Abstract
PAX8 is expressed in thymic epithelial neoplasms and a subset of neuroendocrine carcinomas of gastrointestinal origin but not pulmonary neuroendocrine carcinomas. Thyroid transcription factor 1 (TTF-1) is known to be positive in pulmonary neuroendocrine carcinomas, but studies investigating its expression in thymic neuroendocrine carcinomas are lacking. To date, there are no comprehensive studies focusing on the comparative expression of PAX8 or TTF-1 in pulmonary and thymic neuroendocrine carcinoma. Twenty-five cases of low and intermediate grade neuroendocrine carcinomas of pulmonary and thymic origin, respectively, were selected for immunohistochemical studies using antibodies directed against PAX8 and TTF-1. The percentage of positive tumor cells as well as the intensity of staining were evaluated and scored. Twenty-one of the pulmonary neuroendocrine carcinomas were classified as low grade (typical carcinoid) and 4 as intermediate grade (atypical carcinoid) tumors; the thymic tumors consisted of 8 low grade and 17 intermediate grade neuroendocrine carcinomas. Only 2 (8%) of the pulmonary tumors showed nuclear expression of PAX8 while 19 (76%) expressed TTF-1. Of the thymic tumors, 8 (32%) were positive for PAX8 and 2 (8%) showed TTF-1 positivity. Primary neuroendocrine carcinomas of the thymus are rare neoplasms that display a more aggressive clinical course than pulmonary neuroendocrine carcinomas, highlighting the importance of the separation of these tumors. To date, there are no specific immunomarkers to distinguish between neuroendocrine carcinomas of pulmonary and thymic origin. The differential expression of PAX8 and TTF-1 may prove useful in this context as a PAX8+/TTF-1− immunophenotype appears to be more common in thymic neuroendocrine carcinomas, whereas the reverse (PAX8−/TTF-1+) is true for most pulmonary neuroendocrine carcinomas.
Similar content being viewed by others
Main
Neuroendocrine carcinomas are a group of neoplasms that can occur in many organ systems, with the highest incidence in the lung (30%) and intestinal tract (30%), while primary thymic neuroendocrine carcinomas account for only 0.4% of all neuroendocrine tumors.1 In general, the pathological spectrum of these tumors ranges from low to high grade neuroendocrine carcinomas, although their classification is not uniform throughout the body but is rather defined by organ site and, to date, is still an evolving process. In the lung and thymus, low grade lesions correspond to ‘typical carcinoid tumors’, intermediate grade tumors to ‘atypical carcinoid tumors’ and high grade malignancies are divided into large cell and small cell neuroendocrine carcinomas.2, 3 Compared with other tumors, neuroendocrine carcinomas are rare in the lung representing about 1% of all primary lung neoplasms4 and are extremely rare in the thymic gland where they account for <5% of all mediastinal tumors.5 Similar morphology and routine immunohistochemical markers often do not differentiate between neuroendocrine carcinomas of different locations, thus making it difficult to establish the exact site of origin in cases of metastatic tumors. Therefore, the objective of this study was to compare the staining patterns of two immunohistochemical markers associated with lung and thymic tumors, thyroid transcription factor-1 (TTF-1) and PAX8 in 25 cases of low and intermediate grade neuroendocrine carcinomas of pulmonary and thymic origin, respectively, in order to evaluate their use for the differential diagnosis of these tumors.
