Abstract
Resident macrophages densely populate the normal arterial wall, yet their origins and the mechanisms that sustain them are poorly understood. Here we use gene-expression profiling to show that arterial macrophages constitute a distinct population among macrophages. Using multiple fate-mapping approaches, we show that arterial macrophages arise embryonically from CX3CR1+ precursors and postnatally from bone marrow–derived monocytes that colonize the tissue immediately after birth. In adulthood, proliferation (rather than monocyte recruitment) sustains arterial macrophages in the steady state and after severe depletion following sepsis. After infection, arterial macrophages return rapidly to functional homeostasis. Finally, survival of resident arterial macrophages depends on a CX3CR1-CX3CL1 axis within the vascular niche.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
Primary accessions
Gene Expression Omnibus
Referenced accessions
Gene Expression Omnibus
References
Akazaki, K. A concept of reticuloendothelial system. Tohoku J. Exp. Med. 76, 107–118 (1962).
Naito, M., Yamamura, F., Nishikawa, S. & Takahashi, K. Development, differentiation, and maturation of fetal mouse yolk sac macrophages in cultures. J. Leukoc. Biol. 46, 1–10 (1989).
van Furth, R. & Cohn, Z.A. The origin and kinetics of mononuclear phagocytes. J. Exp. Med. 128, 415–435 (1968).
Samokhvalov, I.M., Samokhvalova, N.I. & Nishikawa, S. Cell tracing shows the contribution of the yolk sac to adult haematopoiesis. Nature 446, 1056–1061 (2007).
Ginhoux, F. et al. Fate mapping analysis reveals that adult microglia derive from primitive macrophages. Science 330, 841–845 (2010).
Schulz, C. et al. A lineage of myeloid cells independent of Myb and hematopoietic stem cells. Science 336, 86–90 (2012).
Hoeffel, G. et al. Adult Langerhans cells derive predominantly from embryonic fetal liver monocytes with a minor contribution of yolk sac-derived macrophages. J. Exp. Med. 209, 1167–1181 (2012).
Epelman, S. et al. Embryonic and adult-derived resident cardiac macrophages are maintained through distinct mechanisms at steady state and during inflammation. Immunity 40, 91–104 (2014).
Molawi, K. et al. Progressive replacement of embryo-derived cardiac macrophages with age. J. Exp. Med. 211, 2151–2158 (2014).
Hoeffel, G. et al. C-myb+ erythro-myeloid progenitor-derived fetal monocytes give rise to adult tissue-resident macrophages. Immunity 42, 665–678 (2015).
Gomez Perdiguero, E. et al. Tissue-resident macrophages originate from yolk-sac-derived erythro-myeloid progenitors. Nature 518, 547–551 (2015).
Hashimoto, D. et al. Tissue-resident macrophages self-maintain locally throughout adult life with minimal contribution from circulating monocytes. Immunity 38, 792–804 (2013).
Yona, S. et al. Fate mapping reveals origins and dynamics of monocytes and tissue macrophages under homeostasis. Immunity 38, 79–91 (2013).
Gautier, E.L. et al. Gene-expression profiles and transcriptional regulatory pathways that underlie the identity and diversity of mouse tissue macrophages. Nat. Immunol. 13, 1118–1128 (2012).
Majesky, M.W., Dong, X.R., Hoglund, V., Mahoney, W.M.J. & Daum, G. The adventitia: a dynamic interface containing resident progenitor cells. Arterioscler. Thromb. Vasc. Biol. 31, 1530–1539 (2011).
Jakubzick, C. et al. Minimal differentiation of classical monocytes as they survey steady-state tissues and transport antigen to lymph nodes. Immunity 39, 599–610 (2013).
Meredith, M.M. et al. Expression of the zinc finger transcription factor zDC (Zbtb46, Btbd4) defines the classical dendritic cell lineage. J. Exp. Med. 209, 1153–1165 (2012).
Satpathy, A.T. et al. Zbtb46 expression distinguishes classical dendritic cells and their committed progenitors from other immune lineages. J. Exp. Med. 209, 1135–1152 (2012).
