Featured
-
-
Article
| Open Access Plant carbonic anhydrase-like enzymes in neuroactive alkaloid biosynthesis
Plant carbonic anhydrase-like enzymes in neuroactive alkaloid biosynthesis
We show how neuroactive alkaloids from clubmosses are biosynthesized, which reveals an unexpected role for carbonic anhydrase-like enzymes in alkaloid scaffold formation.
- Ryan S. Nett
- , Yaereen Dho
- & Elizabeth S. Sattely
-
Article |
 Biosynthesis of selenium-containing small molecules in diverse microorganisms
Biosynthesis of selenium-containing small molecules in diverse microorganisms
Using genome mining, a dedicated pathway for the production of selenosugars and selenoneine is uncovered, thereby expanding the use of biological selenium.
- Chase M. Kayrouz
- , Jonathan Huang
- & Mohammad R. Seyedsayamdost
-
Article |
 A kinase-cGAS cascade to synthesize a therapeutic STING activator
A kinase-cGAS cascade to synthesize a therapeutic STING activator
Leveraging enzymatic selectivity, a single reaction stream provides a single diastereomer of the cyclic dinucleotide MK-1454, a promising immune-oncology drug candidate, without the use of protecting groups or chiral auxiliaries.
- John A. McIntosh
- , Zhijian Liu
- & Matthew L. Maddess
-
Article
| Open Access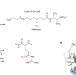 Crystallographic snapshots of a B12-dependent radical SAM methyltransferase
Crystallographic snapshots of a B12-dependent radical SAM methyltransferase
Structural and spectroscopic studies show how a B12-dependent radical SAM enzyme catalyses unique and challenging alkylation chemistry, including protein post-translational modification required for methane biosynthesis.
- Cameron D. Fyfe
- , Noelia Bernardo-García
- & Olivier Berteau
-
Article |
 A naturally inspired antibiotic to target multidrug-resistant pathogens
A naturally inspired antibiotic to target multidrug-resistant pathogens
The discovery and synthesis of a colistin congener provide a promising clinical lead against mcr-1-encoding colistin-resistant pathogens, which are responsible for an increasing number of deaths from antibiotic-resistant infections.
- Zongqiang Wang
- , Bimal Koirala
- & Sean F. Brady
-
Article |
 Discovery, characterization and engineering of ligases for amide synthesis
Discovery, characterization and engineering of ligases for amide synthesis
A family of enzymes—coronafacic acid ligases, involved in the synthesis of bacterial phytotoxins—are found to catalyse amide bond formation with a wide range of substrates.
- Michael Winn
- , Michael Rowlinson
- & Jason Micklefield
-
Article |
 An enzymatic Alder-ene reaction
An enzymatic Alder-ene reaction
Analysis of two homologous groups of fungal pericyclases demonstrates how they can catalyse either an Alder-ene reaction—which has not previously been found in nature—or a hetero-Diels–Alder reaction.
- Masao Ohashi
- , Cooper S. Jamieson
- & Yi Tang
-
Letter |
 Enzyme-catalysed [6+4] cycloadditions in the biosynthesis of natural products
Enzyme-catalysed [6+4] cycloadditions in the biosynthesis of natural products
Enzymatic catalysis of pericyclic [6+4] cycloaddition reactions to form ten-membered rings is observed during biosynthesis of the macrocyclic antibiotic streptoseomycin, and the mechanism of these transformations is established.
- Bo Zhang
- , Kai Biao Wang
- & Hui Ming Ge
-
Letter |
 Discovery of a pathway for terminal-alkyne amino acid biosynthesis
Discovery of a pathway for terminal-alkyne amino acid biosynthesis
Microbial generation of a terminal-alkyne-containing amino acid can be encoded into E. coli and provides the potential for in vivo generation of proteins and natural products for click chemistry.
- J. A. Marchand
- , M. E. Neugebauer
- & M. C. Y. Chang
-
Letter |
 An N-nitrosating metalloenzyme constructs the pharmacophore of streptozotocin
An N-nitrosating metalloenzyme constructs the pharmacophore of streptozotocin
The metalloenzyme SznF catalyses the formation of an N–N bond in the biosynthesis of streptozotocin, providing insights into the enzymatic assembly of an N-nitroso group.