Materials and methods
Twenty-five cases of pulmonary and thymic neuroendocrine carcinomas, respectively, from the surgical pathology files of MD Anderson Cancer Center, Houston, TX, USA form the basis of this study. The tissue was obtained from resection specimens in all cases. The pulmonary tumors were classified as 21 low grade (typical carcinoids) and 4 intermediate grade (atypical carcinoids) neuroendocrine carcinomas based on the classification schema proposed by Moran et al.2 The tumors of the thymic gland were categorized as 8 low grade (typical carcinoids) and 17 intermediate grade (atypical carcinoids) neuroendocrine carcinomas according to Moran and Suster.3 Before this study, all tumors were confirmed as neuroendocrine carcinomas using pancytokeratin, synaptophysin and chromogranin A immunohistochemical stains. In addition, clinical and radiological correlation had confirmed their respective primary sites. For the purpose of the present study, representative unstained sections obtained from formalin-fixed and paraffin-embedded blocks were used to perform immunohistochemical studies. The sections were incubated with 3% H2O2 in methanol and fetal bovine serum to block endogenous peroxidase activity and non-specific protein–protein interactions, respectively. Pretreatment consisted of Dako High ph Target Retrival (ph 9). Immunostaining was performed using a horseradish peroxidase-labelled polymer system. Tissue sections were incubated with TTF-1 (Dako, Carpinteria, CA, USA: 1:200) and PAX8 (Proteintech Group, Chicago, IL, USA; 1:100) antibodies for 65 and 45 min at room temperature, respectively. Diaminobenzidine was used as a chromogen for antigen localization. Adequate positive and negative controls were run, respectively. Nuclear staining in the tumor cells was scored on a sliding scale of 0 to 4+ according to the percentage of reactive cells (0, negative; 1+, 1–25%; 2+, 26–50%; 3+, 51–75%; 4+, 76–100%). The staining intensity was graded as weak, intermediate or strong. Cytoplasmic staining alone was scored as negative. Clinical and follow-up information were obtained from the patients’ charts or from the referring physicians.
Results
Clinical Features
Among the patients with pulmonary neuroendocrine carcinomas were 17 females and 8 males with an age range from 31 to 75 years (mean 54 years) (Table 1). Patients primarily presented with symptoms of chest pain, cough and shortness of breath. Twenty-three patients were treated with surgical resection and clinical follow-up only, while 2 patients with intermediate grade tumors received surgery followed by adjuvant chemotherapy.
The patients with thymic neuroendocrine carcinomas were 19 males and 6 females aged 20 to 87 years (mean 50 years) (Table 1). Chest pain and shortness of breath were the main symptoms, and in 3 patients the tumors were detected incidentally. Complete surgical resection was performed in all the cases.
Histological Features
The pulmonary tumors categorized as low grade neuroendocrine carcinomas (carcinoid tumors) were tumors with a mitotic rate of ≤3 mitoses per 10 high power fields (HPF). If present, necrosis was only focally identified and did not amount to more than punctuate necrosis. The intermediate grade tumors (atypical carcinoid) differed from the lower grade tumors by the presence of 4–10 mitoses per 10 HPF and/or more extensive areas of necrosis, often of the comedo-type. Architecturally, both tumor types showed the typical neuroendocrine growth pattern characterized by a nested, organoid, trabecular or rosette-like architecture (Figure 1a); however, the intermediate grade tumors showed focal areas of more diffuse growth and an often higher degree of cytological atypia.
In a similar fashion to the pulmonary neuroendocrine carcinomas, the thymic tumors were classified according to their mitotic activity and the presence or absence of necrosis. Low grade thymic neuroendocrine carcinomas, in addition to neuroendocrine morphology, had a mitotic activity of up to 3 mitoses per 10 HPF and were allowed focal punctuate necrosis. Tumors displaying 4–10 mitoses per 10 HPF and/or the presence of more extensive areas of necrosis, more diffuse growth and increased cytological atypia were categorized as intermediate grade tumors (Figure 1b).
Immunohistochemical Features
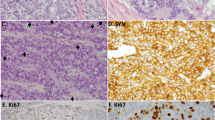
The results of the immunohistochemical study are summarized in Tables 1, 2, 3, 4, 5. In the pulmonary neuroendocrine carcinomas TTF-1 was expressed in a total of 19 of 25 cases (76%) (Figure 2a). It has to be noted, however, that although diffuse staining (≥3+) was seen in all of the positive cases, the staining intensity was always weak (score 1). Contrary to the high number of TTF-1 positive cases, PAX8-positive tumor cells were only seen in two of the pulmonary tumors (8%), both of which also demonstrated diffuse yet weak nuclear staining (Figure 2b). Both TTF-1 and PAX8 reactivity were seen in low and intermediate grade pulmonary neuroendocrine carcinomas.