McGovern, N. et al. Human dermal CD14+ cells are a transient population of monocyte-derived macrophages. Immunity 41, 465–477 (2014).
Galkina, E. et al. Lymphocyte recruitment into the aortic wall before and during development of atherosclerosis is partially L-selectin dependent. J. Exp. Med. 203, 1273–1282 (2006).
Choi, J.H. et al. Identification of antigen-presenting dendritic cells in mouse aorta and cardiac valves. J. Exp. Med. 206, 497–505 (2009).
Choi, J.H. et al. Flt3 signaling-dependent dendritic cells protect against atherosclerosis. Immunity 35, 819–831 (2011).
Guilliams, M. et al. Alveolar macrophages develop from fetal monocytes that differentiate into long-lived cells in the first week of life via GM-CSF. J. Exp. Med. 210, 1977–1992 (2013).
Medvinsky, A. & Dzierzak, E. Definitive hematopoiesis is autonomously initiated by the AGM region. Cell 86, 897–906 (1996).
Parkhurst, C.N. et al. Microglia promote learning-dependent synapse formation through brain-derived neurotrophic factor. Cell 155, 1596–1609 (2013).
Prinz, M. & Priller, J. Microglia and brain macrophages in the molecular age: from origin to neuropsychiatric disease. Nat. Rev. Neurosci. 15, 300–312 (2014).
Karsunky, H., Merad, M., Cozzio, A., Weissman, I.L. & Manz, M.G. Flt3 ligand regulates dendritic cell development from Flt3+ lymphoid and myeloid–committed progenitors to Flt3+ dendritic cells in vivo. J. Exp. Med. 198, 305–313 (2003).
Bain, C.C. et al. Constant replenishment from circulating monocytes maintains the macrophage pool in the intestine of adult mice. Nat. Immunol. 15, 929–937 (2014).
Davies, L.C., Jenkins, S.J., Allen, J.E. & Taylor, P.R. Tissue-resident macrophages. Nat. Immunol. 14, 986–995 (2013).
Ginhoux, F. & Jung, S. Monocytes and macrophages: developmental pathways and tissue homeostasis. Nat. Rev. Immunol. 14, 392–404 (2014).
Kim, K.W. et al. In vivo structure/function and expression analysis of the CX3C chemokine fractalkine. Blood 118, e156–e167 (2011).
Serbina, N.V. & Pamer, E.G. Monocyte emigration from bone marrow during bacterial infection requires signals mediated by chemokine receptor CCR2. Nat. Immunol. 7, 311–317 (2006).
Robbins, C.S. et al. Local proliferation dominates lesional macrophage accumulation in atherosclerosis. Nat. Med. 19, 1166–1172 (2013).
Goldmann, T. et al. A new type of microglia gene targeting shows TAK1 to be pivotal in CNS autoimmune inflammation. Nat. Neurosci. 16, 1618–1626 (2013).
Foudi, A. et al. Analysis of histone 2B-GFP retention reveals slowly cycling hematopoietic stem cells. Nat. Biotechnol. 27, 84–90 (2009).
Mildner, A. et al. Microglia in the adult brain arise from Ly-6ChiCCR2+ monocytes only under defined host conditions. Nat. Neurosci. 10, 1544–1553 (2007).
Ajami, B., Bennett, J.L., Krieger, C., Tetzlaff, W. & Rossi, F.M. Local self-renewal can sustain CNS microglia maintenance and function throughout adult life. Nat. Neurosci. 10, 1538–1543 (2007).
Sieweke, M.H. & Allen, J.E. Beyond stem cells: self-renewal of differentiated macrophages. Science 342, 1242974 (2013).
Barth, M.W., Hendrzak, J.A., Melnicoff, M.J. & Morahan, P.S. Review of the macrophage disappearance reaction. J. Leukoc. Biol. 57, 361–367 (1995).
Kierdorf, K. et al. Microglia emerge from erythromyeloid precursors via Pu.1- and Irf8-dependent pathways. Nat. Neurosci. 16, 273–280 (2013).
Chorro, L. et al. Langerhans cell (LC) proliferation mediates neonatal development, homeostasis, and inflammation-associated expansion of the epidermal LC network. J. Exp. Med. 206, 3089–3100 (2009).
