target
Inflammation Research (2023)
-
The pro-inflammatory effect of Staphylokinase contributes to community-associated Staphylococcus aureus pneumonia
Communications Biology (2022)