Abstract
Obesity is associated with early cardiovascular dysfunction and reduced muscle strength. Whole-body vibration (WBV) training may improve arterial function and muscle strength. The effects of WBV training on arterial stiffness (brachial-ankle pulse wave velocity, baPWV), wave reflection (augmentation index, AIx), brachial systolic blood pressure (bSBP), aortic systolic blood pressure (aSBP), heart rate variability, and muscle strength (one-repetition maximum, 1RM) were examined in 10 young (21±2 year) overweight/obese women (body mass index, BMI=29.9±0.8 kg m–2). Participants were randomized to a 6-week WBV training or non-exercising control (CON) period in a crossover design. WBV training (3 days × week) consisted of static and dynamic squats and calf raises with vibration intensity at 25–30 Hz and 1–2 mm amplitude (2.83–4.86 G). There were significant (P<0.05) decreases in baPWV (−0.9±0.3 m s–1), AIx (−8.0±2.2 %), bSBP (−5.3±1.5 mm Hg), aSBP (−5.2±2.1 mm Hg), low-frequency power (−0.13±0.05 nu) and sympathovagal balance (LF/HF, −0.42±0.16) after WBV training compared with CON. Significant (P<0.05) increases in high-frequency power (HF, 0.19±0.04 nu) and leg extension 1RM (8.2±2.3 kg) occurred after WBV training compared with CON. Six weeks of WBV training decreased systemic arterial stiffness and aSBP via improvements in wave reflection and sympathovagal balance in young overweight/obese normotensive women. WBV training may benefit arterial function and muscle strength in deconditioned individuals who cannot perform conventional exercise.
Similar content being viewed by others
Introduction
Vascular damage can be identified as increased arterial stiffness (pulse wave velocity, PWV), wave reflection (augmentation index, AIx), and aortic systolic blood pressure (aSBP) in young obese adults with normal brachial SBP (bSBP),1, 2, 3 suggesting that PWV, AIx and aSBP are more sensitive markers of early arterial dysfunction than bSBP. Sympathetic overactivity in obese normotensive women4 may contribute to the increase in PWV5 and AIx6 by increasing the vascular tone of small arteries. The predominance of cardiac sympathetic over parasympathetic activity (increased sympathovagal balance) is associated with increased brachial-ankle PWV (baPWV),7 a marker of systemic arterial stiffness that is highly correlated with aortic PWV.8
The baPWV has been inversely associated with muscle mass in adults with metabolic syndrome.9 Moreover, muscle strength has been inversely associated with aortic PWV10 and the prevalence of obesity.11 This evidence suggests that improvement of muscle function can occur concurrently with improved endothelial function in obese women.12, 13 It is well recognized that high-intensity resistance exercise training (RET) is an effective intervention for improving muscle strength and mass in overweight/obese women.13, 14 However, RET may increase central PWV and AIx in young women.15 Alternatively, whole-body vibration (WBV) training consisting of isometric and dynamic leg exercise has shown to effectively increase muscle strength to levels comparable to RET in young women.16, 17 Moreover, muscle strength and muscle mass gains have been reported to occur after only 618 and 1019 weeks, respectively, of WBV training in older women. Thus, muscular adaptations may be apparent after a 6-week WBV training in young women. To date, only acute arterial responses to WBV exercise have been examined. It has been reported that baPWV20 and AIx21 were decreased within 30 to 40 min after a session of static squat with WBV in young healthy men. Recent evidence suggests that acute WBV attenuates the increases in baPWV, wave reflection and aSBP in response to sympathetic overactivity induced by post-exercise muscle ischemia.22 However, the effects of WBV training on arterial function and autonomic modulation have not been examined.
The purpose of this study was to evaluate the effectiveness of a 6-week WBV training program on arterial function, autonomic function, and muscle strength in young overweight/obese women. We hypothesized that WBV training would beneficially affect muscle strength, baPWV, AIx, aSBP and sympathovagal balance in overweight/obese women.
Methods
Subjects
A total of 10 young (18–35 years) overweight/obese (body mass index, BMI >25 kg m–2) and normotensive (<140/90 mm Hg) women volunteered for this study. Subjects were nonsmokers, sedentary (⩽90 min of regular exercise per week), free of apparent cardiovascular diseases as assessed by medical history, not pregnant and not taking oral contraceptives. Individuals were excluded if they had chronic diseases, joint prosthetic devices and recent thrombosis or wounds in the legs. The Florida State University Human Subject committee approved the protocol and all subjects gave written informed consent before their inclusion.
