Abstract
The augmentation index (AIx) is a measure of pulse wave reflection from peripheral muscular conduit arteries, and is assumed to increase with stiffened arteries. A white matter lesion (WML) is generally regarded as a chronic ischemic lesion, which is associated with cerebral small-vessel arteriosclerosis. The aim of this study is to elucidate the effect of pulse wave reflection on the brain. Carotid AIx was measured in a total of 205 neurologically normal adults. The correlations between AIx and WML were investigated. Two categories of WML were evaluated, periventricular hyperintensity (PVH) and deep and subcortical white matter hyperintensity (DSWMH). On univariate analysis, AIx was significantly correlated with the grades of PVH and DSWMH (P=0.0001, respectively). On multivariate analysis, AIx was correlated with PVH and DSWMH, independent of other risk factors such as age or blood pressure. Pulse wave reflection from small vessels may influence the genesis of WML.
Similar content being viewed by others
Introduction
White matter lesions (WMLs) have been reported to be a predictive factor in vascular dementia1, 2, 3, 4 and stroke,5, 6, 7 and are generally regarded as chronic ischemic lesions of the brain. Prior attempts to attribute their cause to general arteriosclerosis have been inconclusive. Some reports analyzed the presence of an association between WML and pulse wave velocity (PWV), which is the velocity at which a pulse wave travels between two arterial points and is considered to reflect an advanced stage of arteriosclerosis, but the results were controversial.8, 9, 10 Henskens emphasized that an important methodological limitation is the lack of information on carotid wave reflection, and it is difficult to interpret hemodynamic profile from the PWV alone, while the carotid augmentation index (AIx) should be determined as an index of the effect of arterial stiffening on cerebral blood flow.9 AIx is represented as the ratio of the late systolic component, which consists mainly of the pressure wave reflected back from the peripheral arteries, to the early systolic component in the pulse, and is thought to detect a degree of functional arterial stiffening in the distal muscular conduit arteries.11, 12, 13, 14 Thus, it is hypothesized that the AIx of the carotid artery includes information related to intracranial vascular changes. Based on this hypothesis, we investigated the association between WML and AIx in order to elucidate the effect of pulse wave reflection as an early marker of arterial stiffening on the brain.
Methods
Subjects
The recruited subjects consisted of 205 neurologically normal adults in whom brain dock examination was carried out, which is a health check for the status of the brain, in the affiliated hospital. They underwent brain MRI and measurement of AIx and PWV as one of a series of health examinations. None showed signs of stroke or brain tumor. Systolic blood pressure, diastolic blood pressure and body mass index were also measured. Fasting blood sugar, triglyceride and HDL-cholesterol were also examined in every case as part of a laboratory examination.
AIx and PWV measurement
AIx and PWV were measured with an automatic device, the Form PWV/ABI (Colin Medical Technology, Komaki, Japan). A tonometric sensor was placed on the left common carotid artery of the patient to detect carotid artery pulse. AIx was calculated automatically by dividing the difference between early- and late-stage systolic blood pressure by the pulse pressure. PWV was measured at the same time. After a 5-min rest, pulse waves were detected in the right upper arm and ankles simultaneously by an introducer, and the time difference between the upper arm waveform and ankle waveforms was measured with the same device. The PWV was calculated automatically as the distance between arm and ankle divided by the time difference.15 The validity and reproducibility of the AIx and PWV measurements by means of this automatic device have already been reported.16 Of the PWV values in the right and left ankles, the larger one was used for analyses as described in a previous report.17 AIx and PWV were measured on the same day when the MRI was examined.