- Tai L. Ng
- , Roman Rohac
- & Emily P. Balskus
-
Letter |
 Enzymatic assembly of carbon–carbon bonds via iron-catalysed sp3 C–H functionalization
Enzymatic assembly of carbon–carbon bonds via iron-catalysed sp3 C–H functionalization
Evolved iron-containing enzymes based on cytochrome P450 achieve selective intermolecular alkylation of sp3 C–H bonds through a carbene C–H insertion strategy.
- Ruijie K. Zhang
- , Kai Chen
- & Frances H. Arnold
-
Letter |
 Trapping biosynthetic acyl-enzyme intermediates with encoded 2,3-diaminopropionic acid
Trapping biosynthetic acyl-enzyme intermediates with encoded 2,3-diaminopropionic acid
A method for encoding the non-canonical amino acid 2,3-diaminopropionate into proteins allows key acyl intermediates in the biosynthesis of valinomycin to be trapped, providing insight into the oligomerization and cyclization reactions involved.
- Nicolas Huguenin-Dezot
- , Diego A. Alonzo
- & Jason W. Chin
-
Letter |
 Resistance-gene-directed discovery of a natural-product herbicide with a new mode of action
Resistance-gene-directed discovery of a natural-product herbicide with a new mode of action
Fungal genome mining targeted to self-resistance genes close to biosynthetic gene clusters identifies a pathway that produces aspterric acid, which proves to be a potent inhibitor of plant growth.
- Yan Yan
- , Qikun Liu
- & Yi Tang
-
Letter |
 Genetically programmed chiral organoborane synthesis
Genetically programmed chiral organoborane synthesis
A genetically encoded platform can produce chiral organoboranes in bacteria with high turnover, enantioselectivity and chemoselectivity, and can be tuned and configured through DNA manipulation.
- S. B. Jennifer Kan
- , Xiongyi Huang
- & Frances H. Arnold
-
Letter |
 SAM-dependent enzyme-catalysed pericyclic reactions in natural product biosynthesis
SAM-dependent enzyme-catalysed pericyclic reactions in natural product biosynthesis
The enzyme LepI is found to be capable of catalysing several natural-product pericyclic transformations, including a hetero-Diels–Alder reaction and a retro-Claisen rearrangement.
- Masao Ohashi
- , Fang Liu
- & Yi Tang
-
Article |
 Elucidation of the biosynthesis of the methane catalyst coenzyme F430
Elucidation of the biosynthesis of the methane catalyst coenzyme F430
The enzymes and pathway involved in the biosynthesis of coenzyme F430 are identified, completing our understanding of how members of the cyclic modified tetrapyrrole family are constructed.
- Simon J. Moore
- , Sven T. Sowa
- & Martin J. Warren
-
Letter |
 Mycocerosic acid synthase exemplifies the architecture of reducing polyketide synthases
Mycocerosic acid synthase exemplifies the architecture of reducing polyketide synthases
A hybrid crystal structure of Mycobacterium smegmatis mycocerosic acid synthase, a multienzyme involved in the biosynthesis of mycobacterial branched-chain fatty acids, exemplifies the organization of fully-reducing polyketide synthases.
- Dominik A. Herbst
- , Roman P. Jakob
- & Timm Maier
-
Letter |
 Synthetic cycle of the initiation module of a formylating nonribosomal peptide synthetase
Synthetic cycle of the initiation module of a formylating nonribosomal peptide synthetase
WebX-ray crystal structures are presented of each major step of the assembly-line synthesis by the initiation module of the nonribosomal peptide synthetase (NRPS) LgrA; the structures reveal large conformational changes, demonstrating a requirement for NRPSs to be very dynamic.
- Janice M. Reimer
- , Martin N. Aloise
- & T. Martin Schmeing
-
Letter |
 Structures of two distinct conformations of holo-non-ribosomal peptide synthetases
Structures of two distinct conformations of holo-non-ribosomal peptide synthetases
X-ray crystal structures of two distinct steps in the catalytic cycle of non-ribosomal peptide synthetases are described, offering the potential to generate novel products through engineering enzyme activity.
