Abstract
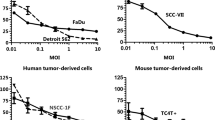
Oncolytic adenoviruses are being investigated as potential anti-cancer agents. Selective lytic replication in cancer cells is essential for an effective and safe treatment. In this study, we compared 11 oncolytic adenoviruses in relevant cell cultures to assess their use for treating oral cancer and pre-cancerous lesions. We determined the cytotoxicity of oncolytic adenovirus infection and calculated selectivity indices for cytotoxicity to cancer cells compared with normal oral keratinocytes and fibroblasts. Keratinocytes were very sensitive to wild-type adenovirus serotype 5 (Ad5); 1- to 3-log more than head and neck squamous cell carcinoma (HNSCC) cells. The potencies of oncolytic adenoviruses to kill HNSCC cells within 7 days after infection ranged from approximately 10 times less potent to approximately 10 times more potent than Ad5. The selectivity indices determined on fibroblasts and keratinocytes differed markedly. Two oncolytic adenoviruses were more selective than Ad5 for HNSCC cells compared with fibroblasts; and five viruses showed selective replication on HNSCC cells compared with keratinocytes. Overall, CRAd-S.RGD with E1A driven by the survivin promoter and an infectivity-enhancing capsid modification showed the most favourable cytotoxicity pattern; being very potent in killing HNSCC cells, only slightly less effective than Ad5 in killing pre-neoplastic keratinocytes and the least toxic to normal keratinocytes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Aghi M, Martuza RL . Oncolytic viral therapies—the clinical experience. Oncogene 2005; 24: 7802–7816.
Lin E, Nemunaitis J . Oncolytic viral therapies. Cancer Gene Ther 2004; 11: 643–664.
Alemany R . Cancer selective adenoviruses. Mol Aspects Med 2007; 28: 42–58.
Haviv YS, Curiel DT . Engineering regulatory elements for conditionally-replicative adeno-viruses. Curr Gene Ther 2003; 3: 357–385.
Fukuda K, Abei M, Ugai H, Seo E, Wakayama M, Murata T et al. E1A, E1B double-restricted adenovirus for oncolytic gene therapy of gallbladder cancer. Cancer Res 2003; 63: 4434–4440.
Gomez-Manzano C, Balague C, Alemany R, Lemoine MG, Mitlianga P, Jiang H et al. A novel E1A-E1B mutant adenovirus induces glioma regression in vivo. Oncogene 2004; 23: 1821–1828.
Howe JA, Demers GW, Johnson DE, Neugebauer SE, Perry ST, Vaillancourt MT et al. Evaluation of E1-mutant adenoviruses as conditionally replicating agents for cancer therapy. Mol Ther 2000; 2: 485–495.
Johnson L, Shen A, Boyle L, Kunich J, Pandey K, Lemmon M et al. Selectively replicating adenoviruses targeting deregulated E2F activity are potent, systemic antitumor agents. Cancer Cell 2002; 1: 325–337.
Sauthoff H, Pipiya T, Heitner S, Chen S, Bleck B, Reibman J et al. Impact of E1a modifications on tumor-selective adenoviral replication and toxicity. Mol Ther 2004; 10: 749–757.
Hermiston TW, Kuhn I . Armed therapeutic viruses: strategies and challenges to arming oncolytic viruses with therapeutic genes. Cancer Gene Ther 2002; 9: 1022–1035.
Guedan S, Gros A, Cascallo M, Vile R, Mercade E, Alemany R . Syncytia formation affects the yield and cytotoxicity of an adenovirus expressing a fusogenic glycoprotein at a late stage of replication. Gene Therapy 2008; 15: 1240–1245.
Mi J, Li ZY, Ni S, Steinwaerder D, Lieber A . Induced apoptosis supports spread of adenovirus vectors in tumors. Hum Gene Ther 2001; 12: 1343–1352.
