Abstract
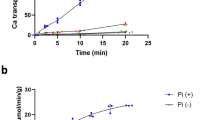
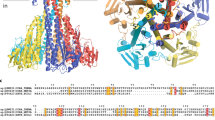
Eukaryotic Ca2+ regulation involves sequestration into intracellular organelles, and expeditious Ca2+ release into the cytosol is a hallmark of key signalling transduction pathways. Bulk removal of Ca2+ after such signalling events is accomplished by members of the Ca2+:cation (CaCA) superfamily1,2,3,4,5. The CaCA superfamily includes the Na+/Ca2+ (NCX) and Ca2+/H+ (CAX) antiporters, and in mammals the NCX and related proteins constitute families SLC8 and SLC24, and are responsible for the re-establishment of Ca2+ resting potential in muscle cells, neuronal signalling and Ca2+ reabsorption in the kidney1,6. The CAX family members maintain cytosolic Ca2+ homeostasis in plants and fungi during steep rises in intracellular Ca2+ due to environmental changes, or following signal transduction caused by events such as hyperosmotic shock, hormone response and response to mating pheromones7,8,9,10,11,12,13. The cytosol-facing conformations within the CaCA superfamily are unknown, and the transport mechanism remains speculative. Here we determine a crystal structure of the Saccharomyces cerevisiae vacuolar Ca2+/H+ exchanger (Vcx1) at 2.3 Å resolution in a cytosol-facing, substrate-bound conformation. Vcx1 is the first structure, to our knowledge, within the CAX family, and it describes the key cytosol-facing conformation of the CaCA superfamily, providing the structural basis for a novel alternating access mechanism by which the CaCA superfamily performs high-throughput Ca2+ transport across membranes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Crespo, L. M., Grantham, C. J. & Cannell, M. B. Kinetics, stoichiometry and role of the Na-Ca exchange mechanism in isolated cardiac myocytes. Nature 345, 618–621 (1990)
Cui, J. et al. Simulating calcium influx and free calcium concentrations in yeast. Cell Calcium 45, 123–132 (2009)
Miseta, A., Kellermayer, R., Aiello, D. P., Fu, L. & Bedwell, D. M. The vacuolar Ca2+/H+ exchanger Vcx1p/Hum1p tightly controls cytosolic Ca2+ levels in S. cerevisiae. FEBS Lett. 451, 132–136 (1999)
Philipson, K. D. & Nicoll, D. A. Sodium-calcium exchange: a molecular perspective. Annu. Rev. Physiol. 62, 111–133 (2000)
Nicoll, D. A., Longoni, S. & Philipson, K. D. Molecular cloning and functional expression of the cardiac sarcolemmal Na+-Ca2+ exchanger. Science 250, 562–565 (1990)
Lytton, J. Na+/Ca2+ exchangers: three mammalian gene families control Ca2+ transport. Biochem. J. 406, 365–382 (2007)
Hirschi, K. D., Zhen, R. G., Cunningham, K. W., Rea, P. A. & Fink, G. R. CAX1, an H+/Ca2+ antiporter from Arabidopsis. Proc. Natl Acad. Sci. USA 93, 8782–8786 (1996)
Pozos, T. C., Sekler, I. & Cyert, M. S. The product of HUM1, a novel yeast gene, is required for vacuolar Ca2+/H+ exchange and is related to mammalian Na+/Ca2+ exchangers. Mol. Cell. Biol. 16, 3730–3741 (1996)
Cheng, N.-H., Pittman, J. K., Barkla, B. J., Shigaki, T. & Hirschi, K. D. The Arabidopsis cax1 mutant exhibits impaired ion homeostasis, development, and hormonal responses and reveals interplay among vacuolar transporters. Plant Cell 15, 347–364 (2003)
Cho, D. et al. Vacuolar CAX1 and CAX3 influence auxin transport in guard cells via regulation of apoplastic pH. Plant Physiol. 160, 1293–1302 (2012)
Cunningham, K. W. Acidic calcium stores of Saccharomyces cerevisiae. Cell Calcium 50, 129–138 (2011)
