Abstract
The increasing number of people with dementia globally illustrates the urgent need to reduce dementia’s scale and impact. Lifetime social participation may affect dementia risk by increasing cognitive reserve, and through brain maintenance by reducing stress and improving cerebrovascular health. It may therefore have important implications for individual behavior and public health policy aimed at reducing dementia burden. Observational study evidence indicates that greater social participation in midlife and late life is associated with 30–50% lower subsequent dementia risk, although some of this may not be causal. Social participation interventions have led to improved cognition but, partly due to short follow-up and small numbers of participants, no reduction in risk of dementia. We summarize the evidence linking social participation with dementia, discuss potential mechanisms by which social participation is likely to reduce and mitigate the impact of neuropathology in the brain, and consider the implications for future clinical and policy dementia prevention interventions.
Similar content being viewed by others
Main
Dementia is highly prevalent with 50 million people globally estimated to currently have dementia, and is forecast to grow markedly so that by 2050 there will be 152 million people living with dementia worldwide1. Dementia has a substantial impact on individuals and their families and is reported to be the most feared condition in older people2. Health and social care costs are estimated to be US $263 billion per year globally, with this figure forecast to rise to US $1.6 trillion by 2050, accounting for 11–17% of all health care spending3. The public health impact of dementia calls attention to the need to identify approaches to delay or prevent dementia. Projections indicate that delaying dementia onset by 5 years would halve prevalence4, and a 3-year delay would reduce dementia cost by one-third5.
There is suggestive evidence that changes in behaviors and improvements in health during the life course affect the risk of developing dementia. This is supported by studies of successive generations of older people showing that age-standardized dementia incidence and prevalence rates in several high-income countries have changed6, attributed to better health and education7. For example, in the UK Cognitive Function and Ageing Studies, a 20% reduced incidence rate was observed between 1990 and 2010 (ref. 8) and in the US Health and Retirement Study, the prevalence rate decreased by 13% between 2000 and 2012 (ref. 7). These findings have been replicated in other settings, although the opposite trend has been found in Japan9. Dementia rates may have declined through adoption of a healthier lifestyle, such as reduced tobacco smoking10 or alcohol intake, and earlier and more effective treatment of hypertension11 and diabetes12, or better access to education7. Conversely, an increasingly Western diet and physical inactivity leading to metabolic syndrome may account for Japan’s increased dementia prevalence rate13.
The overall evidence was summarized in the 2020 Lancet Commission for Dementia Prevention, Intervention and Care, which estimated that 12 potentially modifiable risk factors—lower education, hypertension, hearing impairment, smoking, obesity, depression, physical inactivity, diabetes, alcohol intake, poor sleep, air pollution and low social participation—may account for 40% of worldwide dementia risk14. The proportion may be higher in low-income and middle-income countries where these risk factors overall are more common, although there is variation between and within countries in the prevalence of individual risk factors15,16.
Social participation has attracted interest as a potential factor influencing general health and well-being, as well as specifically cognitive health and risk of dementia; it may promote healthy lifestyles, reduce stress and build cognitive health. Furthermore, longstanding societal changes and those related to the coronavirus disease 2019 (COVID-19) pandemic have increased the focus on social isolation and loneliness17,18. The 2019 World Health Organization’s (WHO) guidelines on risk reduction for cognitive decline and dementia19 did not include social participation, but subsequent research findings in this area necessitate reexamination of the role of social participation.
Understanding the association between different aspects of social participation with dementia risk is important as it could guide measures to improve social participation behaviors at the individual and societal levels. While societal changes may contribute to social isolation and reducing social participation, there is little research evidence on social participation trends over past decades. Some studies show that formal social participation increased in older adults in the USA20, and loneliness levels in Swedish older adults were stable between 1992 and 2014 (ref. 21). The COVID-19 pandemic brought focus on social isolation and participation as measures to curb disease spread led to social distancing in most countries whereby reducing social participation was mandatory, with subsequent effects on mental health22. Social participation has not returned to pre-pandemic levels, and the effect of these changes on the health of older adults and on the risk of dementia remains unclear. However, some evidence suggests that people with dementia deteriorated more rapidly during the pandemic23, emphasizing the importance of considering whether and how to target increasing social participation.
Social participation is defined as ‘a person’s involvement in activities providing interactions with others in community life and in important shared spaces … based on the societal context and what individuals want and is meaningful to them’24. We will use the term here to encompass engagement of individuals in sociable leisure activities (with focus on activities that are undertaken with other people), contact with social networks and their satisfaction with this participation. Definitions of this and related constructs are in Table 1.
In this Review, we aim to elucidate the links between social participation and dementia risk to inform current and future approaches to prevent dementia through clinical interventions and policy measures to increase social participation. We do so by examining the current observational and interventional evidence on social participation and the associated risk of dementia and the mechanisms underlying these links. We conclude this Review by summarizing the implications for future treatments and policy.
