Abstract
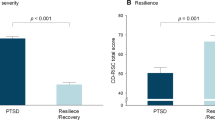
Many survivors of severe disasters, even those without posttraumatic stress disorder (PTSD), need psychological support. To understand the pathogenesis of PTSD symptoms and prevent the development of PTSD, the critical issue is to distinguish neurological abnormalities as vulnerability factors from acquired signs of PTSD symptoms in the early stage of adaptation to the trauma in the normal population. The neurological underpinnings of PTSD have been well characterized, but the causal relationships with the traumatic event are still unclear. We examined 42 non-PTSD subjects to find brain morphometric changes related to the severity of PTSD symptoms in a longitudinal magnetic resonance imaging study extending through the Great East Japan Earthquake. We found that regional grey matter volume (rGMV) in the right ventral anterior cingulate cortex (ACC) before the earthquake, and decreased rGMV in the left orbitofrontal cortex (OFC) through the earthquake were negatively associated with PTSD symptoms. Our results indicate that subjects with smaller GMV in the ACC before the earthquake, and subjects with decreased GMV in the OFC through the earthquake were likely to have PTSD symptoms. As the ACC is involved in processing of fear and anxiety, our results indicate that these processing are related to vulnerability for PTSD symptoms. In addition, decreased OFC volume was induced by failing to extinct conditioned fear soon after the traumatic event. These findings provide a better understanding of posttraumatic responses in early stage of adaptation to the trauma and may contribute to the development of effective methods to prevent PTSD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Fukuda S, Morimoto K, Mure K, Maruyama S . Posttraumatic stress and change in lifestyle among the Hanshin-Awaji earthquake victims. Prev Med 1999; 29: 147–151.
Kessler RC, Sonnega A, Bromet E, Hughes M, Nelson CB . Posttraumatic stress disorder in the National Comorbidity Survey. Arch Gen Psychiatry 1995; 52: 1048–1060.
Perrin MA, DiGrande L, Wheeler K, Thorpe L, Farfel M, Brackbill R . Differences in PTSD prevalence and associated risk factors among World Trade Center disaster rescue and recovery workers. Am J Psychiatry 2007; 164: 1385–1394.
Furukawa K, Arai H . Earthquake in Japan. Lancet 2011; 377: 1652.
Pitman RK, Gilbertson MW, Gurvits TV, May FS, Lasko NB, Metzger LJ et al. Clarifying the origin of biological abnormalities in PTSD through the study of identical twins discordant for combat exposure. Ann N Y Acad Sci 2006; 1071: 242–254.
Karl A, Schaefer M, Malta LS, Dorfel D, Rohleder N, Werner A . A meta-analysis of structural brain abnormalities in PTSD. Neurosci Biobehav Rev 2006; 30: 1004–1031.
Yamasue H, Kasai K, Iwanami A, Ohtani T, Yamada H, Abe O et al. Voxel-based analysis of MRI reveals anterior cingulate gray-matter volume reduction in posttraumatic stress disorder due to terrorism. Proc Natl Acad Sci USA 2003; 100: 9039–9043.
Kasai K, Yamasue H, Gilbertson MW, Shenton ME, Rauch SL, Pitman RK . Evidence for acquired pregenual anterior cingulate gray matter loss from a twin study of combat-related posttraumatic stress disorder. Biol Psychiatry 2008; 63: 550–556.
Woodward SH, Kaloupek DG, Streeter CC, Martinez C, Schaer M, Eliez S . Decreased anterior cingulate volume in combat-related PTSD. Biol Psychiatry 2006; 59: 582–587.
Chen S, Xia W, Li L, Liu J, He Z, Zhang Z et al. Gray matter density reduction in the insula in fire survivors with posttraumatic stress disorder: a voxel-based morphometric study. Psychiatry Res 2006; 146: 65–72.