Sauthoff H, Pipiya T, Heitner S, Chen S, Norman RG, Rom WN et al. Late expression of p53 from a replicating adenovirus improves tumor cell killing and is more tumor cell specific than expression of the adenoviral death protein. Hum Gene Ther 2002; 13: 1859–1871.
van Beusechem VW, van den Doel PB, Grill J, Pinedo HM, Gerritsen WR . Conditionally replicative adenovirus expressing p53 exhibits enhanced oncolytic potency. Cancer Res 2002; 62: 6165–6171.
Braakhuis BJ, Tabor MP, Kummer JA, Leemans CR, Brakenhoff RH . A genetic explanation of Slaughter's concept of field cancerization: evidence and clinical implications. Cancer Res 2003; 63: 1727–1730.
Tabor MP, Brakenhoff RH, Ruijter-Schippers HJ, Van Der Wal JE, Snow GB, Leemans CR et al. Multiple head and neck tumors frequently originate from a single preneoplastic lesion. Am J Pathol 2002; 161: 1051–1060.
Khuri FR, Nemunaitis J, Ganly I, Arseneau J, Tannock IF, Romel L et al. A controlled trial of intratumoral ONYX-015, a selectively-replicating adenovirus, in combination with cisplatin and 5-fluorouracil in patients with recurrent head and neck cancer. Nat Med 2000; 6: 879–885.
Morley S, MacDonald G, Kirn D, Kaye S, Brown R, Soutar D . The dl1520 virus is found preferentially in tumor tissue after direct intratumoral injection in oral carcinoma. Clin Cancer Res 2004; 10: 4357–4362.
Nemunaitis J, Ganly I, Khuri F, Arseneau J, Kuhn J, McCarty T et al. Selective replication and oncolysis in p53 mutant tumors with ONYX-015, an E1B-55kD gene-deleted adenovirus, in patients with advanced head and neck cancer: a phase II trial. Cancer Res 2000; 60: 6359–6366.
Garber K . China approves world's first oncolytic virus therapy for cancer treatment. J Natl Cancer Inst 2006; 98: 298–300.
Rudin CM, Cohen EE, Papadimitrakopoulou VA, Silverman Jr S, Recant W, El-Naggar AK et al. An attenuated adenovirus, ONYX-015, as mouthwash therapy for premalignant oral dysplasia. J Clin Oncol 2003; 21: 4546–4552.
Tu CL, Chang W, Xie Z, Bikle DD . Inactivation of the calcium sensing receptor inhibits E-cadherin-mediated cell–cell adhesion and calcium-induced differentiation in human epidermal keratinocytes. J Biol Chem 2008; 283: 3519–3528.
Herz J, Gerard RD . Adenovirus-mediated transfer of low density lipoprotein receptor gene acutely accelerates cholesterol clearance in normal mice. Proc Natl Acad Sci USA 1993; 90: 2812–2816.
Jelsma TN, Howe JA, Evelegh CM, Cunniff NF, Skiadopoulos MH, Floroff MR et al. Use of deletion and point mutants spanning the coding region of the adenovirus 5 E1A gene to define a domain that is essential for transcriptional activation. Virology 1988; 163: 494–502.
Fueyo J, Gomez-Manzano C, Alemany R, Lee PS, McDonnell TJ, Mitlianga P et al. A mutant oncolytic adenovirus targeting the Rb pathway produces anti- glioma effect in vivo. Oncogene 2000; 19: 2–12.
Heise C, Hermiston T, Johnson L, Brooks G, Sampson-Johannes A, Williams A et al. An adenovirus E1A mutant that demonstrates potent and selective systemic anti-tumoral efficacy. Nat Med 2000; 6: 1134–1139.
Bischoff JR, Kirn DH, Williams A, Heise C, Horn S, Muna M et al. An adenovirus mutant that replicates selectively in p53-deficient human tumor cells. Science 1996; 274: 373–376.
Shen Y, Kitzes G, Nye JA, Fattaey A, Hermiston T . Analyses of single-amino-acid substitution mutants of adenovirus type 5 E1B-55K protein. J Virol 2001; 75: 4297–4307.