- Eric J. Drake
- , Bradley R. Miller
- & Andrew M. Gulick
-
Letter |
 A new cyanogenic metabolite in Arabidopsis required for inducible pathogen defence
A new cyanogenic metabolite in Arabidopsis required for inducible pathogen defence
Untargeted metabolomics and coexpression analysis uncovers the complete biosynthetic pathway of a previously unknown Arabidopsis metabolite, 4-hydroxyindole-3-carbonyl nitrile (4-OH-ICN), which harbours cyanogenic functionality.
- Jakub Rajniak
- , Brenden Barco
- & Elizabeth S. Sattely
-
Letter |
 X-domain of peptide synthetases recruits oxygenases crucial for glycopeptide biosynthesis
X-domain of peptide synthetases recruits oxygenases crucial for glycopeptide biosynthesis
Glycopeptide antibiotics are biosynthesized by non-ribosomal peptide synthetases, which contain a previously uncharacterized ‘X-domain’ now shown to recruit three cytochrome P450 oxygenases that are necessary for the antibiotics to achieve their final, active conformation.
- Kristina Haslinger
- , Madeleine Peschke
- & Max J. Cryle
-
Letter |
 β-Lactam formation by a non-ribosomal peptide synthetase during antibiotic biosynthesis
β-Lactam formation by a non-ribosomal peptide synthetase during antibiotic biosynthesis
The monocyclic β-lactam rings of the nocardicin family of antibiotics are biosynthesized by an unprecedented activity of non-ribosomal peptide synthetases, a mechanism that is distinct from the pathways to the other classes of β-lactam antibiotics.
- Nicole M. Gaudelli
- , Darcie H. Long
- & Craig A. Townsend
-
Letter |
 Metabolic coupling of two small-molecule thiols programs the biosynthesis of lincomycin A
Metabolic coupling of two small-molecule thiols programs the biosynthesis of lincomycin A
Mycothiol and ergothioneine are shown to have important roles in the biosynthesis of lincomycin A, a sulfur-containing antibiotic that is used to treat severe Gram-positive bacterial infections in people who cannot receive penicillin antibiotics.
- Qunfei Zhao
- , Min Wang
- & Wen Liu
-
Letter |
 Structure and mechanism of the tRNA-dependent lantibiotic dehydratase NisB
Structure and mechanism of the tRNA-dependent lantibiotic dehydratase NisB
Structural and biochemical studies show that the biosynthesis of the food preservative nisin involves the tRNA-dependent glutamylation of serine and threonine.
- Manuel A. Ortega
- , Yue Hao
- & Satish K. Nair
-
Letter |
 Co-opting sulphur-carrier proteins from primary metabolic pathways for 2-thiosugar biosynthesis
Co-opting sulphur-carrier proteins from primary metabolic pathways for 2-thiosugar biosynthesis
How sulphur is incorporated into sulphur-containing secondary metabolites is poorly understood; here, the bacterium Amycolatopsis orientalis is shown to co-opt sulphur-carrier proteins from primary metabolic pathways to facilitate the biosynthesis of sulphur-containing natural products.
- Eita Sasaki
- , Xuan Zhang
- & Hung-wen Liu
-
Letter |
 Trapping the dynamic acyl carrier protein in fatty acid biosynthesis
Trapping the dynamic acyl carrier protein in fatty acid biosynthesis
A highly specific chemical crosslinking method is used to trap a complex between an acyl carrier protein and a fatty acid dehydratase during fatty acid biosynthesis; subsequent X-ray crystallography, NMR spectroscopy and molecular dynamics simulations techniques enable the detailed study of this complex.
- Chi Nguyen
- , Robert W. Haushalter
- & Michael D. Burkart
-
Letter |
 Chasing acyl carrier protein through a catalytic cycle of lipid A production
Chasing acyl carrier protein through a catalytic cycle of lipid A production
The crystal structures of three forms of Escherichia coli acyl carrier protein engaging LpxD, an acyltransferase in the lipid A biosynthetic pathway, are reported.
- Ali Masoudi
- , Christian R. H. Raetz
- & Charles W. Pemble IV
-
Letter |
 Vinylogous chain branching catalysed by a dedicated polyketide synthase module
Vinylogous chain branching catalysed by a dedicated polyketide synthase module
This study shows the structural and biochemical characterization of a new type of polyketide synthase module that catalyses the vinylogous addition of a malonyl unit to an unsaturated thioester, generating a branch in the growing polyketide chain; this characterization provides a mechanism by which the structural diversity of polyketide natural products can be increased.
