Abstract
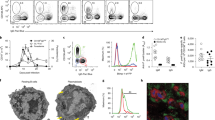
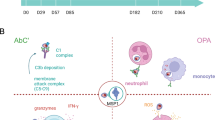
Infection of erythrocytes with Plasmodium species induces clinical malaria. Parasite-specific CD4+ T cells correlate with lower parasite burdens and severity of human malaria and are needed to control blood-stage infection in mice. However, the characteristics of CD4+ T cells that determine protection or parasite persistence remain unknown. Here we show that infection of humans with Plasmodium falciparum resulted in higher expression of the inhibitory receptor PD-1 associated with T cell dysfunction. In vivo blockade of the PD-1 ligand PD-L1 and the inhibitory receptor LAG-3 restored CD4+ T cell function, amplified the number of follicular helper T cells and germinal-center B cells and plasmablasts, enhanced protective antibodies and rapidly cleared blood-stage malaria in mice. Thus, chronic malaria drives specific T cell dysfunction, and proper function can be restored by inhibitory therapies to enhance parasite control.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Mackinnon, M.J. & Marsh, K. The selection landscape of malaria parasites. Science 328, 866–871 (2010).
World Health Organization. World Malaria Report 2010. <http://www.who.int/malaria/world_malaria_report_2010/en/index.html>.
Snow, R.W. & Marsh, K. The consequences of reducing transmission of Plasmodium falciparum in Africa. Adv. Parasitol. 52, 235–264 (2002).
Wykes, M. & Good, M.F. A case for whole-parasite malaria vaccines. Int. J. Parasitol. 37, 705–712 (2007).
Good, M.F. & Doolan, D.L. Malaria vaccine design: immunological considerations. Immunity 33, 555–566 (2010).
Pinzon-Charry, A. et al. Low doses of killed parasite in CpG elicit vigorous CD4+ T cell responses against blood-stage malaria in mice. J. Clin. Invest. 120, 2967–2978 (2010).
Elliott, S.R., Kuns, R.D. & Good, M.F. Heterologous immunity in the absence of variant-specific antibodies after exposure to subpatent infection with blood-stage malaria. Infect. Immun. 73, 2478–2485 (2005).
Pombo, D.J. et al. Immunity to malaria after administration of ultra-low doses of red cells infected with Plasmodium falciparum. Lancet 360, 610–617 (2002).
Riley, E.M., Wahl, S., Perkins, D.J. & Schofield, L. Regulating immunity to malaria. Parasite Immunol. 28, 35–49 (2006).
Amante, F.H. & Good, M.F. Prolonged Th1-like response generated by a Plasmodium yoelii-specific T cell clone allows complete clearance of infection in reconstituted mice. Parasite Immunol. 19, 111–126 (1997).
Amante, F.H. & Good, M.F. Experimental asexual blood stage malaria immunity. Curr. Protoc. Immunol. 29, 19.4.1–19.4.18 (2001).
Kumar, S. & Miller, L.H. Cellular mechanisms in immunity to blood stage infection. Immunol. Lett. 25, 109–114 (1990).
Vinetz, J.M. et al. Adoptive transfer of CD8+ T cells from immune animals does not transfer immunity to blood-stage Plasmodium yoelii malaria. J. Immunol. 144, 1069–1074 (1990).
Miller, L.H., Good, M.F. & Milon, G. Malaria pathogenesis. Science 264, 1878–1883 (1994).
Krajden, S., Panisko, D.M., Tobe, B., Yang, J. & Keystone, J.S. Prolonged infection with Plasmodium falciparum in a semi-immune patient. Trans. R. Soc. Trop. Med. Hyg. 85, 731–732 (1991).
Revel, M.P. et al. Plasmodium falciparum malaria after three years in a non-endemic area. Trans. R. Soc. Trop. Med. Hyg. 82, 832 (1988).
Theunissen, C. et al. Falciparum malaria in patient 9 years after leaving malaria-endemic area. Emerg. Infect. Dis. 15, 115–116 (2009).
Li, C., Seixas, E. & Langhorne, J. Rodent malarias: the mouse as a model for understanding immune responses and pathology induced by the erythrocytic stages of the parasite. Med. Microbiol. Immunol. (Berl.) 189, 115–126 (2001).
Day, C.L. et al. PD-1 expression on HIV-specific T cells is associated with T-cell exhaustion and disease progression. Nature 443, 350–354 (2006).