Design
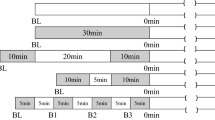
All subjects were examined during the early- to mid-follicular (days 1–10) and late-luteal phases (⩾day 19) of the menstrual cycle to avoid possible effects of endogenous estrogens on autonomic function and arterial function during the late-follicular and early-luteal phases.23, 24 After baseline measurements, subjects were randomly assigned to 6 weeks of WBV training or non-exercising control (CON) period, followed by crossover after a 4-week washout period (Figure 1). During the control and washout period, subjects were instructed to refrain from any mode of structured exercise training. Before and 6 weeks after WBV and CON periods, all subjects had cardiovascular function, muscle strength and body composition measured. Subjects were asked to refrain from caffeine and alcohol for 24 h before each visit. After WVB training, because arterial responses to acute WBV are normalized 40 min after exercise cessation,20 cardiovascular testing was performed 24 h after the last WBV session. All measurements were conducted at the same time of the day (8 AM to10 AM) after an overnight fast in a quiet temperature-controlled room (23±1 °C). After instrumentation, subjects rested in the supine position for at least 10 min before data collection. Subjects were advised not to change their regular lifestyles during the study.
Pulse-wave velocity and brachial blood pressure
The baPWV, bSBP and brachial diastolic blood pressure (bDBP) were measured using a PWV/ABI automatic device (VP-2000, Omron Healthcare Inc., Vernon Hills, IL, USA). ECG electrodes were placed on the forearms, whereas a heart sound microphone was placed on the left parasternal border of the second intercostal space. BP cuffs were wrapped around both arms (brachial artery) and ankles (posterior tibial artery). BP, ECG and pulse waveforms were simultaneously recorded during 10 s. The feet of the pulse waveforms were related to the R-wave of the ECG to calculate transit time between the brachium and ankle (ΔTba). The path distances from the suprasternal notch to the brachial (Db) and to the ankle (Da) sensor were calculated automatically by the device based on the subject's height.8 BaPWV was calculated as Da–Db/ΔTba. The average of the left and right baPWV was used in the analysis. The validity and reproducibility of noninvasive baPWV has been previously shown.8 In our laboratory, the interclass correlation coefficient for baPWV calculated on two separate days is 0.95.
Pulse wave analysis
The bSBP and bDBP were used to calibrate radial waveforms, which were obtained from a 10 s epoch using a high-fidelity tonometer (SPT-301B; Millar Instruments, Houston, TX, USA). Aortic pressure waveforms were derived using a validated generalized transfer function (SphygmoCor, AtCor Medical, Sydney, NSW, Australia).25 The aortic pressure wave is composed of a forward wave, caused by stroke volume ejection, and a reflected wave that returns to the aorta from peripheral sites.26 Augmentation pressure is the difference between the second and first systolic peaks. The AIx was defined as the augmentation pressure expressed as a percentage of the aortic pulse pressure. AIx normalized for a HR of 75 beats per min (AIx@75) was also calculated. Transit time of the reflected wave (Tr) indicates the round-trip travel of the forward wave to the peripheral reflecting sites and back to the aorta.26 The average of two measurements of aortic hemodynamic with high-quality (operator index ⩾80%) was used in the analysis. In our laboratory, the intraclass correlation coefficients for resting aSBP, AIx and Tr taken on two separate days are 0.97, 0.95 and 0.97, respectively.
Heart rate variability
Five-min ECG recordings from a modified CM5 lead were collected at a sampling rate of 1000 Hz using a Biopac data acquizition system (Biopac Systems Inc., Goleta, CA, USA). Following visual inspection of noise and ectopic beats, power spectral analysis of the R-R intervals was performed by the autoregressive model using the WinCPRS software (Absolute Aliens Oy, Turku, Finland). Total power (TP), low-frequency power (LF, 0.04–0.15 Hz), and high-frequency power (HF, 0.15–0.4 Hz) were calculated. The LF/HF ratio reflects the complex interplay between sympathetic and parasympathetic modulation (sympathovagal balance). Increased sympathovagal balance was considered to reflect predominant sympathetic activity. Normalized units (nu) were assessed by dividing the power of HF or LF by (LF+HF). The LFnu power is an indicator of sympathetic modulation, whereas the HF power either in ms2 (absolute units) or nu reflects cardiovagal modulation.27
Muscle strength
Quadriceps muscle strength was measured by the one-repetition maximum (1RM) test using variable resistance equipment for the knee extension exercise (MedX Corp., Ocala, FL, USA). The highest weight moved one time through the full range of motion using a good form was considered the 1RM.