Hakamata Y, Matsuoka Y, Inagaki M, Nagamine M, Hara E, Imoto S et al. Structure of orbitofrontal cortex and its longitudinal course in cancer-related post-traumatic stress disorder. Neurosci Res 2007; 59: 383–389.
Woodward SH, Schaer M, Kaloupek DG, Cediel L, Eliez S . Smaller global and regional cortical volume in combat-related posttraumatic stress disorder. Arch Gen Psychiatry 2009; 66: 1373–1382.
Eckart C, Stoppel C, Kaufmann J, Tempelmann C, Hinrichs H, Elbert T et al. Structural alterations in lateral prefrontal, parietal and posterior midline regions of men with chronic posttraumatic stress disorder. J Psychiatry Neurosci 2011; 36: 176–186.
Papagni SA, Benetti S, Arulanantham S, McCrory E, McGuire P, Mechelli A . Effects of stressful life events on human brain structure: a longitudinal voxel-based morphometry study. Stress 2011; 14: 227–232.
Ganzel BL, Kim P, Glover GH, Temple E . Resilience after 9/11: multimodal neuroimaging evidence for stress-related change in the healthy adult brain. Neuroimage 2008; 40: 788–795.
Gilbertson MW, Shenton ME, Ciszewski A, Kasai K, Lasko NB, Orr SP et al. Smaller hippocampal volume predicts pathologic vulnerability to psychological trauma. Nat Neurosci 2002; 5: 1242–1247.
Bonne O, Brandes D, Gilboa A, Gomori JM, Shenton ME, Pitman RK et al. Longitudinal MRI study of hippocampal volume in trauma survivors with PTSD. Am J Psychiatry 2001; 158: 1248–1251.
De Bellis MD, Hall J, Boring AM, Frustaci K, Moritz G . A pilot longitudinal study of hippocampal volumes in pediatric maltreatment-related posttraumatic stress disorder. Biol Psychiatry 2001; 50: 305–309.
Asukai N, NishizonoMaher A . The Japanese Version of Clinician-Administered PTSD Scale Tokyo. Tokyo Institute of Psychiatry, 1998.
Sheehan DV, Lecrubier Y, Sheehan KH, Amorim P, Janavs J, Weiller E et al. The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry 1998; 59 (Suppl 20): 22–33, ; quiz 4–57.
Otsubo T, Tanaka K, Koda R, Shinoda J, Sano N, Tanaka S et al. Reliability and validity of Japanese version of the Mini-International Neuropsychiatric Interview. Psychiatry Clin Neurosci 2005; 59: 517–526.
Oldfield RC . The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia 1971; 9: 97–113.
World Medical Association. Declaration of Helsinki. Law Med Health Care 1991; 19: 264–265.
Blake DD, Weathers FW, Nagy LM, Kaloupek DG, Gusman FD, Charney DS et al. The development of a Clinician-Administered PTSD Scale. J Trauma Stress 1995; 8: 75–90.
Weathers FW, Keane TM, Davidson JR . Clinician-administered PTSD scale: a review of the first ten years of research. Depress Anxiety 2001; 13: 132–156.
Spielberger C, Gorsuch R, Lushene R, Vagg P, Jacobs G . Manual for the State-Trait Anxiety Inventory. Palo Alto, CA: Consulting Psychologists Press, 1983.
Nakazato K, Mizuguchi T . How to use STAI. Kyoto: Sankyoubou Corp, 1982.
Radloff L . The CES-D scale: a self-report depression scale for research in the general population. Appl Psychol Meas 1977; 1: 385–401.
Shima S, Shikano T, Kitamura T, Asai M . New self-rated scale for depression (in Japanese). Japn J Clin Psychiatry 1985; 27: 717–723.
Gaser C . VBM Toolbox for SPM2, VBM Toolbox for SPM5. 2007 ; Available from: http://dbm.neuro.uni-jena.de/vbm/.
Good CD, Johnsrude IS, Ashburner J, Henson RN, Friston KJ, Frackowiak RS . A voxel-based morphometric study of ageing in 465 normal adult human brains. Neuroimage 2001; 14 (1 Part 1): 21–36.