White matter lesion
Two subcategories of WML were evaluated on MRI, periventricular hyperintensity (PVH) and deep and subcortical white matter hyperintensity (DSWMH). PVH and DSWMH were graded according to the classification of the PICA Study Group.18 PVH grade 0: absent or ‘rim’ only, grade I: localized lesions such as ‘caps’, grade II: PVH extended along the entire periventricular area, grade III: irregular PVH extended into deep white matter, grade IV: extending throughout deep and subcortical white matter. DSWMH grade 0: absent, grade 1: état criblé, grade 2: spotty or scattered lesions more than 3 mm in the subcortical and deep white matter showing hyperintensity on T2WI or Flair image and isointensity on T1WI, grade 3: fused, margin-unclear lesions in deep white matter showing hyperintensity on T2WI and isointensity on T1WI, grade 4: fused lesions widely extended to almost the whole of the white matter, showing hyperintensity on T2WI and isointensity on T1WI. The correlation between the grades of PVH or DSWMH and AIx was investigated by univariate and multivariate analyses. The correlation between those grades and PWV was investigated as well.
Statistical analyses
Statistical analyses were performed with SPSS version 17.0 computer software (SPSS, Chicago, IL, USA) with Mac OS 10.5.5. One-way analysis of variance was used to evaluate the correlation between grades of PVH or DSWMH and AIx or PWV. Multiple logistic regression analysis was used to determine the independent risk factors of PVH and DSWMH. First, AIx, age, systolic blood pressure, diastolic blood pressure, body mass index and laboratory data such as fasting blood sugar, HDL-cholesterol and triglyceride were selected as the independent variates for PVH and DSWMH. Next, instead of AIx, PWV was added as an independent variate, and multiple logistic regression analysis was performed similarly for PVH and DSWMH. Values of P<0.05 were considered significant.
Results
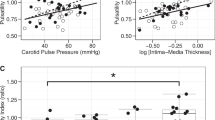
The characteristics of the subjects are summarized in Table 1. The total of 205 subjects included 110 males and 95 females, and mean age was 57 years (28–86). There were 148 people in grade 0, 49 in grade I, 7 in grade II and 1 in grade IV for PVH grading. For DSWMH grading, there were 87 people in grade 0, 86 in grade 1, 26 in grade 2 and 6 in grade 3. On one-way analysis of variance, AIx was significantly correlated with the grades of PVH and DSWMH (P<0.0001, respectively) (Figure 1). PWV was also significantly correlated with the grades of PVH and DSWMH (P<0.0001 and 0.002, respectively) (Figure 2).
On multiple logistic regression analysis, AIx was the independent risk factor for PVH (odds ratio 1.049 (95% confidence interval (CI): 1.014–1.080), P=0.047) and DSWMH (odds ratio 1.042 (95% CI: 1.015–1.069), P<0.0001), and age was also the independent risk factor for PVH (odds ratio 1.110 (95% CI: 1.066–1.155), P<0.0001) and DSWMH (odds ratio 1.068 (95% CI: 1.032–1.106), P=0.001) (Table 2). When PWV was added as the independent variate instead of AIx, it was not found to be an independent risk factor for both PVH and DSWMH, but age was found to be an independent risk factor for PVH (odds ratio 1.130 (95% CI: 1.083–1.178), P<0.0001) and DSWMH (odds ratio 1.074 (95% CI: 1.038–1.110), P<0.0001). For DSWMH, systolic blood pressure was also the independent risk factor (odds ratio 1.029 (95% CI: 1.008–1.050), P=0.021) (Table 3).
Discussion
Although there have been past reports that have attempted to connect WML to PWV, they obtained different results. Ohmine et al.8 investigated the PWV of 132 elderly people, with the result that PWV values were significantly greater in subjects with PVH than in those without. In contrast, there was no significant association between PWV and PVH in 196 elderly people with hypertension after adjustment for age, according to Kim et al.10 The antipodal results may reflect the differences in the backgrounds of the study subjects. Although both subject groups were older Asian people, one consisted of normal people while the other consisted of hypertensive patients.