Anthropometry and body composition
Height was measured to the nearest 0.5 cm using a stadiometer and body weight was measured to the nearest 0.1 kg using a seca scale (Sunbeam Products Inc., Boca Raton, FL, USA). BMI was calculated as kg m–2. Total fat mass and bilateral arm and leg lean soft tissue (LST) mass were determined from whole body dual-energy x-ray absorptiometry scans (GE Lunar DPX-IQ, Madison, WI, USA).
WBV training
All subjects underwent three supervised training sessions per week separated by at least 48 h for 6 weeks. The WBV training included leg exercises standing on a WBV platform (PowerPlate, Irvine, CA, USA). Exercises consisted of dynamic and static semi-squats with a 120° knee angle (considering 180° as full knee extension), wide-stand semi-squat and calf-raises. The dynamic exercises were performed with slow movements at a rate of 2 and 3 s for concentric and eccentric phases, respectively. The vibration intensity was progressed by increasing the frequency (25–30 Hz) and amplitude (1–2 mm). The duration of the sets and rest periods was progressively increased (30–60 s) and decreased (60–30 s), respectively. During the last 2 weeks, subjects used a weight vest with 5% and 10% of their body weight during week 5 and 6, respectively.
Statistical analyses
Data distribution was confirmed using the Shapiro–Wilk test. All statistical analyses were performed with SPSS version 17.0 (SPSS Inc., Chicago, IL, USA). Possible differences between WBV and CON in baseline variables were analyzed using Student's t-test. Changes in all variables were analyzed by a 2 × 2 ((period: WBV vs. CON) × time (0 vs. 6 week)) ANOVA with repeated measures. When a significant group-by-time interaction was observed, a paired t-test was used for post hoc comparisons. A Pearson's correlation was used to find the relationship between changes in two variables. Values are shown as mean±s.e. and statistical significance was set at P<0.05. A previous study was used to estimate that 10 subjects would enable 80% power to detect an 8% decrease in baPWV.28
Results
Subject characteristics
There were no significant differences in all measurements between CON and WBV training at baseline. Age, height and BMI were 22.4±1.8 years, 1.62±0.02 m, and 29.9±0.8 kg m–2, respectively. Table 1 shows body mass, LST mass and 1RM before and after 6 weeks of CON and WBV training. There was a significant increase in leg LST mass (0.36±0.11 kg, P<0.05) after WBV training, which was not significantly different than CON. A significant treatment-by-time interaction was detected for leg 1RM (P<0.01), such that it increased (8.2±2.3 kg, P<0.05) after WBV training, but not after CON. Body weight, total fat mass, and arm LST mass did not change after CON and WBV training.
Arterial function
There were significant treatment-by-time interactions for aSBP (P<0.05, Table 2), bSBP (P<0.01, Table 2), AIx@75 (P<0.01, Table 2), baPWV (P<0.05, Figure 2a) and AIx (P<0.05, Figure 2b). There was a significant decrease in aSBP (−5.2±2.1 mm Hg, P<0.01), bSBP (−5.3±1.5 mm Hg, P<0.01), AIx@75 (−10.7±2.4%, P<0.01), baPWV (−0.9±0.3 m s–1, P<0.05) and AIx (−8.0±2.2%, P<0.05) after WBV training, but not after CON.
Cardiac autonomic function
There were significant treatment-by-time interactions for TP (P<0.05), LF (P<0.05) and HR (P<0.01) (Table 2), and for HFnu, LFnu and LFnu/HFnu (P<0.05, Figure 3). LF power (−376±90 ms2, P<0.01), HR (−6±1, P<0.05), LFnu (−0.13±0.05, P<0.05) and LFnu/HFnu (−0.42±0.16, P<0.05) were significantly decreased, whereas TP (825±258 ms2, P<0.01) and HFnu (0.19±0.04, P<0.05) were significantly increased after WBV training with no changes after CON.