Association between social participation and dementia
There is growing epidemiological evidence for an association between aspects of social participation and risk of dementia. These observational studies have examined social participation in midlife or late life and followed participants to compare dementia incidence, usually ascertained from clinical examination using diagnostic criteria or health records, between those with more and less social participation. Diagnosis of the cause of dementia (for example, Alzheimer’s disease or vascular dementia) is typically not ascertained with high-quality examination in these studies. Methods used to assess social participation have largely relied on self-report questionnaires completed by participants. These include validated scales such as the Berkman–Syme Index25 for aspects of social network contact, and the UCLA scale26 for assessing loneliness. Several unvalidated scales, created for the purposes of a study, have been used to assess participation in social activities.
Social activity engagement
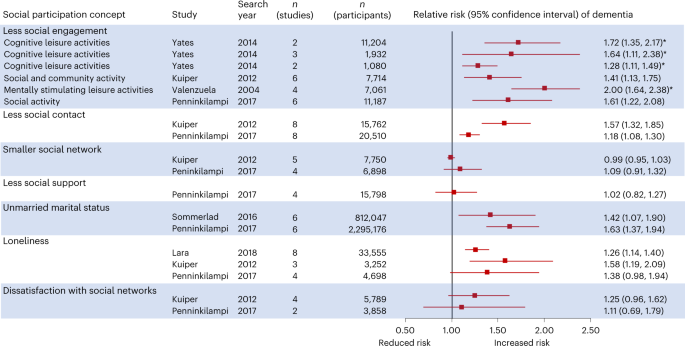
Accumulating evidence suggests that having less engagement in social activities is associated with higher relative risk of dementia27,28,29,30 (Fig. 1). Such studies generally combine activities into a composite activity scale regardless of how social these activities are; some involve other people (board games or group discussions), while others are solitary (crosswords and reading).
Summary of the evidence from six systematic reviews27,37,38,39,44,144 about the association in epidemiological studies of different aspects of social participation with dementia risk. Relative risks indicate the risk of dementia associated with having less social participation; those marked with an asterisk were inverted from the published paper for consistency.
However, a major challenge of observational research of risk factors for dementia is the potential for reverse causation underpinning observed associations, so follow-up duration must be considered in this literature. Pathological changes develop decades before the diagnostic threshold for dementia is reached, and some symptoms of dementia precede diagnosis by many years during this preclinical phase31,32. Thus, establishing that a particular characteristic or behavior is present before dementia diagnosis does not confirm its status as a risk factor, as that characteristic may instead be an early consequence of dementia pathology arising during dementia development.
Many studies have a short follow-up; for example, five of the seven studies in one review of the effect of cognitive leisure activities had less than 6 years of an interval between activity measurement and dementia assessment, and all these studies found significant associations, whereas the remaining two studies, with 9 and 12 years of follow-up did not27. To reduce the risk of reverse causation, studies require a long interval between measurement of social participation and assessment for dementia and should exclude people with impaired cognitive status at baseline. Recent studies in the UK, Japan and China with 8–15 years of follow-up have linked more social engagement, participation in cognitive, intellectual and social activities, membership of social organizations and community engagement with lower dementia risk28,29,30,33,34.
Further insight can be gained from examination of trajectories of engagement in social activities leading up to dementia diagnosis using data from studies with repeated assessments of social participation. In two such studies of UK adults, not participating in social or leisure activities was associated with higher risk of dementia only with less than 10 years of follow-up35,36. And two studies found that declining social activity engagement was associated with higher dementia risk28,35 suggesting that this decline occurs during the preclinical phase of dementia.
Altogether, less frequent engagement with social and leisure activities is linked with elevated subsequent dementia risk, but this association is at least partly due to reverse causation. Social activity engagement frequently co-occurs with cognitively stimulating activity, and social contact with others, so the specific contribution of activity engagement is unclear.
Social networks and social contact
Less frequent social contact with others is shown in two reviews to be associated with higher risk of dementia37,38 (Fig. 1.). Being unmarried, taken as a marker for having less lifetime social contact with others, is associated with elevated risk of dementia in two systematic reviews38,39 (Fig. 1). One study found that having children was associated with lower dementia risk40, partly linked to marital status. Marital and parental status may also influence individuals’ ability to manage their health, and access informal care, which may account for some of this association. By contrast, simply having a smaller social network has not been found to be associated with dementia risk37,38, suggesting that interacting with people frequently, rather than interacting with more people, is important.
Duration of follow-up may again explain the inconsistency in results from these studies; seven of the eight studies in one of the reviews had less than 4 years of follow-up, and so it is impossible to judge the direction of causation37. However, a subsequent study with a longer duration of follow-up found that less frequent social contact in UK adults at age 60, particularly with friends rather than relatives, was associated with greater dementia risk and worse cognitive decline 15 years later41. Two studies of participants in the UK Biobank study with 9 years42 and 12 years43 of mean follow-up found that dementia risk was higher for those who were more socially isolated, with this finding consistent across different levels of genetic risk42 and linked to lower gray matter volume43.
Hence, there is strong evidence that frequent social contact with others, but not having a larger social network, reduces the risk of dementia.