(a) TTF-1 positivity is noted in a case of pulmonary low grade neuroendocrine carcinoma with spindle cell morphology. (b) Weak PAX8 reactivity was seen in two cases of pulmonary neuroendocrine carcinoma. (c) Faint TTF-1 reactivity was identified in a case of intermediate grade neuroendocrine carcinoma of the thymus. (d) Strong expression of PAX8 in an intermediate grade thymic neuroendocrine carcinoma.
The expression of these markers in the thymic tumors showed the reverse staining pattern. In these lesions, TTF-1 positivity was noted in only two of the cases (8%), one low and one intermediate grade tumor (Figure 2c). Again, the staining was diffuse in both the cases, but staining intensity was weak. PAX8, on the other hand, was found to be positive in approximately one-third of the cases (8/25; 32%) (Figure 2d). In these cases, the staining intensity ranged from weak to strong among a generally diffuse staining pattern. Again, both low and intermediate grade thymic neuroendocrine carcinomas were among the positive cases.
Clinical Follow-up
Clinical follow-up among the patients with pulmonary neuroendocrine carcinomas showed that 22 patients were alive with a follow-up ranging from 32 to 105 months (mean 68 months) and 2 patients had died 26 and 41 months after their diagnosis, both from unrelated causes.
Of the patients with thymic neuroendocrine carcinomas, only 6 were alive (follow-up range 24–204 months; mean 116 months) while 11 patients had died from their mediastinal tumors 12–228 months after their initial diagnosis (mean 79 months). Eight patients were lost to follow-up.
Discussion
Neuroendocrine carcinomas are a unique group of tumors characterized by a specific morphological appearance, ultrastructural evidence of neuroendocrine granules and immunohistochemical expression of neuroendocrine markers, such as synaptophysin or chromogranin A. Neuroendocrine carcinomas can occur throughout the body and most often originate in the pulmonary and gastrointestinal systems.1, 6, 7 All neuroendocrine carcinomas have the potential to metastasize, including those tumors that belong to the low or intermediate grade spectrum (carcinoid and atypical carcinoid tumors) despite their relatively bland morphological appearance. This can cause diagnostic difficulty when the differential diagnosis includes the possibility of metastatic disease, as the morphological and immunohistochemical features of these tumors often overlap regardless of their site of origin. This is particularly true for neuroendocrine carcinomas that present in the lung or thymic gland, as both these sites can not only harbor either primary neuroendocrine carcinomas but are also often the site of metastatic deposits. Important to note in this context is the fact that low and intermediate neuroendocrine carcinomas of the lung and thymus demonstrate very different biological behavior, making careful assessment as to their site of origin of utmost importance.
Low and intermediate grade neuroendocrine carcinomas of the lung tend to occur at a younger age than other lung carcinomas, with an average age at presentation in the sixth decade.8, 9, 10 Females are more commonly affected. For low grade pulmonary neuroendocrine carcinomas, survival rates vary from 87–100% at 5 years and 87–93% at 10 years while intermediate grade neuroendocrine carcinomas show 40–66% 5-year and 31–59% 10-year survival.9, 10, 11, 12, 13 On the other hand, low and intermediate grade neuroendocrine carcinomas arising from the thymic gland have a strong male predominance with a mean age at diagnosis of 58 years. Contrary to pulmonary neuroendocrine carcinomas, these tumors are often associated with endocrine disturbances.3 Most importantly, the survival rate for patients with thymic neuroendocrine carcinomas is significantly worse than for patients with pulmonary primaries. In the largest series on this topic,3 the survival for carcinoid tumors of the thymus was calculated at only 50% at 5 years while that rate decreased to 25% for patients with atypical carcinoid tumors. This underscores the importance of correct classification of neuroendocrine carcinomas based on their site of origin.