Correlations
Sympathovagal balance and baPWV at baseline (r=0.67, P<0.05) and their changes (r=0.72, P<0.05) after WBV training were positively correlated. There was no significant association between changes in LF/HF with changes in baPWV and AIx. No significant correlation was found between changes in baPWV with changes in leg LST and strength.
Discussion
The novelty of this study is that 6 weeks of WBV training improved systemic arterial stiffness, aortic hemodynamics, heart rate variability and muscle strength in young overweight/obese women. To the best of our knowledge, this is the first report of the effects of WBV training on arterial function and cardiac autonomic control.
The primary finding of the present study was that baPWV and AIx, indicators of systemic arterial stiffness and wave reflection, were significantly decreased after 6 weeks of WBV training. Currently, only acute arterial responses to WBV exercise have been examined in young healthy men. It has been shown that a 10-min session of intermittent static squat with WBV results in acute decreases in baPWV,20 leg PWV and AIx,21 which persist for 30 to 40 min after exercise cessation. Interestingly, a continuous 10-min session of vibration on the legs without exercise also decreases baPWV, leg PWV and AIx that is maintained during 15 to 30 min post vibration.29, 30 These previous findings suggest that active and passive vibration acutely decrease baPWV and AIx through local arterial effects independently of aortic stiffness as shown by no changes in aortic PWV and Tr. The present data showed no change in Tr, suggesting that the reduction in AIx was not influenced by aortic stiffness. Although AIx and HR are inversely related, a small decrease in HR (8 beats per min) occurs with a decrease in AIx after vasodilating drug therapy.31 The observed 5 beats per min decrease following WBV training did not influence the changes in AIx and AIx@75. Thus, it is likely that repetition of the acute vascular effects during 6 weeks would be responsible for the improvements in baPWV and AIx after WBV training.
We found that WBV training decreased bSBP (−5.3 mm Hg) and aSBP (−5.2 mm Hg). This hypotensive effect may be due in part to the static component of the WBV protocol. Similar to our findings, decreases in bSBP have been reported after 4 to 8 weeks of static training using leg extension (−4.9 mm Hg) and handgrip (−10 mm Hg) in normotensive young men32 and older adults,33 suggesting that the magnitude of the hypotensive effect of static exercise training may be inversely related to the initial level of bSBP. Although the effect of static training on aSBP is unknown, Taaffe et al.34 showed a significant decrease in aSBP (−6 mm Hg) in older adults after 20 weeks of RET, which has an important static component specially when performed with slow movement.35 We previously reported acute decreases in AIx following a session of either leg resistance exercise with slow movement36 or static squat with WBV.21 As Tr was not affected by WBV training, the decrease in AIx was attributed to a reduction in the magnitude of the reflected wave, which corresponds with aSBP in individuals with increased AIx.37 Because our participants were normotensives, the improved arterial function may have influenced the decrease in aSBP rather than the reduction in aSBP caused a passive decrease in baPWV.
The possible mechanisms underlying the effects of WBV training on arterial function and SBP are the improvement of endothelial and autonomic functions. WBV may acutely increase local nitric oxide production.38 Nitrate administration decreases SBP and AIx via a reduction in vascular tone of small arteries independently from aortic PWV.39 In a previous study, we showed that acute WBV attenuated the increases in reflected wave magnitude, aSBP, leg PWV and baPWV during metaboreflex-induced sympathetic activation following a continuous 4 min session of static squat, suggesting that vibration-related factors induced functional sympatholysis in the legs.22 In the current study, WBV training decreased the sympathovagal balance (LFnu/HFnu) due to a concurrent decrease in sympathetic (LFnu) and increase in cardiovagal (HFnu) modulation. Our data showed a positive relationship between the sympathovagal balance and baPWV at baseline in young obese women, as previously reported in young men.7 After WBV training, the decreases in sympathovagal balance and baPWV were strongly correlated. A decrease in sympathovagal balance may reduce the cardiovascular risk in normotensive obese women.4 Saul et al.40 reported that LFnu power of heart rate variability is associated with muscle sympathetic nerve activity. Recent work has shown that the association between sympathetic activity and AIx is positive in men and negative in women.6 This sex difference in the sympathetic control of wave reflection6 may explain the lack of relationship between changes in sympathovagal balance and AIx in obese women after WBV training. Our data suggest that the improvement in baPWV would be partially attributed to a decrease in sympathetic dominance after WBV training.