Zhu ZB, Chen Y, Makhija SK, Lu B, Wang M, Rivera AA et al. Survivin promoter-based conditionally replicative adenoviruses target cholangiocarcinoma. Int J Oncol 2006; 29: 1319–1329.
Zhu ZB, Rivera AA, Makhija SK, Lu B, Wang M, Izumi M et al. Targeting lung cancer using an infectivity enhanced CXCR4-CRAd. Lung Cancer 2007; 55: 145–156.
Suzuki K, Alemany R, Yamamoto M, Curiel DT . The presence of the adenovirus E3 region improves the oncolytic potency of conditionally replicative adenoviruses. Clin Cancer Res 2002; 8: 3348–3359.
Tollefson AE, Scaria A, Hermiston TW, Ryerse JS, Wold LJ, Wold WS . The adenovirus death protein (E3-11.6K) is required at very late stages of infection for efficient cell lysis and release of adenovirus from infected cells. J Virol 1996; 70: 2296–2306.
Suzuki K, Fueyo J, Krasnykh V, Reynolds PN, Curiel DT, Alemany R . A conditionally replicative adenovirus with enhanced infectivity shows improved oncolytic potency. Clin Cancer Res 2001; 7: 120–126.
Bazan-Peregrino M, Carlisle RC, Hernandez-Alcoceba R, Iggo R, Homicsko K, Fisher KD et al. Comparison of molecular strategies for breast cancer virotherapy using oncolytic adenovirus. Hum Gene Ther 2008; 19: 873–886.
Lockley M, Fernandez M, Wang Y, Li NF, Conroy S, Lemoine N et al. Activity of the adenoviral E1A deletion mutant dl922-947 in ovarian cancer: comparison with E1A wild-type viruses, bioluminescence monitoring, and intraperitoneal delivery in icodextrin. Cancer Res 2006; 66: 989–998.
Geoerger B, Vassal G, Opolon P, Dirven CM, Morizet J, Laudani L et al. Oncolytic activity of p53-expressing conditionally replicative adenovirus AdDelta24-p53 against human malignant glioma. Cancer Res 2004; 64: 5753–5759.
Goodrum FD, Ornelles DA . The early region 1B 55-kilodalton oncoprotein of adenovirus relieves growth restrictions imposed on viral replication by the cell cycle. J Virol 1997; 71: 548–561.
Gaballah K, Hills A, Curiel D, Hallden G, Harrison P, Partridge M . Lysis of dysplastic but not normal oral keratinocytes and tissue-engineered epithelia with conditionally replicating adenoviruses. Cancer Res 2007; 67: 7284–7294.
Hutchin ME, Pickles RJ, Yarbrough WG . Efficiency of adenovirus-mediated gene transfer to oropharyngeal epithelial cells correlates with cellular differentiation and human coxsackie and adenovirus receptor expression. Hum Gene Ther 2000; 11: 2365–2375.
van Zeeburg HJ, Snijders PJ, Pals G, Hermsen MA, Rooimans MA, Bagby G et al. Generation and molecular characterization of head and neck squamous cell lines of Fanconi anemia patients. Cancer Res 2005; 65: 1271–1276.
Whyte P, Ruley HE, Harlow E . Two regions of the adenovirus early region 1A proteins are required for transformation. J Virol 1988; 62: 257–265.
He TC, Zhou S, da Costa LT, Yu J, Kinzler KW, Vogelstein B . A simplified system for generating recombinant adenoviruses. Proc Natl Acad Sci USA 1998; 95: 2509–2514.
Acknowledgements
This work was supported by Dutch Cancer Society Grant VU 2004-3045.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Gene Therapy website
Rights and permissions
About this article
Cite this article
van Zeeburg, H., Huizenga, A., Brink, A. et al. Comparison of oncolytic adenoviruses for selective eradication of oral cancer and pre-cancerous lesions. Gene Ther 17, 1517–1524 (2010). https://doi.org/10.1038/gt.2010.99
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2010.99
Keywords
This article is cited by
-
Winners and losers and oncolytic adenoviruses: who should be down in the mouth?
Gene Therapy (2010)