Subjective aspects of social participation
Three reviews found that reporting loneliness is associated with greater dementia risk compared to people who were not lonely37,38,44 (Fig. 1). Five subsequent studies of loneliness in the USA over 10 years45,46,47, over 14 years in Sweden48 and over 5 years in Japan49 have found it to be associated with 34–91% elevated dementia risk. Some of these studies found the association to persist after adjusting for potential confounders including social isolation46,49,50. However, studies of UK adults found that loneliness was not independently associated with dementia risk and that the unadjusted association was probably mediated by depression symptoms42,43. Reporting less social support was also unrelated to dementia risk in a systematic review38, although a recent study of Korean adults with 10 years of follow-up found low emotional, but not practical, support was associated with elevated risk of all-cause dementia and Alzheimer’s disease in women, but not men51. The inconsistency in these findings may be due to measurement error because few of the included studies used validated scales measuring loneliness; cultural variation in conceptualization and expression of loneliness; duration and perceived intensity of loneliness exposure; and differences in studies’ adjustment for potentially explanatory covariates especially social isolation, socioeconomic status and depression.
These findings suggest that loneliness but not perceived social support is associated with higher dementia risk, but it is unclear whether this association is only a consequence of having more social contact with others.
Social participation and cognitive decline
Our Review focused on dementia as the most clinically important syndrome, but some studies considered the link between social participation and conditions along the disease pathway such as mild cognitive impairment52, providing further supportive evidence. Several studies have also examined the association of social participation with decline in cognitive function, which is relevant as cognitive decline is the core feature of dementia. While associations between potential risk factors and cognitive decline cannot directly indicate elevated dementia risk, the finding of an association is consistent with this possibility53. Two systematic reviews of social relationships and cognitive decline found that both the quantity and quality of social relationship participation were linked to subsequent increased risk of cognitive impairment54,55. However, the methodological quality of included studies precluded strong conclusions about beneficial aspects of social relationships for cognition, and the direction underpinning the association was unclear as 15 of the 19 studies had less than 5 years of follow-up, meaning that reverse causation was likely.
A further review found associations between social activity, networks and support and global cognitive function, although similar limitations apply to this Review56. Subsequent studies of German adults over 85 years found that small and declining social networks were associated with worse cognitive decline57 and this finding was replicated in a study of Japanese adults who were followed for over 10 years58. Employment is frequently a source of social contact for many, and previous studies have found that retirement is associated with cognitive decline59,60,61, which may indicate that withdrawal from work-related social networks has detrimental effects on cognitive function. However, leaving employment does not only cause loss of social contact, but may reflect loss of cognitively stimulating occupation, physical labor, social status and income, and the direction of the relationship is not clear as individuals with already declining cognitive function may be more likely to retire if given a choice. The report that countries in which people retire younger have a higher rate of declining cognition compared to countries with later retirement age suggests that there may be some causal relationship62. In line with this is the finding of more memory loss in retirees in a study adjusting for health as well as sociodemographic factors63.
A systematic review of studies on loneliness and cognitive decline found that loneliness was associated with worse general cognitive function, intelligence quotient, processing speed and delayed and immediate recall64. However, confounding factors such as depression and social isolation may also have contributed to these findings. Furthermore, the directional relationship between cognitive impairment and loneliness was not clear as, for example, one study found that poor verbal ability predicted loneliness later in life65.
More social network contact and less loneliness is associated with lower cognitive decline, which is consistent with a protective effect against dementia, although methodological limitations in this literature persist. Retirement is associated with cognitive decline, which may partly be due to less social participation.
Limitations of observational evidence
The epidemiological evidence is limited by the probability of reverse causation in studies with a short follow-up that have typically found strong associations of social participation with lower dementia risk. Studies with a longer follow-up have found mixed results and, where a protective effect was reported, the magnitude of associations has generally been small. Whether this is because of lower bias from reverse causation in these studies or greater effect dilution bias due to exposure misclassification remains unclear. In long-term follow-up studies, a single baseline measurement may not accurately indicate social participation across the life course, and the longer the follow-up is, the more likely changes in social participation are to occur, contributing to exposure misclassification, resulting in artificial attenuation of effect estimates. These findings highlight the importance of studies with longer-term follow-up and repeated assessment of social participation, ideally comprising age ranges from early adulthood or even earlier. Such studies would allow further exploration of the life-course influence of social participation on dementia risk, including the ability to identify whether there are critical or sensitive risk periods, or whether an effect of social participation on dementia results from accumulation or chains of risk, guiding future interventions. Ultimately, it is likely that there is a bidirectional relationship between social participation and dementia, whereby low participation accelerates cognitive decline, and this decline impairs social participation.
Confounding is a major source of bias in observational evidence. Associations persisted in studies that adjusted for a wide range of potential confounders, but it is possible that residual confounding persists. Other study designs, such as Mendelian randomization, may help to tackle reverse causation and minimize other sources of confounding, and evidence from one study suggests only partial benefit of social engagement43,66. There is evidence of publication bias in the literature as reported in two reviews38,54. Sharing data to combine cohorts in larger, individual participant-data meta-analyses67 may help to address publication bias, and increase power and generalizability of the findings. Many studies tend to recruit people with lower risk68,69 who are less likely to develop dementia than the general population meaning that findings may not be generalizable.