Leg muscle strength was increased by 6.3% after WBV training. Consistent with the present study, muscle strength improvements (7.2–9.0%) have been reported after 12 to 24 weeks of static and dynamic exercises with WBV in young women.16, 17 Our findings are in agreement with a 2.2% increase in body fat-free mass reported in young women after 24 weeks of unloaded static and dynamic exercises with WBV.17 Because the increase in LST mass after 6 weeks of WBV training was no different than the CON period in our study, muscle strength gains would be primarily attributed to neural adaptations rather than muscle hypertrophy.16, 17, 19 Indeed, increased muscle strength per se may offer protection from obesity.11 In addition, Bogaerts et al.41 showed improvements in muscle strength and oxygen consumption after WBV training that included static and dynamic exercises that were comparable to a fitness group that combined RET and aerobic training, suggesting that WBV training is an efficient alternative for overall training effects. Because young obese individuals may develop high SBP, metabolic syndrome and sarcopenic obesity, which are associated with increased baPWV,9, 42 WBV training may be a beneficial exercise modality for the prevention of these comorbidities through improvements in arterial function and muscle strength.
There are several limitations of the present study. Because only young overweight/obese otherwise healthy women were evaluated, our findings may not be generalizable to overweight/obese men or individuals of different ages and with different health conditions. It might be argued that a limitation of this study is the lack of a parallel non-exercising control group. Because it could be ethically questionable not to provide WBV training to obese women, we used a cross-over design. Moreover, exercise without WBV does not induce acute decreases in hemodynamics and PWV;20, 21 thus, a non-WBV exercise period or group was not included. It is possible that a longer WBV training may be required to show significant correlations between changes in muscle strength and arterial function. Future research is needed to examine the effects of WBV training on arterial function in these populations.
In conclusion, 6 weeks of WBV training was effective at decreasing baPWV, AIx and aSBP through improvements in wave reflection magnitude and autonomic regulation in young overweight/obese normotensive women.
References
Wykretowicz A, Adamska K, Guzik P, Krauze T, Wysocki H . Indices of vascular stiffness and wave reflection in relation to body mass index or body fat in healthy subjects. Clin Exp Pharmacol Physiol 2007; 34: 1005–1009.
Kolade OO, O’Moore-Sullivan TM, Stowasser M, Coombes JS, Fassett RG, Marwick TH, Sharman JE . Arterial stiffness, central blood pressure and body size in health and disease. Int J Obes 2012; 36: 93–99.
Wildman RP, Mackey RH, Bostom A, Thompson T, Sutton-Tyrrell K . Measures of obesity are associated with vascular stiffness in young and older adults. Hypertension 2003; 42: 468–473.
Esposito K, Marfella R, Gualdiero P, Carusone C, Pontillo A, Giugliano G, Nicoletti G, Giugliano D . Sympathovagal balance, nighttime blood pressure, and QT intervals in normotensive obese women. Obes Res 2003; 11: 653–659.
Swierblewska E, Hering D, Kara T, Kunicka K, Kruszewski P, Bieniaszewski L, Boutouyrie P, Somers VK, Narkiewicz K . An independent relationship between muscle sympathetic nerve activity and pulse wave velocity in normal humans. J Hypertens 2010; 28: 979–984.
Casey DP, Curry TB, Joyner MJ, Charkoudian N, Hart EC . Relationship between muscle sympathetic nerve activity and aortic wave reflection characteristics in young men and women. Hypertension 2011; 57: 421–427.
Nakao M, Nomura K, Karita K, Nishikitani M, Yano E . Relationship between brachial-ankle pulse wave velocity and heart rate variability in young Japanese men. Hypertens Res 2004; 27: 925–931.
Yamashina A, Tomiyama H, Takeda K, Tsuda H, Arai T, Hirose K, Koji Y, Hori S, Yamamoto Y . Validity, reproducibility, and clinical significance of noninvasive brachial-ankle pulse wave velocity measurement. Hypertens Res 2002; 25: 359–364.
Kim TN, Park MS, Lim KI, Yang SJ, Yoo HJ, Kang HJ, Song W, Seo JA, Kim SG, Kim NH, Baik SH, Choi DS, Choi KM . Skeletal muscle mass to visceral fat area ratio is associated with metabolic syndrome and arterial stiffness: The Korean Sarcopenic Obesity Study (KSOS). Diabetes Res Clin Pract 2011; 93: 285–291.