Betts, M.R. et al. HIV nonprogressors preferentially maintain highly functional HIV-specific CD8+ T cells. Blood 107, 4781–4789 (2006).
Migueles, S.A. et al. HIV-specific CD8+ T cell proliferation is coupled to perforin expression and is maintained in nonprogressors. Nat. Immunol. 3, 1061–1068 (2002).
Zajac, A.J. et al. Viral immune evasion due to persistence of activated T cells without effector function. J. Exp. Med. 188, 2205–2213 (1998).
Barber, D.L. et al. Restoring function in exhausted CD8 T cells during chronic viral infection. Nature 439, 682–687 (2006).
Blackburn, S.D. et al. Coregulation of CD8+ T cell exhaustion by multiple inhibitory receptors during chronic viral infection. Nat. Immunol. 10, 29–37 (2009).
Weiss, G.E. et al. The Plasmodium falciparum–specific human memory B cell compartment expands gradually with repeated malaria infections. PLoS Pathog. 6, e1000912 (2010).
Butler, N.S. et al. Superior antimalarial immunity after vaccination with late liver stage-arresting genetically attenuated parasites. Cell Host Microbe 9, 451–462 (2011).
Rai, D., Pham, N.L., Harty, J.T. & Badovinac, V.P. Tracking the total CD8 T cell response to infection reveals substantial discordance in magnitude and kinetics between inbred and outbred hosts. J. Immunol. 183, 7672–7681 (2009).
McDermott, D. & Varga, S.M. Quantifying antigen-specific CD4 T cells during a viral infection: CD4 T cell responses are larger than we think. J. Immunol. (31 October 2011) doi:10.4049/jimmunol.1102104.
Oxenius, A., Bachmann, M.F., Zinkernagel, R.M. & Hengartner, H. Virus-specific MHC-class II-restricted TCR-transgenic mice: effects on humoral and cellular immune responses after viral infection. Eur. J. Immunol. 28, 390–400 (1998).
Brown, K.E., Freeman, G.J., Wherry, E.J. & Sharpe, A.H. Role of PD-1 in regulating acute infections. Curr. Opin. Immunol. 22, 397–401 (2010).
Sharpe, A.H., Wherry, E.J., Ahmed, R. & Freeman, G.J. The function of programmed cell death 1 and its ligands in regulating autoimmunity and infection. Nat. Immunol. 8, 239–245 (2007).
Goldberg, M.V. & Drake, C.G. LAG-3 in Cancer Immunotherapy. Curr. Top. Microbiol. Immunol. 344, 269–278 (2011).
Huang, C.T. et al. Role of LAG-3 in regulatory T cells. Immunity 21, 503–513 (2004).
Achtman, A.H., Stephens, R., Cadman, E.T., Harrison, V. & Langhorne, J. Malaria-specific antibody responses and parasite persistence after infection of mice with Plasmodium chabaudi chabaudi. Parasite Immunol. 29, 435–444 (2007).
Langhorne, J. The role of CD4+ T-cells in the immune response to Plasmodium chabaudi. Parasitol. Today 5, 362–364 (1989).
Meding, S.J. & Langhorne, J. CD4+ T cells and B cells are necessary for the transfer of protective immunity to Plasmodium chabaudi chabaudi. Eur. J. Immunol. 21, 1433–1438 (1991).
Titanji, K. et al. Acute depletion of activated memory B cells involves the PD-1 pathway in rapidly progressing SIV-infected macaques. J. Clin. Invest. 120, 3878–3890 (2010).
Velu, V. et al. Enhancing SIV-specific immunity in vivo by PD-1 blockade. Nature 458, 206–210 (2009).
Crotty, S. Follicular helper CD4 T cells (TFH). Annu. Rev. Immunol. 29, 621–663 (2011).
Fahey, L.M. et al. Viral persistence redirects CD4 T cell differentiation toward T follicular helper cells. J. Exp. Med. 208, 987–999 (2011).
Wipasa, J. et al. Effect of Plasmodium yoelii exposure on vaccination with the 19-kilodalton carboxyl terminus of merozoite surface protein 1 and vice versa and implications for the application of a human malaria vaccine. Infect. Immun. 77, 817–824 (2009).