Most meta-analyses dichotomize the social exposure variables although social participation is not a binary concept, so nuanced examination to clarify dose–response associations and different dimensions of social participation, as used in some studies41, would be beneficial. The heterogeneity in social participation measurement related to the different construct definitions and the lack of validated established instruments is another contributing factor to the heterogeneity in findings. There is also limited evidence for whether social participation has a differential effect on different subtypes of dementia, as relatively few studies have examined dementia subtypes. One study found that the association of leisure activity participation was similar for Alzheimer’s dementia and vascular dementia70, and results were inconsistent for whether marital status association with dementia differed for Alzheimer’s and vascular etiology39.
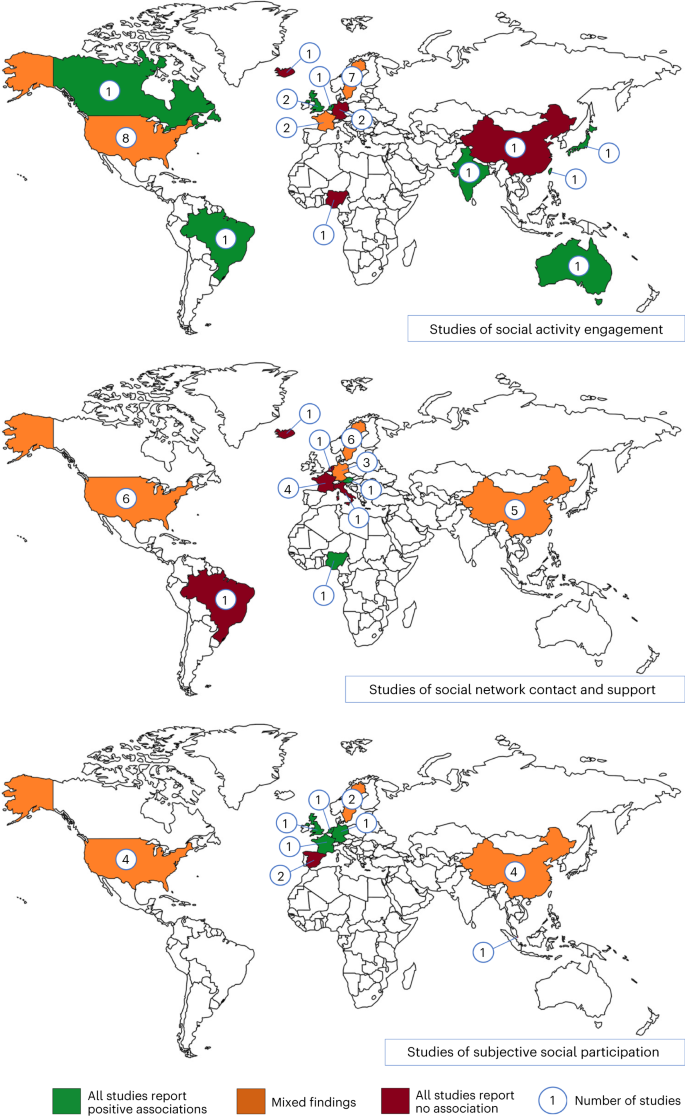
Social participation is culturally patterned, making it important to understand how the contribution of social participation varies in different settings. Most studies have been conducted in North America, Europe and China, and there is little consistency in the findings (Fig. 2). This may be due to differences in study design and measurement of social participation in these studies. It may also reflect that social participation effects differ across settings as the nature of the participation and associated health behaviors vary. The prevalence of social behaviors is also likely to be conditioned by contextual factors, for example, social isolation is reported less commonly in Latin American countries, India and China than the global average15. Within countries, there is also ethnic and socioeconomic variation in social participation, for example, in Aotearoa/New Zealand, the contribution of social isolation to dementia risk was greater in European and Māori populations groups than Asian and Pasifika populations71, and in Brazil it was greater in poor regions than wealthy ones, although there was no ethnic variation16. Interventions therefore need to be designed with consideration to culture and social environments and target different components of social participation to be acceptable and effective in different cultures.
Summary of the findings of studies included in six systematic reviews of observational studies related to social participation and dementia27,37,38,39,44,144, according to the country studied. Studies were divided into those examining social activity engagement, social network contact and support and subjective aspects of social participation. The number of studies of each kind conducted in each country and the consistency of findings are shown. Green indicates countries where all included studies found that more social participation was associated with lower dementia risk (positive association). Red indicates countries where all studies found no association. Orange indicates mixed findings.
In summary, major methodological limitations of previous observational literature on social participation and dementia risk include reverse causation bias, confounding, publication bias, poor generalizability and measurement error. There are few studies in lower-income and middle-income countries, thus we do not know whether these findings generalize to these settings.