Fahs CA, Heffernan KS, Ranadive S, Jae SY, Fernhall B . Muscular strength is inversely associated with aortic stiffness in young men. Med Sci Sports Exerc 2010; 42: 1619–1624.
Jackson AW, Lee DC, Sui X, Morrow Jr JR, Church TS, Maslow AL, Blair SN . Muscular strength is inversely related to prevalence and incidence of obesity in adult men. Obesity 2010; 18: 1988–1995.
Kingsley JD, Figueroa A . Effects of resistance exercise training on resting and post-exercise forearm blood flow and wave reflection in overweight and obese women. J Hum Hypertens (e-pub ahead of print, 15 September 2011; doi: 10.1038/jhh.2011.82).
Olson TP, Dengel DR, Leon AS, Schmitz KH . Moderate resistance training and vascular health in overweight women. Med Sci Sports Exerc 2006; 38: 1558–1564.
Figueroa A, Going SB, Milliken LA, Blew RM, Sharp S, Teixeira PJ, Lohman TG . Effects of exercise training and hormone replacement therapy on lean and fat mass in postmenopausal women. J Gerontol A Biol Sci Med Sci 2003; 58: 266–270.
Cortez-Cooper MY, DeVan AE, Anton MM, Farrar RP, Beckwith KA, Todd JS, Tanaka H . Effects of high intensity resistance training on arterial stiffness and wave reflection in women. Am J Hypertens 2005; 18: 930–934.
Delecluse C, Roelants M, Verschueren S . Strength increase after whole-body vibration compared with resistance training. Med Sci Sports Exerc 2003; 35: 1033–1041.
Roelants M, Delecluse C, Goris M, Verschueren S . Effects of 24 weeks of whole body vibration training on body composition and muscle strength in untrained females. Int J Sports Med 2004; 25: 1–5.
Sanudo B, de Hoyo M, Carrasco L, McVeigh JG, Corral J, Cabeza R, Rodríguez C, Oliva A . The effect of 6-week exercise programme and whole body vibration on strength and quality of life in women with fibromyalgia: a randomised study. Clin Exp Rheumatol 2010; 28 (6 Suppl 63): S40–S45.
Machado A, Garcia-Lopez D, Gonzalez-Gallego J, Garatachea N . Whole-body vibration training increases muscle strength and mass in older women: a randomized-controlled trial. Scand J Med Sci Sports 2010; 20: 200–207.
Otsuki T, Takanami Y, Aoi W, Kawai Y, Ichikawa H, Yoshikawa T . Arterial stiffness acutely decreases after whole-body vibration in humans. Acta physiologica 2008; 194: 189–194.
Figueroa A, Vicil F, Sanchez-Gonzalez M . Acute exercise with whole-body vibration decreases wave reflection and leg arterial stiffness. Am J Cardiovasc Dis 2011; 1: 60–67.
Figueroa A, Gil R, Sanchez-Gonzalez MA . Whole-body vibration attenuates the increase in leg arterial stiffness and aortic systolic blood pressure during post-exercise muscle ischemia. Eur J Appl Physiol 2011; 111: 1261–1268.
Adkisson EJ, Casey DP, Beck DT, Gurovich AN, Martin JS, Braith RW . Central, peripheral and resistance arterial reactivity: fluctuates during the phases of the menstrual cycle. Exp Biol Med 2010; 235: 111–118.
Saeki Y, Atogami F, Takahashi K, Yoshizawa T . Reflex control of autonomic function induced by posture change during the menstrual cycle. J Auton Nerv Syst 1997; 66: 69–74.
O’Rourke MF, Pauca A, Jiang XJ . Pulse wave analysis. Br J Clin Pharmacol 2001; 51: 507–522.
Nichols WW, Singh BM . Augmentation index as a measure of peripheral vascular disease state. Curr Opin Cardiol 2002; 17: 543–551.
Malik M, Bigger JT, Camm AJ, Kleiger RE, Malliani A, Moss AJ et al. Heart rate variability: standards of measurement, physiological interpretation and clinical use. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Circulation 1996; 93: 1043–1065.
Okamoto T, Masuhara M, Ikuta K . Effects of low-intensity resistance training with slow lifting and lowering on vascular function. J Hum Hypertens 2008; 22: 509–511.
Sanchez-Gonzalez MA, Wong A, Vicil F, Gil R, Park SY, Figueroa A . Impact of passive vibration on pressure pulse wave characteristics. J Hum Hypertens (e-pub ahead of print, 23 June 2011; doi:10.1038/jhh.2011.63).