In the past, numerous studies have focused on how to separate pulmonary from non-pulmonary neuroendocrine carcinomas, especially those of gastrointestinal or pancreatic origin using immunohistochemical markers.7, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 In this scenario, the use of TTF-1 has attracted a lot of attention. TTF-1 is a transcription factor that has been linked with expression in lung and thyroid neoplasms.7, 24, 25, 26 The documented expression of TTF-1 in typical and atypical carcinoids of lung origin has been very variable ranging from 0 to 94% and 0 to 100%, respectively.7, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 27, 28, 29, 30, 31 This corresponds to an average expression rate of 34.3% for carcinoid tumors and 37.3% for atypical carcinoid tumors. This contrasts with neuroendocrine carcinomas of non-pulmonary origin, the majority which are reportedly negative for this marker,15, 16, 18, 19, 23 although isolated cases of low grade gastrointestinal neuroendocrine carcinoma may show some reactivity with TTF-1.14, 16 When applied to thymic neuroendocrine carcinomas, TTF-1 has shown more consistent results: all reported cases, although low in number, failed to show expression of this antibody.7, 14, 18
A novel marker that has recently attracted a lot of attention in surgical pathology is PAX8. PAX8 is a transcription factor that is commonly expressed in epithelial tumors of the thyroid gland, parathyroid gland, kidneys and Müllerian tract.24, 32, 33, 34 In addition, PAX8 has recently also been described to label neoplastic epithelial cells of thymic epithelial neoplasms.35 Interestingly, PAX8 expression has also been described in a subset of neuroendocrine neoplasms, such as pancreatic neuroendocrine tumors as well as duodenal and rectal carcinoid tumors.21, 22 In the same studies, primary pulmonary neuroendocrine carcinomas failed to stain with this marker.21, 22
The fact that the immunohistochemical staining pattern of TTF-1 and PAX8 has not been investigated for neuroendocrine carcinomas of thymic origin has led us to study the expression pattern of these markers in 25 low and intermediate grade neuroendocrine carcinomas of the thymus and compare it with that of pulmonary neuroendocrine carcinomas in order to evaluate whether these markers could assist in the identification of the primary site in the context of thoracic neuroendocrine tumors.
From a clinical point of view, the results of our study largely confirm previous observations. Age at presentation, gender predominance and survival mirrored the data of previous studies.3, 8, 9, 10, 11, 12, 13 On an immunohistochemical level, expression of TTF-1 was noted in 76% of our pulmonary neuroendocrine carcinoma cases, although the staining was often weak requiring careful evaluation of the material. Interestingly, two of the pulmonary neuroendocrine carcinomas also showed weak staining with PAX8, a finding that has not been noted before. A reverse staining pattern was noted in the primary thymic tumors. We were able to demonstrate PAX8 reactivity in up to 1/3 of the cases confirming that PAX8 expression is not only restricted to thymic epithelial cells but can also be identified in neoplastic neuroendocrine cells of the thymus. Careful evaluation is also required when TTF-1 positivity is encountered in tumors of the thymic gland. This does not automatically rule out a primary thymic process as demonstrated by two of our thymic neuroendocrine carcinomas that were positive for this marker. This finding is in support of previous observations that showed TTF-1 expression in rare cases of thymoma36, 37 and emphasizes once again the importance of close clinical correlation in addition to the use of immunohistochemical studies.
In summary, we have described the differential immunohistochemical expression pattern of TTF-1 and PAX8 in 25 cases of neuroendocrine carcinomas of the lung and thymic gland, respectively. Our results indicate that these markers may aid in the differential diagnosis of thoracic neuroendocrine carcinomas. Close evaluation of the submitted material, however, may be required due to often weak expression of these markers. In addition, both TTF-1 and PAX8 staining may be seen in a small subset of thymic and pulmonary neuroendocrine carcinomas, respectively, making close clinical correlation invaluable in the context of thoracic neuroendocrine neoplasms.
References
Modlin IM, Lye KD, Kidd MA . 5-Decade analysis of 13,715 carcinoid tumors. Cancer 2003;97:934–959.
Moran CA, Suster S, Coppola D et al. Neuroendocrine carcinomas of the lung: a critical analysis. Am J Clin Pathol 2009;131:206–221.
Moran CA, Suster S . Neuroendocrine carcinomas (carcinoid tumor) of the thymus. a clinicopathologic analysis of 80 cases. Am J Clin Pathol 2000;114:100–110.
Hage R, de la Rivière AB, Seldenrijk CA et al. Update in pulmonary carcinoid tumors: a review article. Ann Surg Oncol 2003;10:697–704.