Shigaki, T., Rees, I., Nakhleh, L. & Hirschi, K. D. Identification of three distinct phylogenetic groups of CAX cation/proton antiporters. J. Mol. Evol. 63, 815–825 (2006)
Denis, V. & Cyert, M. S. Internal Ca2+ release in yeast is triggered by hypertonic shock and mediated by a TRP channel homologue. J. Cell Biol. 156, 29–34 (2002)
Schwarz, E. M. & Benzer, S. Calx, a Na-Ca exchanger gene of Drosophila melanogaster. Proc. Natl Acad. Sci. USA 94, 10249–10254 (1997)
Cai, X. & Lytton, J. The cation/Ca2+ exchanger superfamily: phylogenetic analysis and structural implications. Mol. Biol. Evol. 21, 1692–1703 (2004)
Iwamoto, T. et al. Unique topology of the internal repeats in the cardiac Na+/Ca2+ exchanger. FEBS Lett. 446, 264–268 (1999)
Liao, J. et al. Structural insight into the ion-exchange mechanism of the sodium/calcium exchanger. Science 335, 686–690 (2012)
Nicoll, D. A., Hryshko, L. V., Matsuoka, S., Frank, J. S. & Philipson, K. D. Mutation of amino acid residues in the putative transmembrane segments of the cardiac sarcolemmal Na+-Ca2+ exchanger. J. Biol. Chem. 271, 13385–13391 (1996)
Winkfein, R. J. et al. Scanning mutagenesis of the alpha repeats and of the transmembrane acidic residues of the human retinal cone Na/Ca-K exchanger. Biochemistry 42, 543–552 (2003)
Kang, K.-J. Residues contributing to the Ca2+ and K+ binding pocket of the NCKX2 Na+/Ca2+-K+ exchanger. J. Biol. Chem. 280, 6823–6833 (2005)
Pittman, J. K., Sreevidya, C. S., Shigaki, T., Ueoka-Nakanishi, H. & Hirschi, K. D. Distinct N-terminal regulatory domains of Ca2+/H+ antiporters. Plant Physiol. 130, 1054–1062 (2002)
Pittman, J. K. & Hirschi, K. D. Regulation of CAX1, an Arabidopsis Ca2+/H+ antiporter. Identification of an N-terminal autoinhibitory domain. Plant Physiol. 127, 1020–1029 (2001)
Ivey, D. M. et al. Cloning and characterization of a putative Ca2+/H+ antiporter gene from Escherichia coli upon functional complementation of Na+/H+ antiporter-deficient strains by the overexpressed gene. J. Biol. Chem. 268, 11296–11303 (1993)
Ohsumi, Y. & Anraku, Y. Calcium transport driven by a proton motive force in vacuolar membrane vesicles of Saccharomyces cerevisiae. J. Biol. Chem. 258, 5614–5617 (1983)
Dunn, T., Gable, K. & Beeler, T. Regulation of cellular Ca2+ by yeast vacuoles. J. Biol. Chem. 269, 7273–7278 (1994)
Kamiya, T. & Maeshima, M. Residues in internal repeats of the rice cation/H+ exchanger are involved in the transport and selection of cations. J. Biol. Chem. 279, 812–819 (2004)
Shigaki, T. et al. Identification of a crucial histidine involved in metal transport activity in the Arabidopsis cation/H+ exchanger CAX1. J. Biol. Chem. 280, 30136–30142 (2005)
Matsuoka, S., Nicoll, D. A., Reilly, R. F., Hilgemann, D. W. & Philipson, K. D. Initial localization of regulatory regions of the cardiac sarcolemmal Na+-Ca2+ exchanger. Proc. Natl Acad. Sci. USA 90, 3870–3874 (1993)
Nicoll, D. A. et al. The crystal structure of the primary Ca2+ sensor of the Na+/Ca2+ exchanger reveals a novel Ca2+ binding motif. J. Biol. Chem. 281, 21577–21581 (2006)
Martínez-Muñoz, G. A. & Kane, P. Vacuolar and plasma membrane proton pumps collaborate to achieve cytosolic pH homeostasis in yeast. J. Biol. Chem. 283, 20309–20319 (2008)
Li, M. et al. Selecting optimum eukaryotic integral membrane proteins for structure determination by rapid expression and solubilization screening. J. Mol. Biol. 385, 820–830 (2009)
Ridilla, M., Narayanan, A., Bolin, J. T. & Yernool, D. A. Identification of the dimer interface of a bacterial Ca2+/H+ antiporter. Biochemistry 51, 9603–9611 (2012)