Boehme, S.A., Lio, F.M., Maciejewski-Lenoir, D., Bacon, K.B. & Conlon, P.J. The chemokine fractalkine inhibits Fas-mediated cell death of brain microglia. J. Immunol. 165, 397–403 (2000).
Lionakis, M.S. et al. CX3CR1-dependent renal macrophage survival promotes Candida control and host survival. J. Clin. Invest. 123, 5035–5051 (2013).
Zheng, J. et al. Chemokine receptor CX3CR1 contributes to macrophage survival in tumor metastasis. Mol. Cancer 12, 141 (2013).
Landsman, L. et al. CX3CR1 is required for monocyte homeostasis and atherogenesis by promoting cell survival. Blood 113, 963–972 (2009).
Gautier, E.L. et al. Gene-expression profiles and transcriptional regulatory pathways that underlie the identity and diversity of mouse tissue macrophages. Nat. Immunol. 13, 1118–1128 (2012).
Jongstra-Bilen, J. et al. Low-grade chronic inflammation in regions of the normal mouse arterial intima predisposed to atherosclerosis. J. Exp. Med. 203, 2073–2083 (2006).
Paulson, K.E. et al. Resident intimal dendritic cells accumulate lipid and contribute to the initiation of atherosclerosis. Circ. Res. 106, 383–390 (2010).
Carvalho, B.S. & Irizarry, R.A. A framework for oligonucleotide microarray preprocessing. Bioinformatics 26, 2363–2367 (2010).
Acknowledgements
We acknowledge the administrative assistance of B. Bali. Supported by a Canadian Institutes of Health Research (CIHR) New Investigator Award (MSH136670), a CIHR operating grant (MOP133390), an Ontario Lung Association/Pfizer Award and the Peter Munk Chair in Aortic Disease Research (C.S.R.) and an Ontario Graduate Scholarship (S.E.).
Author information
Authors and Affiliations
Contributions
S.E., A.L. and R.B. conceived the project, designed and performed experiments and analyzed and interpreted data. N.D., J.C., M.R., M.E.M., J.W., E.S., C.L., B.L., L.R., T.J.Y., J.S.L., P.W., I.H., S.E., R.K. and K.F. performed experiments and helped interpret the data. M.O., J.B., C.C.J.Z., G.A.L., C.M.T.B., P.L., M.H., F.K.S., C.C., M.P., I.H., G.J.R., S.E., A.O.G., M.C. and B.B.R. provided materials and intellectual input and edited the manuscript. C.S.R. conceived the project, designed and performed experiments, supervised the study and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
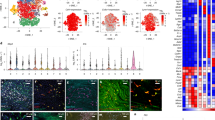
Supplementary Figure 1 Phenotype and gene-expression profiling of arterial macrophages.
(a) Representative dot plots identifying macrophages in the liver, lung, and brain by flow cytometry. (b) Heat map of mRNA transcripts increased in each single macrophage population by fivefold or more relative to their expression in the remaining 3 populations. (c) Heat map of mRNA transcripts decreased in each single macrophage population by fivefold or more relative to their expression in the remaining 3 populations. (d) Flow cytometry analysis of blood monocytes and arterial macrophages following treatment with clodronate liposomes (n = 3). (e) Number of CD45+ leukocytes isolated from adventitia and intima/media of aorta (mean ± SEM; adventitia n = 27, intima/media n = 22).* P = 0.0018 (unpaired Student’s t-test). (f) Visualization of arterial dendritic cells (DC) in CD11ceYFP/+ mice. (g) Representative dot plots showing presence of macrophages in carotid and femoral arteries.
Supplementary Figure 2 Arterial macrophages have embryonic and postnatal origins.
E8.5-induced CX3CR1-TdTomato expression in F4/80highCD11blow liver macrophages and F4/80intCD11bhigh liver monocytes at E16.5 (mean ± SD; n = 6). Data show a representative histogram from one of 6 mice analyzed. (b) E-selectin, ICAM-1, and CCL5 expression detected by RT-PCR and conducted on aortic tissue taken from mice at indicated time points (mean ± SD; n = 4 mice per time point except t = 1 where n = 3).* P < 0.05 (unpaired t-test).
