Abstract
Objective:
The objective of this study was to determine the effect of incrementally higher doses of iron on the zinc protoporphyrin to heme ratio (ZnPP/H) and serum ferritin, and developmental outcomes in premature infants at risk for iron deficiency.
Study Design:
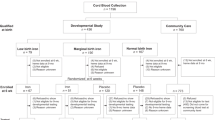
Infants eligible for this prospective, randomized blinded trial were between 27 and 30 completed weeks of gestation, older than 1 week of age and tolerating 100 ml kg−1 per day of enteral feedings. The control group was treated with 2.2 mg kg−1 per day of ferrous sulfate and the treatment group was treated with 3 to 12 mg kg−1 per day based on the ZnPP/H. Infants had follow-up with Bayley exams at 6 and 24 months corrected age. Statistical evaluation included Student’s t-tests and Fisher’s exact test.
Result:
Eighty-one infants were enrolled (40 control, 41 treatment). The average total iron dose for the control group was 2.2 mg kg−1 per day and for the treatment group was 10.4 mg kg−1 per day (P<0.05). The ZnPP/H was not different between the two groups. The ferritin at the end of the study was decreased in the control group but remained stable in the treatment group (control initial 202±109 ng ml−1, final 168±141 ng ml−1 (P<0.05); treatment initial 187±131 ng ml−1, final 176±118 ng ml−1). At 24 months, infants with psychomotor development index <85 occurred in 25% of the subjects in the control group and in 7% of subjects in the treatment group in a post hoc analysis (odds ratio, 4.2; 95% confidence interval, 0.7 to 43, P=0.07).
Conclusion:
The ZnPP/H may not be a reliable marker of iron status when used in a short period of time during iron supplementation. Infants treated with a lower dose of ferrous sulfate had a decreasing serum ferritin and a trend toward increased motor delays at 24 months.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Rao R, Georgieff MK . Neonatal iron nutrition. Semin Neonatol 2001; 6 (5): 425–435.
Rao R, Georgieff M . Perinatal aspects of iron metabolism. Acta Paediatr Suppl 2002; 91 (438): 124–129.
Beard JL, Connor JR . Iron status and neural functioning. Annu Rev Nutr 2003; 23: 41–58.
Lozoff B, Beard J, Connor J, Barbara F, Georgieff M, Schallert T . Long-lasting neural and behavioral effects of iron deficiency in infancy. Nutr Rev 2006; 64 (5 Pt 2): S34–S43.
Lozoff B, Brittenham GM, Wolf AW, McClish DK, Kuhnert PM, Jimenez E et al. Iron deficiency anemia and iron therapy effects on infant developmental test performance. Pediatrics 1987; 79 (6): 981–995.
Lozoff B . Iron deficiency and infant development. J Pediatr 1994; 125 (4): 577–578.
Lozoff B, Jimenez E, Hagen J, Mollen E, Wolf AW . Poorer behavioral and developmental outcome more than 10 years after treatment for iron deficiency in infancy. Pediatrics 2000; 105 (4): E51.
Georgieff M . Nutrition and the developing brain: nutrient priorities and measurement. Am J Clin Nutr 2007; 85 (2): 614S–620S.
Chen Q, Connor JR, Beard JL . Brain iron, transferrin and ferritin concentrations are altered in developing iron-deficient rats. J Nutr 1995; 125 (6): 1529–1535.
Beard J, Felt B, Schallert T, Burhans M, Connor J, Georgieff M . Moderate iron deficiency in infancy: biology and behavior in young rats. Behav Brain Res 2006; 170 (2): 224–232.
Petry CD, Eaton MA, Wobken JD, Mills MM, Johnson DE, Georgieff MK . Iron deficiency of liver, heart, and brain in newborn infants of diabetic mothers. J Pediatr 1992; 121 (1): 109–114.
Amin S, Orlando M, Eddins A, MacDonald M, Monczynski C, Wang H . In utero iron status and auditory neural maturation in premature infants as evaluated by auditory brainstem response. J Pediatr 2010; 156 (3): 377–381.
American Academy of Pediatrics. Committee on Nutrition. Iron fortification of infant formulas. Pediatrics 1999; 104 (1 Pt 1): 119–123.