Duh QY, Hybarger CP, Geist R et al. Carcinoids associated with multiple endocrine neoplasia syndromes. Am J Surg 1987;154:142–148.
Tomassetti P, Migliori M, Lalli S et al. Epidemiology, clinical features and diagnosis of gastroenteropancreatic endocrine tumours. Ann Oncol 2001;12 (Suppl 2):S95–S99.
Du EZ, Goldstraw P, Zacharias J et al. TTF-1 expression is specific for lung primary in typical and atypical carcinoids: TTF-1-positive carcinoids are predominantly in peripheral location. Hum Pathol 2004;35:825–831.
Modlin IM, Sandor A . An analysis of 8305 cases of carcinoid tumors. Cancer 1997;79:813–829.
Davini F, Gonfiotti A, Comin C et al. Typical and atypical carcinoid tumours: 20-year experience with 89 patients. J Cardiovasc Surg (Torino) 2009;50:807–811.
Naalsund A, Rostad H, Strøm EH et al. Carcinoid lung tumors—incidence, treatment and outcomes: a population-based study. Eur J Cardiothorac Surg 2011;39:565–569.
Travis WD, Rush W, Flieder DB et al. Survival analysis of 200 pulmonary neuroendocrine tumors with clarification of criteria for atypical carcinoid and its separation from typical carcinoid. Am J Surg Pathol 1998;22:934–944.
Skuladottir H, Hirsch FR, Hansen HH et al. Pulmonary neuroendocrine tumors: incidence and prognosis of histological subtypes. A population-based study in Denmark. Lung Cancer 2002;37:127–135.
Thomas CF Jr, Tazelaar HD, Jett JR . Typical and atypical pulmonary carcinoids: outcome in patients presenting with regional lymph node involvement. Chest 2001;119:1143–1150.
Kaufmann O, Dietel M . Expression of thyroid transcription factor-1 in pulmonary and extrapulmonary small cell carcinomas and other neuroendocrine carcinomas of various primary sites. Histopathology 2000;36:415–420.
Srivastava A, Hornick JL . Immunohistochemical staining for CDX-2, PDX-1, NESP-55, and TTF-1 can help distinguish gastrointestinal carcinoid tumors from pancreatic endocrine and pulmonary carcinoid tumors. Am J Surg Pathol 2009;33:626–632.
Cai YC, Banner B, Glickman J et al. Cytokeratin 7 and 20 and thyroid transcription factor 1 can help distinguish pulmonary from gastrointestinal carcinoid and pancreatic endocrine tumors. Hum Pathol 2001;32:1087–1093.
Sturm N, Rossi G, Lantuejoul S et al. Expression of thyroid transcription factor-1 in the spectrum of neuroendocrine cell lung proliferations with special interest in carcinoids. Hum Pathol 2002;33:175–182.
Oliveira AM, Tazelaar HD, Myers JL et al. Thyroid transcription factor-1 distinguishes metastatic pulmonary from well-differentiated neuroendocrine tumors of other sites. Am J Surg Pathol 2001;25:815–819.
Saqi A, Alexis D, Remotti F et al. Usefulness of CDX2 and TTF-1 in differentiating gastrointestinal from pulmonary carcinoids. Am J Clin Pathol 2005;123:394–404.
Matoso A, Singh K, Jacob R et al. Comparison of thyroid transcription factor-1 expression by 2 monoclonal antibodies in pulmonary and nonpulmonary primary tumors. Appl Immunohistochem Mol Morphol 2010;18:142–149.
Long KB, Srivastava A, Hirsch MS et al. PAX8 Expression in well-differentiated pancreatic endocrine tumors: correlation with clinicopathologic features and comparison with gastrointestinal and pulmonary carcinoid tumors. Am J Surg Pathol 2010;34:723–729.
Sangoi AR, Ohgami RS, Pai RK et al. PAX8 expression reliably distinguishes pancreatic well-differentiated neuroendocrine tumors from ileal and pulmonary well-differentiated neuroendocrine tumors and pancreatic acinar cell carcinoma. Mod Pathol 2011;24:412–424.