Supplementary Figure 3 CX3CL1-CX3CR1 interactions determine survival of arterial macrophages.
(a) Enumeration of aortic macrophages in Csf2r−/− mice (mean ± SEM, n = 5). P = 0.1776 (unpaired Student’s t-test). (b) CD68 staining of aortic adventitia in wild type and Csf1−/− mice (mean ± SEM; wild type n = 9, M-CSF−/− n = 10).* P = 0.0002 (unpaired Student’s t-test). (c) Representative dot plots represent negative controls (C57BL6/J mice) for identifying GFP+ staining in CX3CR1gfp/+ mice. (d) IF showing association of adventitial aortic macrophages (CD68+) with CX3CL1+ cells in CX3CL1cherry mice. Shown is a representative image from one of several mice examined. (e) Representative dot plots showing absence of CX3CL1 expression in CD45+ cells isolated from aortae of CX3CL1cherry mice. 3 animals were analyzed (mean ± SD). (f) Representative IF section showing isotype staining for mcherry (rabbit IgG) and PDGFRα and CD31 (ratIgG2a).
Supplementary Figure 4 Arterial macrophages are maintained independently of monocytes in adulthood.
(a) Representative histogram showing Tdtomato labeling of blood monocytes in CX3CR1creER R26Tomato mice 9 and 11 months after tamoxifen exposure. (b) Adult TetOP-H2B-GFP mice were administered doxycycline for 4 weeks to induce H2B-GFP expression and then loss of fluorescence in aortic macrophages (indicative of cell division) was monitored during a 2 month chase period. (c) Representative histogram showing 5-bromodeoxyuridine (BrdU) incorporation by aortic MΦ.
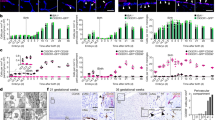
Supplementary Figure 5 Arterial macrophages self-renew after exposure to bacteria.
(a) Enumeration of aortic macrophages in wild type mice following induction of sepsis by cecal puncture (mean ± SEM; day 0 n = 4, day 1 n = 4, day 7 n = 5). (b) Enumeration of neutrophils following exposure to LPS. Data were collected at multiple time points (mean ± SD; day 0 n = 8, 4h n = 3, 12h n = 3, 24h n = 3, 72h n = 4, 120h n = 4, 336h n = 3, 648h n = 3). (c) Flow cytometry characterization of CD11b (left panel) and Ly6C (right panel) expression on CD115+Lyve-1+ and CD115−Lyve-1− macrophages. (d) Representative dot plots showing purification of aortic macrophages following cell sorting. (e) Phagocytosis of pHrodo™ E. coli BioParticles® 7 days after induction of sepsis by cecal puncture (mean ± SD, n = 4). P = 0.26 (unpaired t-test).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 and Supplementary Tables 1–6 (PDF 921 kb)
Supplementary Table 7
Data sets and accession codes. (XLSX 12 kb)
Rights and permissions
About this article
Cite this article
Ensan, S., Li, A., Besla, R. et al. Self-renewing resident arterial macrophages arise from embryonic CX3CR1+ precursors and circulating monocytes immediately after birth. Nat Immunol 17, 159–168 (2016). https://doi.org/10.1038/ni.3343
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.3343
This article is cited by
-
Border-associated macrophages in the central nervous system
Journal of Neuroinflammation (2024)
-
Macrophage-driven cardiac inflammation and healing: insights from homeostasis and myocardial infarction
Cellular & Molecular Biology Letters (2023)
-
Single-cell RNA-Seq analysis of diabetic wound macrophages in STZ-induced mice
Journal of Cell Communication and Signaling (2023)
-
The Characteristics of Macrophage Heterogeneity in Atherosclerotic Aortas
Journal of Cardiovascular Translational Research (2023)
-
Extracellular vesicle miR-32 derived from macrophage promotes arterial calcification in mice with type 2 diabetes via inhibiting VSMC autophagy
Journal of Translational Medicine (2022)