On the one hand, Henskens et al.9 had results showing that a higher PWV was significantly associated with a greater volume of WMLs in 167 hypertensive patients in Netherlands. In their report they emphasized that an important methodological limitation is the lack of information on carotid wave reflection, and it is difficult to interpret hemodynamic profile from the PWV alone, suggesting that the carotid AIx should be used as an index of the effect of arterial stiffening on cerebral blood flow. This was the reason why AIx was requested as a marker of arteriosclerosis in this study. AIx is considered to be a marker of arterial stiffness because reflected waves reach the aorta earlier, and the ratio of the reflected wave to the systolic pressure wave increases in patients with arterial stiffness,19 resulting in different characteristics from PWV at two points. First of all, AIx is thought to detect vascular endothelial dysfunction, an early stage of arteriosclerosis, which causes reduced nitric oxide bioavailability and results in arterial stiffness,12, 13 and may be useful in evaluating the risk of vascular events earlier than PWV. Second, AIx is considered to reflect arterial stiffness in distal muscular conduit arteries or resistant arterioles, while PWV mainly reflects the arterial stiffness of central elastic arteries such as the aorta.20
The discrepancy between AIx and PWV in association with WML in the present study seems to be explained by their differing characteristics. One explanation is that PWV may have only weak detective power for WML because PWV reflects only aortic stiffness in parallel with cerebral arteriosclerosis. Owing to the fact that WMLs are thought to represent chronic ischemia in the terminal zones of the perforating or medullary artery and are caused by arteriosclerosis of these small vessels, there is a possibility that AIx reflects these arteriosclerotic changes when it is detected in the carotid artery.
Another explanation is related to the stage of arteriosclerosis. WMLs are thought to be caused by vascular endothelial dysfunction,21, 22, 23, 24, 25, 26 which is detected by AIx.12, 13 The difference that underlies people with WML vs. patients with the onset of stroke seems to be the severity of cerebral arteriosclerosis as the cause of these disorders. WMLs were thought to represent pre-clinical stroke by many previous authors. According to Kobayashi et al.,27 the presence of these lesions increased the risk of stroke onset significantly in their prospective study. Similarly, Vermeer et al.5 reported that individuals with severe WML had an increased risk of stroke. There is a phased progression of cerebral arteriosclerosis before the onset of stroke. AIx is useful in detecting arteriosclerosis up to a mild degree, corresponding to WML. Stroke is the breakthrough event at a critical level of cerebral arteriosclerosis, which corresponds to an increase in PWV.
The mechanism and pathophysiological aspects of PVH and DSWMH have not yet been identified sufficiently. PVH appears surrounding cerebral ventricles, and is very similar radiologically to periventricular lucency, which represents diffused cerebrospinal fluid throughout the subependymal layer in patients with hydrocephalus. Delano-Wood et al.4 reported that PVH may be derived from demyelination of the subependymal layer and diffusion of cerebrospinal fluid because of aging. On the other hand, DSWMH are thought to represent chronic ischemia in the terminal zones of the perforating or medullary artery and are caused by arteriosclerosis of these small vessels,6, 28, 29 and there is a possibility that AIx reflects these arteriosclerotic changes. But we must await future studies to elucidate the strict pathological differences between them.
Clinically, however, we can prevent the progression of cerebral arteriosclerosis in its early stage by measuring AIx. Identifying high AIx serves to provide an initial education to the patients about the disease, by awakening them to the risk. Quantitative expression of arteriosclerosis shows chronological changes, thus widening their awareness of the risk. In this way, monitoring AIx effectively generates motivation in patients to try to improve their condition.
In conclusion, the association between AIx and WML may suggest that early cerebral arteriosclerosis is related to the genesis of WML. To prevent breakthrough, detection of arteriosclerosis in the early stage with AIx may be useful for risk stratification in the management of WML.
References
Smith EE, Egorova S, Blacker D, Killiany RJ, Muzikansky A, Dickerson BC, Tanzi RE, Albert MS, Greenberg SM, Guttmann CRG . Magnetic resonance imaging white matter hyperintensities and brain volume in the prediction of mild cognitive impairment and dementia. Arch Neurol 2008; 65: 94–100.