Potential underlying mechanisms
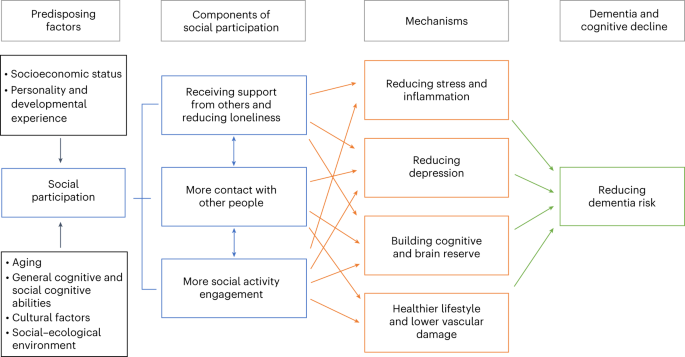
Social participation could plausibly result in reduced dementia risk, irrespective of genetic risk42,72, through one or more mechanisms. These include building cognitive reserve73, and better brain maintenance74 by reducing neurodegeneration by lower vascular damage, reduced stress75, promotion of healthy lifestyles and behaviors and enhanced mental stimulation76 (Fig. 3).
Summary of the mechanisms by which social participation may reduce dementia risk. Neuropsychological factors such as personality, cognition and developmental experiences, and social factors like socioeconomic status and the social and ecological environment predispose individuals to having more or less need for and access to social participation in a range of different domains. Social participation may act through several mechanisms, including brain maintenance by reducing stress, inflammation and cerebrovascular damage, building cognitive reserve and reducing depression to reduce dementia risk.
Cognitive reserve
Social contact with others may build cognitive reserve, implying that increased cognitive adaptability improves the ability of individuals to tolerate neuropathology without corresponding decline in cognition and function73. Socialization is cognitively demanding, requiring deployment of numerous social cognitive domains77, as well as planning, memory and language and, according to the social brain hypothesis, social complexity may be the major driving force for evolutionary brain development78. Social participation may therefore exercise cognitive domains, reducing vulnerability to late-life decline79. An autopsy study found that more social contact before death modified the relationship between neuropathology and cognition, such that more amyloid load and neurofibrillary tangle density was less strongly associated with cognitive decline in people who had more frequent social contacts80, supporting the hypothesis that cognitive reserve was greater in those with more frequent social contact. Another study found that the detrimental effect of diabetes on dementia risk was attenuated by social participation81.
Brain maintenance
Social participation may affect dementia risk through brain maintenance, which refers to reduced accumulation of brain pathology and preservation of a healthy brain conferring better late-life cognitive function74. Better social health, defined as less loneliness, more social support and being married, was associated in one study with greater brain volume82. Another study showed that socially isolated individuals had lower frontal and temporal gray matter volumes and that differences in gray matter volume partly mediated the association between social isolation at baseline and cognitive function at follow-up43. This may partly reflect reverse causation whereby people with larger brains are able to maintain more complex social lives. However, mental stimulation within the occupational context has been linked to greater hippocampal volume83 and lower levels of proteins that inhibit axonogenesis and synaptogenesis84, although it is unclear to what extent the level of mental stimulation during social participation is important. Brain maintenance from social participation may therefore be achieved through several mechanisms.
Lifestyle behaviors and vascular risk
Social participation may improve brain maintenance through healthy lifestyle behaviors. Social isolation is associated with increased mortality through health behaviors such as smoking85, high alcohol consumption86 and cardiovascular diseases87. All-cause dementia and dementia subtypes are related to cardiovascular and cerebrovascular health88,89 and heavy alcohol consumption is an established dementia risk factor14, so the mechanisms by which lower social contact is associated with cardiovascular risk factors and illness may also extend to dementia risk. Contact with others may, in many cases, model and encourage better health behaviors leading to lower risk of vascular disease and subsequent better cognitive health and lower dementia risk. This is supported by a study of Ecuadorean adults in whom poor social relationships and lower support networks were associated with worse progression of cerebrovascular disease90. However, smoking and alcohol consumption may accompany social participation to mitigate socioeconomic stress, so some social contact may not confer cerebrovascular health benefits.
Stress, accelerated aging and inflammation
The effect of social participation on brain maintenance could act through the stress response91. A detrimental effect of stress through, for example, restraint, social isolation or other chronic stressors, on hippocampal networks has been demonstrated in animal models92, and persistent midlife stress has been associated with elevated dementia risk in human epidemiological studies93. Social isolation and poor social support are associated with biological stress markers including dysregulation of the hypothalamic–pituitary–adrenal axis and cortisol responses94, increased sympathetic nervous system activation95, accelerated cellular aging96,97,98 and raised inflammatory markers99, suggesting that social isolation may affect dementia risk through the pathological effect of stress. The important role of systemic inflammation in the etiology of dementia is increasingly being recognized100,101,102,103.
Immunity
Some large-scale studies have linked severe infections to increased dementia risk104,105, suggesting that human-to-human contact could increase dementia risk through the adverse effect of infection. The reduced human contact of socially isolated individuals might therefore potentially decrease their exposure to transmitted pathogens. However, some studies have observed increased risk of severe infections among socially isolated individuals106, reflecting overall adverse impacts of social isolation on general health, and this may also reflect the immune suppression linked to stress induced by social isolation107. In an analysis making a distinction between subjective loneliness and objective social isolation, the occurrence of hospital-treated infections was higher among lonely people than others, while no robust association was observed between social isolation and the incidence of hospital-treated infections108. This preliminary evidence requires further study, and research on long-term impacts of COVID-19-related isolation may be informative.