Ashburner J, Friston KJ . Voxel-based morphometry—the methods. Neuroimage 2000; 11 (6 Part 1): 805–821.
Worsley KJ, Marrett S, Neelin P, Vandal AC, Friston KJ, Evans AC . A unified statistical approach for determining significant signals in images of cerebral activation. Hum Brain Mapp 1996; 4: 58–73.
Lancaster JL, Woldorff MG, Parsons LM, Liotti M, Freitas CS, Rainey L et al. Automated Talairach atlas labels for functional brain mapping. Hum Brain Mapp 2000; 10: 120–131.
Maldjian JA, Laurienti PJ, Kraft RA, Burdette JH . An automated method for neuroanatomic and cytoarchitectonic atlas-based interrogation of fMRI data sets. Neuroimage 2003; 19: 1233–1239.
Tzourio-Mazoyer N, Landeau B, Papathanassiou D, Crivello F, Etard O, Delcroix N et al. Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage 2002; 15: 273–289.
van Wingen GA, Geuze E, Vermetten E, Fernandez G . Perceived threat predicts the neural sequelae of combat stress. Mol Psychiatry 2011; 16: 664–671.
Kroes MC, Rugg MD, Whalley MG, Brewin CR . Structural brain abnormalities common to posttraumatic stress disorder and depression. J Psychiatry Neurosci 2011; 36: 256–265.
Shin LM, Bush G, Milad MR, Lasko NB, Brohawn KH, Hughes KC et al. Exaggerated activation of dorsal anterior cingulate cortex during cognitive interference: a monozygotic twin study of posttraumatic stress disorder. Am J Psychiatry 2011; 168: 979–985.
Thomaes K, Dorrepaal E, Draijer N, de Ruiter MB, Elzinga BM, Sjoerds Z et al. Increased anterior cingulate cortex and hippocampus activation in Complex PTSD during encoding of negative words. Soc Cogn Affect Neurosci; advance online publication, 14 December 2011; doi:10.1093/scan/nsr.084.
Etkin A, Egner T, Kalisch R . Emotional processing in anterior cingulate and medial prefrontal cortex. Trends Cogn Sci 2011; 15: 85–93.
Lanius RA, Bluhm R, Lanius U, Pain C . A review of neuroimaging studies in PTSD: heterogeneity of response to symptom provocation. J Psychiatr Res 2006; 40: 709–729.
Cloninger CR, Svrakic DM, Przybeck TR . A psychobiological model of temperament and character. Arch Gen Psychiatry 1993; 50: 975–990.
Richman H, Frueh BC . Personality and PTSD II: personality assessment of PTSD-diagnosed Vietnam veterans using the cloninger tridimensional personality questionnaire (TPQ). Depress Anxiety 1997; 6: 70–77.
Pujol J, Lopez A, Deus J, Cardoner N, Vallejo J, Capdevila A et al. Anatomical variability of the anterior cingulate gyrus and basic dimensions of human personality. Neuroimage 2002; 15: 847–855.
Frewen PA, Pain C, Dozois DJ, Lanius RA . Alexithymia in PTSD: psychometric and FMRI studies. Ann N Y Acad Sci 2006; 1071: 397–400.
Borsci G, Boccardi M, Rossi R, Rossi G, Perez J, Bonetti M et al. Alexithymia in healthy women: a brain morphology study. J Affect Disord 2009; 114: 208–215.
Butter CM, Mishkin M, Rosvold HE . Conditioning and extinction of a food-rewarded response after selective ablations of frontal cortex in rhesus monkeys. Exp Neurol 1963; 7: 65–75.
Rolls ET, Hornak J, Wade D, McGrath J . Emotion-related learning in patients with social and emotional changes associated with frontal lobe damage. J Neurol Neurosurg Psychiatry 1994; 57: 1518–1524.