- Tom Bretschneider
- , Joel B. Heim
- & Christian Hertweck
-
Letter |
 Pyrimidine homeostasis is accomplished by directed overflow metabolism
Pyrimidine homeostasis is accomplished by directed overflow metabolism
Here, the authors identify a previously unknown regulatory strategy used by Escherichia coli to control end-product levels of the pyrimidine biosynthetic pathway: this involves feedback regulation of the near-terminal pathway enzyme UMP kinase, with accumulation of UMP prevented by its degradation to uridine through UmpH, a phosphatase with a previously unknown function.
- Marshall Louis Reaves
- , Brian D. Young
- & Joshua D. Rabinowitz
-
Letter |
 Mechanistic studies of an unprecedented enzyme-catalysed 1,2-phosphono-migration reaction
Mechanistic studies of an unprecedented enzyme-catalysed 1,2-phosphono-migration reaction
The non-haem-iron-dependent enzyme HppE catalyses the final step in the biosynthesis of fosfomycin, a broad-spectrum, clinically useful antibiotic; here it is shown that HppE can also catalyse a 1,2-phosphono-migration reaction, previously undocumented for any enzyme.
- Wei-chen Chang
- , Mishtu Dey
- & Hung-wen Liu
-
News & Views Forum |
 A constructive debate
A constructive debate
Synthetic chemistry has long been used to prepare useful compounds — especially those that are hard to obtain from natural sources. But synthetic biology is coming of age as an alternative strategy. A biologist and two chemists debate the merits of their fields' synthetic prowess.
- Jay D. Keasling
- , Abraham Mendoza
- & Phil S. Baran
-
News & Views |
 Another aspect of nature's ingenuity
Another aspect of nature's ingenuity
Eyewitnesses are sometimes asked to identify a culprit from a line-up of people associated with a crime scene. An enzyme — iridoid synthase — that catalyses an unusual reaction has been identified by a similar approach. See Letter p.138
- Joe Chappell
-
Letter |
 An alternative route to cyclic terpenes by reductive cyclization in iridoid biosynthesis
An alternative route to cyclic terpenes by reductive cyclization in iridoid biosynthesis
Iridoids are a large family of bicyclic natural products that possess anticancer, anti-inflammatory, antifungal and antibacterial activities; here the essential cyclization step in their biosynthesis is identified, opening up the possibility of production of naturally occurring and synthetic variants of iridoids for use in pharmacy or agriculture.
- Fernando Geu-Flores
- , Nathaniel H. Sherden
- & Sarah E. O’Connor
-
Letter |
 Mitochondrial Atpif1 regulates haem synthesis in developing erythroblasts
Mitochondrial Atpif1 regulates haem synthesis in developing erythroblasts
Atpif1, a mitochondrial ATPase inhibitor, was identified as a zebrafish anemic mutant, pinotage, providing an important link in our understanding of the relationship between mitochondrial homeostasis and haem synthesis and identifying a gene that may have a role in human iron, haem and mitochondrial diseases.
- Dhvanit I. Shah
- , Naoko Takahashi-Makise
- & Barry H. Paw
-
Letter |
 Serine is a natural ligand and allosteric activator of pyruvate kinase M2
Serine is a natural ligand and allosteric activator of pyruvate kinase M2
The M2 isoform of pyruvate kinase (PKM2) is a key glycolytic enzyme that is overexpressed in cancer cells; here, serine is shown to bind to and directly activate PKM2, and the resulting reduction in enzyme activity under serine-deprivation conditions is shown to lead to the diversion of glucose-derived carbon to promote serine biosynthesis required for cell proliferation.
- Barbara Chaneton
- , Petra Hillmann
- & Eyal Gottlieb
-
Research Highlights |
The mystery of high seas methane
-
Article |
 Watching DNA polymerase η make a phosphodiester bond
Watching DNA polymerase η make a phosphodiester bond
Atomic-resolution time courses of phosphodiester bond formation catalysed by DNA polymerase η reveal transient intermediate states and an unexpected third metal ion in the reaction mechanism.