Depression
Social participation, depression and cognitive function are interrelated in a complex fashion. Late-life depression is consistently associated with dementia risk109, although this may be partially due to low mood arising in preclinical dementia110. However, social participation may arise from and result in low mood, so it is plausible that social participation affects cognition and dementia through affecting mood.
Interventions for social participation
Clarifying the potential of social participation as a target for intervention is crucial but the design of research to test whether increasing social participation affects dementia risk is challenging. It would be unethical to deprive some people of aspects of social participation and impractical to enforce greater social participation in others. While desire for social participation is an inherent aspect of human nature that may have neurobiological underpinnings111, and social contact with other people is a core part of individual identity and important for wider society112, it is not clear how to improve social participation. Furthermore, some individuals may not wish to change their social participatory habits as, although social contact with others is usually associated with positive affective experiences, personality characteristics such as introversion113, autistic traits114 or internalized stigma related to mental illness115 may encourage some to seek and prefer solitude.
Local cultures and traditions are important in the roles of individuals within wider society, and an intervention to encourage social participation would need to be acceptable to participants, feasible to administer and sustainable to potentially have an effect. Furthermore, considering the gradual onset of dementia pathology and symptoms, and the likely lifelong contribution of symptoms, interventions may best be delivered at a relatively young age, and many decades of follow-up would be required to assess dementia risk, which is impractical. Interventions have therefore often focused on cognitive change, rather than dementia incidence, as a primary outcome. Randomized controlled trials (RCTs) of social interventions or multicomponent interventions that include a social focus are summarized in Table 2.
Social interventions for cognitive function
Evidence from studies testing social interventions for people at risk of developing dementia is variable. Interventions for improving cognition through social activities, networks and relationships were summarized in a 2017 systematic review56, which found three RCTs, the results of which were not suitable for meta-analysis due to study design heterogeneity. The trials of between 120 and 235 participants tested social activity interventions, which all included facilitator-led group activities. One Finnish study led to a significant but small improvement in cognitive performance on the Alzheimer’s Disease Assessment Scale–Cognitive Subscale (ADAS-cog)116 at 3 months (1 point difference, −2.6 (3.4, −1.8) versus −1.6 (−2.2, −1.0)) and better subjective cognitive function117. The other studies were from China118 and the USA119 and showed better but non-significant overall cognitive performance compared to control groups, although one reported improved immediate recall and verbal fluency118 and a 0.6% increase in total brain volume compared to the control group118. These studies were too small and the follow-up time too short to identify any effect on the risk of incident mild cognitive impairment or dementia.
Interventions aimed at improving loneliness in older adults have usually focused on encouraging study participants to engage in purposeful activity and maintain contact with their social networks120. Although there is some evidence of efficacy of these programs in reducing loneliness compared to control groups, no studies have assessed effects on cognition121.
Interventions to increase social participation through facilitator-led group activities have not consistently been shown in RCTs to improve general cognitive function; studies were too small and short to identify any effect on the development of dementia.
Interventions combining social participation with other strategies
Interventions using a combination of strategies to improve cognition have gained increasing interest as they target several risk factors and can be tailored according to individual risk profiles. A Cochrane review of multi-domain interventions122 was inconclusive about whether these overall affected dementia risk, but the review determined that only two of these interventions targeted social participation—the Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability (FINGER) RCT123 and an RCT of Korean community-dwelling adults124.
The FINGER study was a 2-year intervention for people with elevated vascular risk for dementia where the intervention consisted of advice on diet, physical activity, cognitive training and improved management of vascular risk. There was no explicit social element to the study, but it was achieved implicitly through group meetings123. The intervention resulted in less cognitive decline than the control group with a small effect size (Cohen’s d = 0.13), and this difference persisted irrespective of baseline risk factors125, but results using dementia as an outcome have not yet been published. An RCT of a multi-domain intervention delivered over 18 months in Korea included promotion of cognitive and social activities alongside physical activity, alcohol and smoking interventions and weight loss, lean body mass and healthy diet with four different modes of delivery including, at its most intensive, bimonthly health worker-initiated visits, counseling, and rewards for adherence to the program124. Only the most intensive approach to delivering the intervention led to better cognitive function compared to the control group at 18 months follow-up on the mini-mental state examination (MMSE; mean performance, 0.99 points better than control (standard error, 0.49)). Both interventions were time-consuming, intensive and costly with small effect sizes for cognition.
A subsequent RCT, SUPERBRAIN, tested a multi-domain intervention including social activities through group meetings and additional scheduled monthly social activities126. The Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) score improved significantly at 24 weeks of follow-up of participants in the intervention group who received the intervention in a clinical setting (5.46 ± 7.50, P = 0.004) or at home (5.50 ± 8.14, P = 0.004) compared to the control group (−0.74 ± 11.51)127. This improvement was associated with changes in learning and cognitive neural networks128.