Baker R, Greer F . NUTRITION TCO. Clinical report—diagnosis and prevention of iron deficiency and iron-deficiency anemia in infants and young children (0–3 years of age). Pediatrics 2010; 126 (5): 1040–1050.
Siimes MA, Järvenpää AL . Prevention of anemia and iron deficiency in very low-birth-weight infants. J Pediatr 1982; 101 (2): 277–280.
Domellöf M, Dewey KG, Lönnerdal B, Cohen RJ, Hernell O . The diagnostic criteria for iron deficiency in infants should be reevaluated. J Nutr 2002; 132 (12): 3680–3686.
Miller S, McPherson R, Juul S . Iron sulfate supplementation decreases zinc protoporphyrin to heme ratio in premature infants. J Pediatr 2006; 148 (1): 44–48.
Labbé R, Vreman H, Stevenson D . Zinc protoporphyrin: a metabolite with a mission. Clin Chem 1999; 45 (12): 2060–2072.
Labbé R, Dewanji A . Iron assessment tests: transferrin receptor vis-à-vis zinc protoporphyrin. Clin Biochem 2004; 37 (3): 165–174.
Labbé RF, Finch CA, Smith NJ, Doan RN, Sood SK, Madan N . Erythrocyte protoporphyrin/heme ratio in the assessment of iron status. Clin Chem 1979; 25 (1): 87–92.
Hastka J, Lasserre JJ, Schwarzbeck A, Reiter A, Hehlmann R . Laboratory tests of iron status: correlation or common sense? Clin Chem 1996; 42 (5): 718–724.
Juul S, Zerzan J, Strandjord T, Woodrum D . Zinc protoporphyrin/heme as an indicator of iron status in NICU patients. J Pediatr 2003; 142 (3): 273–278.
Oski F . Iron-fortified formulas and gastrointestinal symptoms in infants: a controlled study, With the cooperation of The Syracuse Consortium for Pediatric Clinical Studies. Pediatrics 1980; 66: 168–170.
Franz A, Mihatsch W, Sander S, Kron M, Pohlandt F . Prospective randomized trial of early versus late enteral iron supplementation in infants with a birth weight of less than 1301 grams. Pediatrics 2000; 106 (4): 700–706.
Winzerling JJ, Kling PJ . Iron-deficient erythropoiesis in premature infants measured by blood zinc protoporphyrin/heme. J Pediatr 2001; 139 (1): 134–136.
Cheng CF, Zerzan JC, Johnson DB, Juul SE . Zinc protoporphyrin-to-heme ratios in high-risk and preterm infants. J Pediatr 2012; 161 (1): 81–87.
Dallman PR, Siimes MA, Manies EC . Brain iron: persistent deficiency following short-term iron deprivation in the young rat. Br J Haematol 1975; 31 (2): 209–215.
Lozoff B, Jimenez E, Wolf AW . Long-term developmental outcome of infants with iron deficiency. N Engl J Med 1991; 325 (10): 687–694.
Lozoff B, Georgieff M . Iron deficiency and brain development. Semin Pediatr Neurol 2006; 13 (3): 158–165.
Stoltzfus RJ, Kvalsvig JD, Chwaya HM, Montresor A, Albonico M, Tielsch JM et al. Effects of iron supplementation and anthelmintic treatment on motor and language development of preschool children in Zanzibar: double blind, placebo controlled study. BMJ 2001; 323 (7326): 1389–1393.
Georgieff MK, Innis SM . Controversial nutrients that potentially affect preterm neurodevelopment: essential fatty acids and iron. Pediatr Res 2005; 57 (5 Pt 2): 99R–103R.
Acknowledgements
I acknowledge the contributions of Sharon Dowd and Diane D’Eugenio for their work, leading to the successful completion of this study. I also wish to thank the Crouse Hospital neonatal intensive care unit nursing staff and physicians for their support of this project. This study was funded by a grant from the Gerber Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no conflict of interest.
Rights and permissions
About this article
Cite this article
Miller, S. Iron supplementation in premature infants using the zinc protoporphyrin to heme ratio: short- and long-term outcomes. J Perinatol 33, 712–716 (2013). https://doi.org/10.1038/jp.2013.40
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jp.2013.40