Lin X, Saad RS, Luckasevic TM et al. Diagnostic value of CDX-2 and TTF-1 expressions in separating metastatic neuroendocrine neoplasms of unknown origin. Appl Immunohistochem Mol Morphol 2007;15:407–414.
Fabbro D, Di Loreto C, Beltrami CA et al. Expression of thyroid-specific transcription factors TTF-1 and PAX-8 in human thyroid neoplasms. Cancer Res 1994;54:4744–4749.
Khoor A, Whitsett JA, Stahlman MT et al. Utility of surfactant protein B precursor and thyroid transcription factor 1 in differentiating adenocarcinoma of the lung from malignant mesothelioma. Hum Pathol 1999;30:695–700.
Saad RS, Liu YL, Han H et al. Prognostic significance of thyroid transcription factor-1 expression in both early-stage conventional adenocarcinoma and bronchioloalveolar carcinoma of the lung. Hum Pathol 2004;35:3–7.
Chang YL, Lee YC, Liao WY et al. The utility and limitation of thyroid transcription factor-1 protein in primary and metastatic pulmonary neoplasms. Lung Cancer 2004;44:149–157.
Bishop JA, Sharma R, Illei PB et al. and thyroid transcription factor-1 expression in carcinomas of the lung, breast, pancreas, colon, kidney, thyroid, and malignant mesothelioma. Hum Pathol 2010;41:20–25.
Zamecnik J, Kodet R . Value of thyroid transcription factor-1 and surfactant apoprotein A in the differential diagnosis of pulmonary carcinomas: a study of 109 cases. Virchows Arch 2002;440:353–361.
Folpe AL, Gown AM, Lamps LW et al. Thyroid transcription factor-1: immunohistochemical evaluation in pulmonary neuroendocrine tumors. Mod Pathol 1999;12:5–8.
Pelosi G, Rodriguez J, Viale G et al. Typical and atypical pulmonary carcinoid tumor overdiagnosed as small-cell carcinoma on biopsy specimens: a major pitfall in the management of lung cancer patients. Am J Surg Pathol 2005;29:179–187.
Laury AR, Perets R, Piao H et al. A comprehensive analysis of PAX8 expression in human epithelial tumors. Am J Surg Pathol 2011;35:816–826.
Tacha D, Zhou D, Cheng L . Expression of PAX8 in normal and neoplastic tissues: a comprehensive immunohistochemical study. Appl Immunohistochem Mol Morphol 2011;19:293–299.
Ozcan A, Shen SS, Hamilton C et al. PAX 8 expression in non-neoplastic tissues, primary tumors, and metastatic tumors: a comprehensive immunohistochemical study. Mod Pathol 2011;24:751–764.
Weissferdt A, Moran CA . PAX8 expression in thymic epithelial neoplasms: an immunohistochemical analysis. Am J Surg Pathol 2011;35:1305–1310.
Weissferdt A, Hernandez JC, Kalhor N et al. Spindle cell thymomas: an immunohistochemical study of 30 cases. Appl Immunohistochem Mol Morphol 2011;19:329–335.
Yan B, Seng SC, Petersson F. . Thymoma with nuclear expression of thyroid transcription factor-1: a potential diagnostic pitfall on core biopsy. Appl Immunohistochem Mol Morphol 2011;19:76–81.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Weissferdt, A., Tang, X., Wistuba, I. et al. Comparative immunohistochemical analysis of pulmonary and thymic neuroendocrine carcinomas using PAX8 and TTF-1. Mod Pathol 26, 1554–1560 (2013). https://doi.org/10.1038/modpathol.2013.111
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.2013.111
Keywords
This article is cited by
-
Neuroendocrine Neoplasms Associated with Germline Pathogenic Variants in the Homologous Recombination Pathway
Endocrine Pathology (2019)
-
Immunohistochemical Biomarkers of Gastrointestinal, Pancreatic, Pulmonary, and Thymic Neuroendocrine Neoplasms
Endocrine Pathology (2018)
-
Thymic large cell neuroendocrine carcinoma – a rare and aggressive tumor: a case report
Journal of Medical Case Reports (2017)
-
Pathology of Neuroendocrine Tumours of the Female Genital Tract
Current Oncology Reports (2017)