Chandele, A., Mukerjee, P., Das, G., Ahmed, R. & Chauhan, V.S. Phenotypic and functional profiling of malaria-induced CD8 and CD4 T cells during blood-stage infection with Plasmodium yoelii. Immunology 132, 273–286 (2011).
Kline, J. & Gajewski, T.F. Clinical development of mAbs to block the PD1 pathway as an immunotherapy for cancer. Curr. Opin. Investig. Drugs 11, 1354–1359 (2010).
Porichis, F. et al. Responsiveness of HIV-specific CD4 T cells to PD-1 blockade. Blood 118, 965–974 (2011).
Golden-Mason, L. et al. Negative immune regulator Tim-3 is overexpressed on T cells in hepatitis C virus infection and its blockade rescues dysfunctional CD4+ and CD8+ T cells. J. Virol. 83, 9122–9130 (2009).
Barber, D.L., Mayer-Barber, K.D., Feng, C.G., Sharpe, A.H. & Sher, A. CD4 T cells promote rather than control tuberculosis in the absence of PD-1-mediated inhibition. J. Immunol. 186, 1598–1607 (2011).
Reiley, W.W. et al. Distinct functions of antigen-specific CD4 T cells during murine Mycobacterium tuberculosis infection. Proc. Natl. Acad. Sci. USA 107, 19408–19413 (2010).
Wherry, E.J. T cell exhaustion. Nat. Immunol. 12, 492–499 (2011).
White, N.J. The treatment of malaria. N. Engl. J. Med. 335, 800–806 (1996).
Barfod, L. et al. Evasion of immunity to Plasmodium falciparum malaria by IgM masking of protective IgG epitopes in infected erythrocyte surface-exposed PfEMP1. Proc. Natl. Acad. Sci. USA 108, 12485–12490 (2011).
Wojciechowski, W. et al. Cytokine-producing effector B cells regulate type 2 immunity to H. polygyrus. Immunity 30, 421–433 (2009).
Nolz, J.C. & Harty, J.T. Protective capacity of memory CD8+ T cells is dictated by antigen exposure history and nature of the infection. Immunity 34, 781–793 (2011).
Alexander, C.M. et al. T regulatory cells participate in the control of germinal center reactions. J. Immunol. 133, 452–456 (2011).
Acknowledgements
We thank the residents of Kambila, Mali, for their participation; F. Lund (University of Rochester) for C57BL/6 Aicda−/− mice lacking the immunoglobulin heavy-chain μ-chain secretory domain; D.A.A. Vignali (St. Jude Children's Research Hospital) for hybridoma clone C9B7W; and S. Perlman, N. Schmidt and V. Badovinac for comments. Supported by the Division of Intramural Research of the National Institute of Allergy and Infectious Diseases, the US National Institutes of Health (for work in Mali; and AI085515 and AI42767 for work in the J.T.H. laboratory) and the Department of Microbiology, University of Iowa (for work in the J.T.H. laboratory).
Author information
Authors and Affiliations
Contributions
N.S.B. and J.M. designed the experiments, did the work, analyzed the data and wrote the manuscript; T.J.W., P.D.C. and J.T.H. designed the experiments, analyzed the data and wrote the manuscript; B.T. and O.K.D. coordinated the field studies and study site participants; and L.L.P. and L.T.T. did the histological studies and analyzed the data.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 (PDF 2347 kb)
Rights and permissions
About this article
Cite this article
Butler, N., Moebius, J., Pewe, L. et al. Therapeutic blockade of PD-L1 and LAG-3 rapidly clears established blood-stage Plasmodium infection. Nat Immunol 13, 188–195 (2012). https://doi.org/10.1038/ni.2180
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.2180
This article is cited by
-
CAR-tropic extracellular vesicles carry tumor-associated antigens and modulate CAR T cell functionality
Scientific Reports (2023)
-
Cytolytic circumsporozoite-specific memory CD4+ T cell clones are expanded during Plasmodium falciparum infection
Nature Communications (2023)
-
The foundations of immune checkpoint blockade and the ipilimumab approval decennial
Nature Reviews Drug Discovery (2022)
-
The plasma membrane calcium ATPase 4 does not influence parasite levels but partially promotes experimental cerebral malaria during murine blood stage malaria
Malaria Journal (2021)
-
Impact of immune checkpoint molecules on FoxP3+ Treg cells and related cytokines in patients with acute and chronic brucellosis
BMC Infectious Diseases (2021)