In all these studies, the specific social contribution to any positive effect is unclear due to the multi-domain nature of the intervention. One study described which health behaviors changed and found that social participation did not significantly increase after the intervention, and analysis instead attributed the cognitive benefit to increased cognitive activity124. Other studies to test combination strategies for dementia prevention with interventions, including AgeWell.de129, MIND China130 and US Pointer131, are still ongoing.
Interventions targeting several domains including social participation have shown cognitive benefit, although they are time-consuming and costly, and no effect on dementia risk has yet been demonstrated. The specific contribution of social participation to improving cognition is not yet established.
Quasi-experimental research
Considering the challenges of RCTs in this area, quasi-experimental research may be informative. COVID-19-related social isolation measures, introduced to contain the disease, resulted in a reduction in social participation, which affected some, such as those living alone, more than others. The evidence on how this affected dementia risk may become evident in future years. However, some studies have found that social restrictions may have affected cognitive function.
A review of the effect of COVID-19-related isolation on the cognition of people with dementia found that 60% of 15 studies reported a detrimental effect on cognition23. Similar findings have been reported in a study of adults without dementia in which adherence to stricter isolation was associated with worse short-term performance on cognitive tasks132, but this deterioration of performance may be related to anxiety or affective symptoms. A study found that the link between COVID-19-related social restrictions and poor cognitive performance was more pronounced in younger than older individuals, which would not be expected if poor cognitive performance were related to acceleration, or greater susceptibility to the effects, of neurodegeneration133. Findings should be interpreted with caution as reverse causation may partly account for findings; for example, one cross-sectional study of Chinese adults found that worse cognition was associated with lower adherence to COVID-19 prevention regulations134 and COVID-19 itself may increase risk of dementia135. Thus, while isolation measures may be required during periods of high COVID-19 rates or other future infectious disease epidemics, the principle of ‘isolating the virus rather than isolating people’ should be used to decide on measures.
Uncertainties of interventional evidence
There are several uncertainties from the interventional research. Although there is preliminary evidence that group social interventions can improve cognition, there is no evidence about prevention of dementia, and interventions that have demonstrated effect on cognition may not carry through to delay or prevent dementia. However, accelerated cognitive decline begins years before dementia diagnosis and this decline predicts dementia risk136. Therefore, the US Food and Drug Administration have encouraged the use of cognitive and functional endpoints, rather than dementia, for drug trials137, so improvement in cognition can be considered necessary but not sufficient evidence for identifying dementia prevention strategies. Extended follow-up of previous studies by attaining dementia status from clinical records will support existing evidence.
Additional follow-up may also clarify whether interventions lead to long-lasting changes in social behavior, which would guide timing of an intervention. Considering that dementia results from accumulated lifetime risk, it may be that changing behavior earlier would confer more benefit. The move toward multi-domain interventions, rather than interventions targeting a single risk factor, is reasonable as it allows tailored interventions to change risk profiles. However, it obscures the understanding whether a single component of the intervention is an active ingredient. This may be helped by process evaluation of trials, examination of mechanisms through assessment of biomarkers and analyses that determine adherence to different intervention components and estimate their effects124.
Policy implications
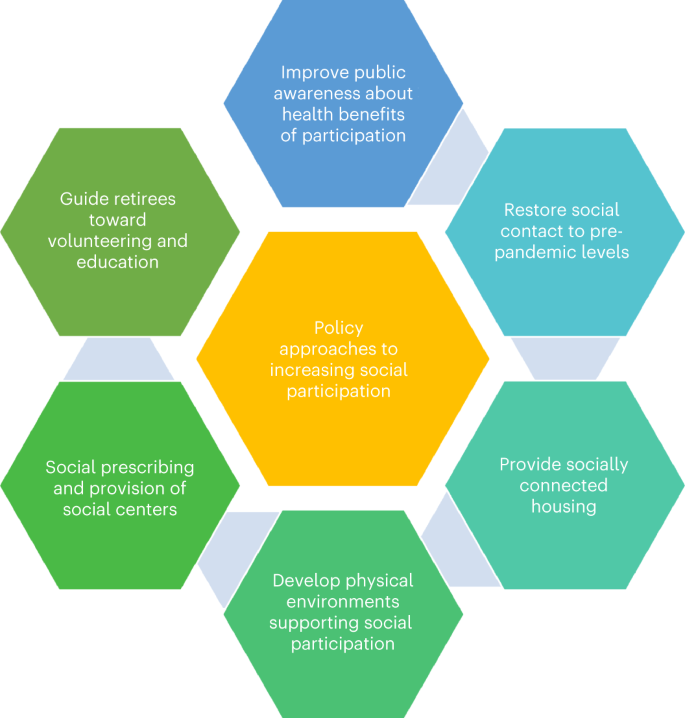
Public health policy could promote social participation through several approaches, focusing particularly on the areas with the strongest evidence for dementia prevention, related to social contact and activity engagement (Fig. 4). Policy should aim to restore pre-pandemic social participation, including promotion of face-to-face contact and support alongside remote approaches where necessary during periods of high contagion. There should be guidance and support for retirees, because retirement is a potential sensitive period, such as by providing volunteering opportunities and further education to promote social participation. Provision of adequate socially connected housing for older people is a focus of several governmental and third-sector organizations138, and has potential to reduce social isolation and loneliness as well as provide support networks for older people. Physical environmental adaptations by optimizing urban planning, accessibility and infrastructure, as recommended in the WHO’s Global Age Friendly Cities: A Guide139, could help to promote social inclusion. Finally, provision of leisure time with social connection through social centers would promote social and cognitive activity and participation.