- Teruya Nakamura
- , Ye Zhao
- & Wei Yang
-
News & Views |
 Favouring the unfavoured
Favouring the unfavoured
The naturally occurring antibiotic lasalocid A contains a chemical structure that is not expected to form readily. The enzyme that catalyses the formation of this structure has been identified, and its activity is a revelation. See Letter p.355
- David E. Cane
-
Letter |
 Enzymatic catalysis of anti-Baldwin ring closure in polyether biosynthesis
Enzymatic catalysis of anti-Baldwin ring closure in polyether biosynthesis
The X-ray crystal structure of the epoxide hydrolase Lsd19 in complex with its substrate and product analogue is determined, providing insight into a general mechanism of enzyme-catalysed formation of polyether natural products.
- Kinya Hotta
- , Xi Chen
- & Chu-Young Kim
-
Letter |
 Saccharomyces cerevisiae THI4p is a suicide thiamine thiazole synthase
Saccharomyces cerevisiae THI4p is a suicide thiamine thiazole synthase
- Abhishek Chatterjee
- , N. Dinuka Abeydeera
- & Tadhg P. Begley
-
Letter |
 S-nitrosylation of NADPH oxidase regulates cell death in plant immunity
S-nitrosylation of NADPH oxidase regulates cell death in plant immunity
- Byung-Wook Yun
- , Angela Feechan
- & Gary J. Loake
-
Letter |
 Polyamine sensing by nascent ornithine decarboxylase antizyme stimulates decoding of its mRNA
Polyamine sensing by nascent ornithine decarboxylase antizyme stimulates decoding of its mRNA
- Leo Kurian
- , R. Palanimurugan
- & R. Jürgen Dohmen
-
Letter |
 Functional genomics reveal that the serine synthesis pathway is essential in breast cancer
Functional genomics reveal that the serine synthesis pathway is essential in breast cancer
- Richard Possemato
- , Kevin M. Marks
- & David M. Sabatini
-
News |
 Domino synthesis mimics nature's chemistry
Domino synthesis mimics nature's chemistry
A new approach to making natural products delivers rapid results.
- James Mitchell Crow
-
News & Views |
 Life imitates art
Life imitates art
The biosynthetic route to a naturally occurring insecticide, spinosyn A, has been established. One of the enzymes involved might catalyse a reaction that, although widely used by chemists, has proved elusive in nature. See Letter p.109
- Wendy L. Kelly
-
Letter |
 Enzyme-catalysed [4+2] cycloaddition is a key step in the biosynthesis of spinosyn A
Enzyme-catalysed [4+2] cycloaddition is a key step in the biosynthesis of spinosyn A
- Hak Joong Kim
- , Mark W. Ruszczycky
- & Hung-wen Liu
-
Letter |
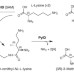 The complete biosynthesis of the genetically encoded amino acid pyrrolysine from lysine
The complete biosynthesis of the genetically encoded amino acid pyrrolysine from lysine
Pyrrolysine is the twenty-second naturally occurring amino acid, and it is required for the formation of methane from methylamines. Pyrrolysine is the last remaining natural amino acid with a poorly characterized biosynthetic pathway. Here it is shown that the radical SAM protein PylB converts lysine to 3-methylornithine, which then undergoes a PylC-catalysed condensation with another lysine to generate a product that is oxidized by PylD to generate pyrrolysine.
- Marsha A. Gaston
- , Liwen Zhang
- & Joseph A. Krzycki
-
Letter |
 Taxadiene synthase structure and evolution of modular architecture in terpene biosynthesis
Taxadiene synthase structure and evolution of modular architecture in terpene biosynthesis
The first X-ray crystal structure of a diterpene cyclase is reported — this enzyme, taxadiene synthase, catalyses the cyclization of an isoprenoid in the first committed step of the biosynthesis of the cancer chemotherapeutic drug Taxol. The C-terminal catalytic domain binds and activates the substrate in a manner seen in class I terpenoid cyclases, but the N-terminal domain and a third 'insertion' domain together adopt the fold of a class II terpenoid cyclase. It is proposed that this enzyme could be the ancestral progenitor of all terpenoid cyclases.
- Mustafa Köksal
- , Yinghua Jin
- & David W. Christianson
-
News |
 Hydrogen production comes naturally to ocean microbe
Hydrogen production comes naturally to ocean microbe
Cyanobacterium gives off hydrogen as by-product of day-to-day processes.
- Katharine Sanderson

 Computational prediction of complex cationic rearrangement outcomes
Computational prediction of complex cationic rearrangement outcomes