Grynkiewicz, G., Poenie, M. & Tsien, R. Y. A new generation of Ca2+ indicators with greatly improved fluorescence properties. J. Biol. Chem. 260, 3440–3450 (1985)
Caffrey, M. & Cherezov, V. Crystallizing membrane proteins using lipidic mesophases. Nature Protocols 4, 706–731 (2009)
Kabsch, W. XDS. Acta Crystallogr. D 66, 125–132 (2010)
Adams, P. D. et al. PHENIX : a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D 66, 213–221 (2010)
Winn, M. D. et al. Overview of the CCP4 suite and current developments. Acta Crystallogr. 67, 235–242 (2011)
Vonrhein, C., Blanc, E., Roversi, P. & Bricogne, G. Automated structure solution with autoSHARP. Methods Mol. Biol. 364, 215–230 (2007)
Terwilliger, T. C. Maximum-likelihood density modification. Acta Crystallogr. D 56, 965–972 (2000)
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004)
Šali, A. & Blundell, T. L. Comparative protein modelling by satisfaction of spatial restraints. J. Mol. Biol. 234, 779–815 (1993)
Pettersen, E. F. et al. UCSF Chimera—a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004)
Pei, J., Kim, B.-H. & Grishin, N. V. PROMALS3D: a tool for multiple protein sequence and structure alignments. Nucleic Acids Res. 36, 2295–2300 (2008)
The PyMOL Molecular Graphics System, Version 1.5.0.4 Schrödinger, LLC
Baker, N. A., Sept, D., Joseph, S., Holst, M. J. & McCammon, J. A. Electrostatics of nanosystems: Application to microtubules and the ribosome. Proc. Natl Acad. Sci. USA 98, 10037–10041 (2001)
Hess, B., Kutzner, C., Van der Spoel, D. & Lindahl, E. GROMACS 4: Algorithms for highly efficient, load-balanced, and scalable molecular simulation. J. Chem. Theory Comput. 4, 435–447 (2008)
Bjelkmar, P., Larsson, P., Cuendet, M. A., Hess, B. & Lindahl, E. Implementation of the CHARMM force field in GROMACS: Analysis of protein stability effects from correction maps, virtual interaction sites, and water models. J. Chem. Theory Comput. 6, 459–466 (2010)
Jorgensen, W. L., Chandrasekhar, J., Madura, J. D., Impey, R. W. & Klein, M. L. Comparison of simple potential functions for simulating liquid water. J. Chem. Phys. 79, 926–935 (1983)
Lomize, M. A., Pogozheva, I. D., Joo, H., Mosberg, H. I. & Lomize, A. L. OPM database and PPM web server: resources for positioning of proteins in membranes. Nucleic Acids Res. 40, D370–D376 (2012)
Jo, S., Lim, J. B., Klauda, J. B. & Im, W. CHARMM-GUI Membrane Builder for mixed bilayers and its application to yeast membranes. Biophys. J. 97, 50–58 (2009)
Berendsen, H. J. C., Postma, J. P. M., Van Gunsteren, W. F., DiNola, A. & Haak, J. R. Molecular dynamics with coupling to an external bath. J. Chem. Phys. 81, 3684–3690 (1984)
Bussi, G., Donadio, D. & Parrinello, M. Canonical sampling through velocity rescaling. J. Chem. Phys. 126, 014101–014101–7 (2007)
Parrinello, M. & Rahman, A. Polymorphic transitions in single crystals: A new molecular dynamics method. J. Appl. Phys. 52, 7182–7190 (1981)
Acknowledgements
We thank J. Holton, G. Meigs, C. Ogata, N. Venugopalan and T. Doukov for assistance with synchrotron data collection at Advanced Light Source, Advanced Photon Source and Stanford Synchrotron Radiation Lightsource; and C. Waddling and P. Wassam for technical assistance. B.P.P. was supported by a postdoctoral fellowship from the Carlsberg Foundation and later by a fellowship from the Danish Cancer Society; A.S. by NIH grants U54 GM094625 and U01 GM61390; R.M.S. by NIH grants U54 GM094625, GM24485 and GM073210.