Price TR, Manolio TA, Kronmal RA, Kittner SJ, Yue NC, Robbins J, Culver HA, O'Leary DH . Silent brain infarction on magnetic resonance imaging and neurological abnormalities in community-dwelling older adults. The cardiovascular health study. Stroke 1997; 28: 1158–1164.
Prins ND, Dijk EJ, Heijer T, Vermeer SE, Koudstaal PJ, Oudkerk M, Hofman A, Breteler MMB . Cerebral white matter lesions and the risk of dementia. Arch Neurol 2004; 61: 1531–1534.
Delano-Wood L, Abeles N, Sacco JM, Wierenga CE, Horne NR, Bozoki A . Regional white matter pathology in mild cognitive impairment. Differential influence of lesion type on neuropsychological functioning. Stroke 2008; 39: 794–799.
Vermeer SE, Hollander M, Dijk EJ, Hofman A, Koudstaal PJ, Breteler MMB . Silent brain infarcts and white matter lesions increase stroke risk in the general population: the Rotterdam scan study. Stroke 2003; 34: 1126–1129.
Thomas AJ, O'Brien JT, Davis S, Ballard C, Barber R, Kalaria RN, Perry RH . Ischemic basis for deep white matter hyperintensities in major depression. Arch Gen Psychiatry 2002; 59: 785–792.
Bokura H, Kobayashi S, Yamaguchi S, Iijima K, Nagai A, Toyoda G, Oguro H, Takahashi K . Silent brain infarction and subcortical white matter lesions increase the risk of stroke and mortality: a prospective cohort study. J Stroke Cerebrovasc Dis 2006; 15: 57–63.
Ohmine T, Miwa Y, Yao H, Yuzuhara T, Takashima Y, Uchino A, Takahashi-Yanaga F, Morimoto S, Maehara Y, Sasaguri T . Association between arterial stiffness and cerebral white matter lesions in community-dwelling elderly subjects. Hypertens Res 2008; 31: 75–81.
Henskens LHG, Kroon AA, Oostenbrugge RJ, Gronenschild EHB, Fuss-Lejeune MMJJ, Hofman PAM, Lodder J, Leeuw PW . Increased aortic pulse wave velocity is associated with silent cerebral small-vessel disease in hypertensive patients. Hypertension 2008; 52: 1120–1126.
Kim DH, Kim J, Kim JM, Lee AY . Increased brachial-ankle pulse wave velocity is independently associated with risk of cerebral ischemic small vessel disease in elderly hypertensive patients. Clin Neurol Neurosurg 2008; 110: 599–604.
Murgo JP, Westerhof N, Giolma JP, Altobelli SA . Aortic input impedance in normal man: relationship to pressure wave forms. Circulation 1980; 62: 105–116.
Wilkinson IB, Cockcroft JR . Cholesterol, endothelial function and cardiovascular disease. Curr Opin Lipidol 1995; 9: 237–242.
Wilkinson IB, Parasad K, Hall IR, Thomas A, Maccallum H, Webb DJ, Frenneaux MP, Cockcroft JR . Increased central pulse pressure and augmentation index in subjects with hypercholesterolemia. J Am Cardiol 2002; 39: 1005–1011.
Bortolotto LA, Blacher J, Kondo T, Takazawa K, Safar ME . Assessment of vascular aging and atherosclerosis in hypertensive subjects: second derivative of photoplethysmogram versus pulse wave velocity. Am J Hypertens 2000; 13: 165–171.
Yamashina A, Tomiyama H, Arai T, Hirose K, Koji Y, Hirayama Y, Yamamoto Y, Hori S . Brachial-ankle pulse wave velocity as a marker of atherosclerotic vascular damage and cardiovascular risk. Hypertens Res 2003; 26: 615–622.
Yamashina A, Tomiyama H, Takeda K, Tsuda H, Arai T, Hirose K, Koji Y, Hori S, Yamamoto Y . Validity, reproducibility, and clinical significance of noninvasive brachial-ankle pulse wave velocity measurement. Hypertens Res 2002; 25: 359–364.