Wong A, Sanchez-Gonzalez M, Gil R, Vicil F, Park S, Figueroa A . Passive vibration on the legs reduces peripheral and systemic arterial stiffness. Hypertens Res 2012; 35: 126–127.
Mahmud A, Feely J . Beta-blockers reduce aortic stiffness in hypertension but nebivolol, not atenolol, reduces wave reflection. Am J Hypertens 2008; 21: 663–667.
Devereux GR, Wiles JD, Swaine IL . Reductions in resting blood pressure after 4 weeks of isometric exercise training. Eur J Appl Physiol 2010; 109: 601–606.
Millar PJ, Bray SR, MacDonald MJ, McCartney N . The hypotensive effects of isometric handgrip training using an inexpensive spring handgrip training device. J Cardiopulm Rehabil Prev 2008; 28: 203–207.
Taaffe DR, Galvao DA, Sharman JE, Coombes JS . Reduced central blood pressure in older adults following progressive resistance training. J Hum Hypertens 2007; 21: 96–98.
Tanimoto M, Ishii N . Effects of low-intensity resistance exercise with slow movement and tonic force generation on muscular function in young men. J Appl Physiol 2006; 100: 1150–1157.
Figueroa A, Vicil F . Post-exercise aortic hemodynamic responses to low-intensity resistance exercise with and without vascular occlusion. Scand J Med Sci Sports 2011; 21: 431–436.
Takazawa K, Tanaka N, Takeda K, Kurosu F, Ibukiyama C . Underestimation of vasodilator effects of nitroglycerin by upper limb blood pressure. Hypertension 1995; 26: 520–523.
Maloney-Hinds C, Petrofsky JS, Zimmerman G, Hessinger DA . The role of nitric oxide in skin blood flow increases due to vibration in healthy adults and adults with type 2 diabetes. Diabetes Technol Ther 2009; 11: 39–43.
Kelly RP, Millasseau SC, Ritter JM, Chowienczyk PJ . Vasoactive drugs influence aortic augmentation index independently of pulse-wave velocity in healthy men. Hypertension 2001; 37: 1429–1433.
Saul JP, Rea RF, Eckberg DL, Berger RD, Cohen RJ . Heart rate and muscle sympathetic nerve variability during reflex changes of autonomic activity. Am J Physiol 1990; 258 (3 Part 2): H713–H721.
Bogaerts AC, Delecluse C, Claessens AL, Troosters T, Boonen S, Verschueren SM . Effects of whole body vibration training on cardiorespiratory fitness and muscle strength in older individuals (a 1-year randomised controlled trial). Age Ageing 2009; 38: 448–454.
Sanada K, Miyachi M, Tanimoto M, Yamamoto K, Murakami H, Okumura S, Gando Y, Suzuki K, Tabata I, Higuchi M . A cross-sectional study of sarcopenia in Japanese men and women: reference values and association with cardiovascular risk factors. Eur J Appl Physiol 2010; 110: 57–65.
Acknowledgements
We thank Phillip Snider, Amber Ramey, Eric Borr and Jenna McNamara for their assistance in data collection. We also thank Edzard Zeinstra and Power Plate International for providing the vibration platforms.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Figueroa, A., Gil, R., Wong, A. et al. Whole-body vibration training reduces arterial stiffness, blood pressure and sympathovagal balance in young overweight/obese women. Hypertens Res 35, 667–672 (2012). https://doi.org/10.1038/hr.2012.15
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/hr.2012.15
Keywords
This article is cited by
-
Effect of exercise training on arterial stiffness in obese and overweight children: a meta-analysis
European Journal of Pediatrics (2022)
-
Impact of low-intensity resistance and whole-body vibration training on aortic hemodynamics and vascular function in postmenopausal women
Hypertension Research (2019)
-
Blood flow restriction increases myoelectric activity and metabolic accumulation during whole-body vibration
European Journal of Applied Physiology (2019)
-
Tai Chi and whole-body vibrating therapy in sarcopenic men in advanced old age: a clinical randomized controlled trial
European Journal of Ageing (2019)
-
Passive exercise to improve quality of life, activities of daily living, care burden and cognitive functioning in institutionalized older adults with dementia – a randomized controlled trial study protocol
BMC Geriatrics (2018)