Author information
Authors and Affiliations
Contributions
A.B.W. optimized the yeast expression system, performed expression, purification and crystallization experiments, collected and processed the data, determined, refined and analysed the structure, and performed reconstitution and transport assays. B.P.P. performed data collection and assisted with structure solution and structural analysis. B.H.C. and A.J.R. assisted in cell collection, membrane preparation and purification experiments. B.H.C. and Z.R.-Z. did cloning and expression tests. A.Sc. constructed Vcx1 comparative models as well as performed bioinformatics and distance plot analysis. M.B. performed molecular dynamics simulations and distance plot analysis. A.B.W., B.P.P. and R.M.S. wrote the paper with input from A.Sc., M.B. and A.Sa.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-9, Supplementary Table 1, Supplementary Methods and Supplementary References. (PDF 14435 kb)
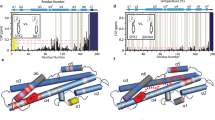
The two-stroke mechanism of alternating access for the VCX1 protein
Interpolation between the VCX1 crystal structure (cytosol-facing) and the VCX1 comparative model based on the mjNCX structure (vacuole-facing) describe the proposed mechanism of alternating access. Translational movements in the M1/M6 'piston' are coordinated with M2a and M7a to cover and uncover the active site to alternating sides of the vacuolar membrane. The proton gradient across the vacuole provides energy which results in a conformational change to the cytosol-facing state. Under conditions of high cytosolic calcium concentrations, Ca2+ ions are coordinated by the acidic helix and Ca2+ can reach the active site. Glu106 coordination to the active site Ca2+ initiates M2a straightening and M1/M6 translation to expose the active site Ca2+ to the vacuole. The vacuole-facing conformation, in combination with acidic interior of the vacuole allows for lowered affinity and release of the bound Ca2+. (MP4 15399 kb)
Rights and permissions
About this article
Cite this article
Waight, A., Pedersen, B., Schlessinger, A. et al. Structural basis for alternating access of a eukaryotic calcium/proton exchanger. Nature 499, 107–110 (2013). https://doi.org/10.1038/nature12233
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature12233
This article is cited by
-
Ca2+ efflux facilitated by co-transport of inorganic phosphate anion in the H+/Ca2+ antiporter YfkE
Communications Biology (2023)
-
Structural insight into the allosteric inhibition of human sodium-calcium exchanger NCX1 by XIP and SEA0400
The EMBO Journal (2023)
-
Genome-wide analysis of the apple CaCA superfamily reveals that MdCAX proteins are involved in the abiotic stress response as calcium transporters
BMC Plant Biology (2021)
-
Fluorescent Silicon-based Nanomaterials Imaging Technology in Diseases
Chemical Research in Chinese Universities (2021)
-
Phylogenetic analysis and protein structure modelling identifies distinct Ca2+/Cation antiporters and conservation of gene family structure within Arabidopsis and rice species
Rice (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.