Abstract
Objective:
The use of transcutaneous bilirubin (TcB) measurements has been studied extensively in the newborn population, but there have been few studies in outpatient populations and none from the offices of practicing pediatricians.
Study Design:
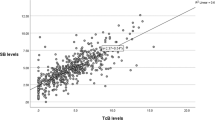
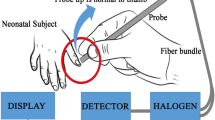
We performed TcB measurements on a mixed-race population of 120 jaundiced infants, ⩾35 weeks of gestation, in two hospital-based outpatient clinics, a regional public health nurse follow-up program and two pediatric office practices. Three individual TcB readings were obtained from the mid-sternum, and the average and maximum values were recorded.
Result:
There was good correlation between the TcB and total serum bilirubin (TSB) measurements (r=0.78, P=0.0). 59% of TSB's were ⩾15 mg dl−1 and, although the number of false-negative readings increased when the TSB values exceeded 15 mg dl−1, it was nevertheless possible to use TcB measurements to accurately predict the risk of TSB levels ⩾15 mg dl−1.
Conclusion:
In outpatient settings, a TcB measurement with the JM-103 provides a reliable screening method for the identification of hyperbilirubinemia even when the TSB level exceeds 15 mg dl−1. Using the maximum of three independent measurements reduces the number of false negatives, but increases the number of false positives. The use of TcB measurements in an outpatient practice should be a valuable tool for the practitioner.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Maisels MJ . Transcutaneous bilirubinometry. Neo Reviews 2006; 7: 217–225.
Engle WD, Jackson GL, Stehel EK, Sendelbach D, Manning MD . Evaluation of a transcutaneous jaundice meter following hospital discharge in term and near-term neonates. J Perinatol 2005; 25: 486–490.
Engle WD, Jackson GL, Sendelbach D, Manning D, Frawley W . Assessment of a transcutaneous device in the evaluation of neonatal hyperbilirubinemia in a primarily hispanic population. Pediatrics 2002; 110: 61–67.
Maisels MJ, Ostrea Jr E, Touch S, Clune S, Cepeda E, Kring E et al. Evaluation of a new transcutaneous bilirubinometer. Pediatrics 2004; 113: 1628–1635.
Ebbesen F, Rasmussen LM, Wimberley PD . A new transcutaneous bilirubinometer, BiliCheck, used in the neonatal intensive care unit and the maternity ward. Acta Paediatr 2002; 91: 203–211.
Rodriguez-Capote K, Kim K, Paes B, Turner D, Grey V . Clinical implication of the difference between transcutaneous bilirubinometry and total serum bilirubin for the classification of newborns at risk of hyperbilirubinemia. Clin Bio 2009; 42: 176–179.
Holland L, Blick K . Implementing and validating transcutaneous bilirubinometry for neonates. Am J Clin Pathol 2009; 132: 555–561.
Slusher TM, Angyo IA, Bode-Thomas F, Akor F, Pam SD, Adetunji AA et al. Transcutaneous bilirubin measurements and serum total bilirubin levels in indigenous African infants. Pediatrics 2004; 113: 1636–1641.
Kazmierczak SC, Robertson AF, Briley KP, Kreamer B, Gourley GR . Transcutaneous measurement of bilirubin in newborns: comparison with an automated Jendrassik-Grof procedure and HPLC. Clin Chem 2004; 50: 433–435.
Bhutani VK, Johnson L, Sivieri EM . Predictive ability of a predischarge hour-specific serum bilirubin for subsequent significant hyperbilirubinemia in healthy-term and near-term newborns. Pediatrics 1999; 103: 6–14.
Bhutani V, Gourley GR, Adler S, Kreamer B, Dalman C, Johnson LH . Noninvasive measurement of total serum bilirubin in a multiracial predischarge newborn population to assess the risk of severe hyperbilirubinemia. Pediatrics 2000; 106: e17.
Maisels MJ, Bhutani VK, Bogen D, Newman TB, Stark AR, Watchko JF . Hyperbilirubinemia in the newborn infant ⩾35 weeks’ gestation: An update with clarifications. Pediatrics 2009; 124 (4): 1193–1198.
Maisels MJ, Kring E . Transcutaneous bilirubin levels in the first 96 hours in a normal newborn population of 35 or more weeks’ of gestation. Pediatrics 2006; 117: 1169–1173.
Engle W, Lai S, Ahmad N, Manning MD, Jackson GL . An hour-specific nomogram for transcutaneous bilirubin values in term and late preterm hispanic neonates. Am J Perinatol 2009; 26 (6): 425–430.
Wainer S, Rabi Y, Parmar SM, Allegro D, Lyon M . Impact of skin tone on the performance of a transcutaneous jaundice meter. Acta Paediatrica 2009; 98: 1909–1915.
Fouzas S, Karatza AA, Skylogianni E, Mantagou L, Varvarigou A . Transcutaneous bilirubin levels in late preterm neonates. J Pediatr 2010; 157: 762–766.
Acknowledgements
Elizabeth Kring and Jennifer DeRidder assisted with data collection and analysis. Thomas Riggs, MD, provided statistical advice.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Dr Maisels has been a consultant for Draegar Medical Inc. and has received funding from Draeger Medical Inc. for previous studies of the JM-103.
Rights and permissions
About this article
Cite this article
Maisels, M., Engle, W., Wainer, S. et al. Transcutaneous bilirubin levels in an outpatient and office population. J Perinatol 31, 621–624 (2011). https://doi.org/10.1038/jp.2011.5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jp.2011.5
Keywords
This article is cited by
-
Validity of BiliDx as a point-of-care bilirubin measurement device to diagnose and monitor neonatal jaundice at Muhimbili National Hospital, an observational study
BMC Pediatrics (2024)
-
Performance of smartphone application to accurately quantify hyperbilirubinemia in neonates: a systematic review with meta-analysis
European Journal of Pediatrics (2023)
-
Transcutaneous bilirubin measurements: useful, but also reproducible?
Pediatric Research (2021)
-
Challenges for the estimation of uncertainty of measurements made in situ
Accreditation and Quality Assurance (2021)
-
Large scale validation of a new non-invasive and non-contact bilirubinometer in neonates with risk factors
Scientific Reports (2020)