Gottfried JA, Dolan RJ . Human orbitofrontal cortex mediates extinction learning while accessing conditioned representations of value. Nat Neurosci 2004; 7: 1144–1152.
Ochsner KN, Gross JJ . The cognitive control of emotion. Trends Cogn Sci 2005; 9: 242–249.
Davidson RJ, Putnam KM, Larson CL . Dysfunction in the neural circuitry of emotion regulation--a possible prelude to violence. Science 2000; 289: 591–594.
Dolcos F, McCarthy G . Brain systems mediating cognitive interference by emotional distraction. J Neurosci 2006; 26: 2072–2079.
Bremner JD, Vermetten E, Schmahl C, Vaccarino V, Vythilingam M, Afzal N et al. Positron emission tomographic imaging of neural correlates of a fear acquisition and extinction paradigm in women with childhood sexual-abuse-related post-traumatic stress disorder. Psychol Med 2005; 35: 791–806.
New AS, Fan J, Murrough JW, Liu X, Liebman RE, Guise KG et al. A functional magnetic resonance imaging study of deliberate emotion regulation in resilience and posttraumatic stress disorder. Biol Psychiatry 2009; 66: 656–664.
Carrion VG, Weems CF, Richert K, Hoffman BC, Reiss AL . Decreased prefrontal cortical volume associated with increased bedtime cortisol in traumatized youth. Biol Psychiatry 2010; 68: 491–493.
Cohen RA, Grieve S, Hoth KF, Paul RH, Sweet L, Tate D et al. Early life stress and morphometry of the adult anterior cingulate cortex and caudate nuclei. Biol Psychiatry 2006; 59: 975–982.
Gianaros PJ, Jennings JR, Sheu LK, Greer PJ, Kuller LH, Matthews KA . Prospective reports of chronic life stress predict decreased grey matter volume in the hippocampus. Neuroimage 2007; 35: 795–803.
Lui S, Huang X, Chen L, Tang H, Zhang T, Li X et al. High-field MRI reveals an acute impact on brain function in survivors of the magnitude 8.0 earthquake in China. Proc Natl Acad Sci USA 2009; 106: 15412–15417.
McCurry J . Anxiety over radiation exposure remains high in Japan. Lancet 2011; 378: 1061–1062.
Wright IC, Ellison ZR, Sharma T, Friston KJ, Murray RM, McGuire PK . Mapping of grey matter changes in schizophrenia. Schizophr Res 1999; 35: 1–14.
Acknowledgements
We thank the study participants and all of our colleagues in the Institute of Development, Aging, and Cancer and in Tohoku University for their support. We also thank A Omoto, N Aikawa, N Saito and Y Watanabe for conducting the CAPS structured interview. AS was supported by a Grant-in-Aid for Young Scientists (B) (KAKENHI 22790611, 24790653) from the Ministry of Education, Culture, Sports, Science and Technology, and Grants-in-Aid for Scientific Research from the Ministry of Health, Labour and Welfare in Japan from the Ministry of Education, Culture, Sports, Science and Technology in Japan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Sekiguchi, A., Sugiura, M., Taki, Y. et al. Brain structural changes as vulnerability factors and acquired signs of post-earthquake stress. Mol Psychiatry 18, 618–623 (2013). https://doi.org/10.1038/mp.2012.51
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/mp.2012.51
Keywords
This article is cited by
-
Effects of lockdowns on neurobiological and psychometric parameters in unipolar depression during the COVID-19 pandemic
Translational Psychiatry (2024)
-
Longitudinal volumetric evaluation of hippocampus and amygdala subregions in recent trauma survivors
Molecular Psychiatry (2023)
-
Large-scale societal dynamics are reflected in human mood and brain
Scientific Reports (2022)
-
The brain at war: effects of stress on brain structure in soldiers deployed to a war zone
Translational Psychiatry (2021)
-
Neural contributors to trauma resilience: a review of longitudinal neuroimaging studies
Translational Psychiatry (2021)