Cortez-Cooper MY, Supak JA, Tanaka H . A new device for automatic measurements of arterial stiffness and ankle-brachial index. Am J Cardiol 2003; 91: 1519–1522.
Shinohara Y, Tohgi H, Hirai S, Terashi A, Fukuuchi Y, Yamaguchi T, Okudera T . Effect of the Ca antagonist nivaldipine on stroke occurrence or recurrence and extension of asymptomatic cerebral infarction in hypertensive patients with or without history of stroke (PICA Study). Cerebrovasc Dis 2007; 24: 202–209.
Saba PS, Roman MJ, Ganau A, Pini R, Jones EC, Pickering TG, Devereux RB . Relationship of effective arterial elastance to demographic and arterial characteristics in normotensive and hypertensive adults. J Hypertens 1995; 13: 971–977.
Asmar RG, London GM, O'Rourke ME, Safar ME . Improvement in blood pressure, arterial stiffness and wave reflections with a very-low-dose perindopril/indapamide combination in hypertensive patient. A combination with atenolol. Hypertension 2001; 38: 922–926.
Young VG, Halliday GM, Kril JK . Neuropathologic correlates of white matter hyperintensities. Neurology 2008; 71: 804–811.
Hoth KF, Tate DF, Poppas A, Forman DE, Gunstad J, Moser DJ, Paul RH, Jefferson AL, Haley AP, Cohen RA . Endothelial function and white matter hyperintensities in older adults with cardiovascular disease. Stroke 2007; 38: 308–312.
Markus HS, Hunt B, Palmer K, Enzinger C, Schmidt H, Schmidt R . Markers of endothelial and hemostatic activation and progression of cerebral white matter hperintensities. Longitudinal results of the Austrian stroke prevention study. Stroke 2005; 36: 1410–1414.
Kahn U, Hassan A, Vallance P, Markus HS . Asymmetric dimethylarginine in cerebral small vessel disease. Stroke 2007; 38: 411–413.
Knottnerus ILH, Cate HT, Lodder J, Kessels F, van Oostenbrugge RJ . Endothelial dysfunction in lacunar stroke: a systemic review. Cerebrovasc Dis 2009; 27: 519–526.
Markus HS . Genes, endothelial dysfunction and cerebral small vessel disease in man. Exp Physiol 2007; 93: 121–127.
Kobayashi S, Okada K, Koide H, Bokura H, Yamaguchi S . Subcortical silent brain infarction as a risk factor for clinical stroke. Stroke 1997; 28: 1932–1939.
Simpson JE, Hosny O, Wharton SB, Heath PR, Holden H, Fernando MS, Matthews F, Forster G, O'Brien JT, Barber R, Kalaria RN, Brayne C, Shaw PJ, Lewis CE, Ince PG . Microarray RNA expression analysis of cerebral white matter lesions reveals changes in multiple functional pathways. Stroke 2009; 40: 369–375.
Fernando MS, Simpson JE, Matthews F, Brayne C, Lewis CE, Barber R, Kalaria RN, Forster G, Esteves F, Wharton SB, Shaw PJ, O'Brien JT, Ince PG . White matter lesions in an unselected cohort of the elderly. Molecular pathology suggests origin from chronic hypoperfusion injury. Stroke 2006; 37: 1391–1398.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nakano, T., Munakata, A., Shimaura, N. et al. Augmentation index is related to white matter lesions. Hypertens Res 35, 729–732 (2012). https://doi.org/10.1038/hr.2012.24
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/hr.2012.24
Keywords
This article is cited by
-
NT-proBNP, cerebral small vessel disease and cardiac function in patients with a recent lacunar infarct
Journal of Human Hypertension (2023)
-
Brain arteriolosclerosis
Acta Neuropathologica (2021)
-
Response to Rai
Hypertension Research (2013)