Conclusion
This Review highlights the current evidence for the effect of social participation on dementia risk. There is a pressing need to clarify risk factors for dementia to guide prevention approaches to mitigate the projected future rise in the number of people with dementia. The evidence for a direct causal link between social participation and dementia is not definitive due to limitations in observational research but is consistent and biologically plausible, with evidence for several potential mechanistic pathways underlying an association. There are few RCTs of social interventions and they are consistent with showing a positive effect on cognition, but none include dementia as an outcome.
Evidence has accumulated since the WHO19 published guidelines on risk reduction for cognitive decline and dementia19, which rated evidence on a range of risk factors based on confidence about whether the desirable effects of the intervention outweigh any undesirable effects. The guidelines reported that there was insufficient evidence to recommend social activity for reducing cognitive decline or dementia due to limited and inconclusive evidence and risk of bias arising from reverse causality. There is now considerably more evidence for the link between social participation and dementia, particularly for social contact with others, although the evidence for activity engagement is weaker, suggesting that these guidelines should be reassessed and that future guidelines should highlight social participation as a modifiable risk factor for dementia. There is good evidence that social participation overall is beneficial for general health and well-being140 and the WHO141 position paper on optimizing brain health concludes that social connection is an important determinant for brain health across the life course141.
Implications for treatment
The US National Academies of Sciences recommend applying quality of evidence standards, which take into account the priority of the problem, how substantial the benefits and harms are, the certainty of the evidence, the value of the outcomes to stakeholders, how an intervention’s clinical and cost effectiveness compares with others, resource requirements, the impact on health equity, the intervention’s acceptability and the feasibility of implementation142. These standards could be applied to evaluate social participation interventions. While trials today suggest potential individual-level strategies, there is currently insufficient RCT effectiveness or cost-effectiveness evidence for definitive recommendations, so further research is required on effective and scalable interventions.
Social isolation is closely linked with socioeconomic deprivation143, so targeting individuals to improve social participation may be inequitable and ineffective, but health status can be shaped by policies with a societal rather than individual focus. The lower numbers of people with dementia due to higher education standards or reduced smoking are a consequence of societal changes, by, for example, increasing the number of mandatory years in education, and improving public understanding of the harm of, and limiting the opportunities for, cigarette smoking. The key area for applying the understanding that social participation is beneficial for brain health is therefore likely to be in social policies, community awareness, and advice around individuals’ choices related to social participation over the life cycle. These policy approaches should particularly aim to support those from socioeconomic and cultural groups at highest risk of dementia who may find it hardest to access social participation, by reducing financial and logistical barriers to social activities and participation to have a substantial benefit for health and society.
References
Patterson, C. World Alzheimer report 2018 (Alzheimer’s Disease International, 2018).
Alzheimer’s Research UK. Dementia Attitudes Monitor: Shaping Breakthroughs in Public Understanding of Dementia and Attitudes to Research (2021).
Velandia, P. P. Global and regional spending on dementia care from 2000–2019 and expected future health spending scenarios from 2020–2050: an economic modelling exercise. EClinicalMedicine 45, 101337 (2022).
Seshadri, S. & Wolf, P. A. Lifetime risk of stroke and dementia: current concepts, and estimates from the Framingham Study. Lancet Neurol. 6, 1106–1114 (2007).
Zissimopoulos, J., Crimmins, E. & Clair, P. S. The value of delaying Alzheimer’s disease onset. Forum Health Econ. Policy 18, 25–39 (2014).
Röhr, S., Pabst, A., Luck, T. & Riedel-Heller, S. G. Is dementia incidence declining in high-income countries? A systematic review and meta-analysis. Clin. Epidemiol. 10, 1233–1247 (2018).
Langa, K. M. et al. A comparison of the prevalence of dementia in the United States in 2000 and 2012. JAMA Intern. Med. 177, 51–58 (2016).
Matthews, F. et al. A two decade dementia incidence comparison from the Cognitive Function and Ageing Studies I and II. Nat. Commun. 7, 11398 (2016).
Wu, Y.-T. et al. The changing prevalence and incidence of dementia over time—current evidence. Nat. Rev. Neurol. 13, 327–339 (2017).
Sreeramareddy, C. T. & Aye, S. N. Changes in adult smoking behaviours in ten global adult tobacco survey (GATS) countries during 2008–2018—a test of ‘hardening’hypothesis’. BMC Public Health 21, 1209 (2021).
Mosterd, A. et al. Trends in the prevalence of hypertension, antihypertensive therapy, and left ventricular hypertrophy from 1950 to 1989. N. Engl. J. Med. 340, 1221–1227 (1999).
Fang, M., Wang, D., Coresh, J. & Selvin, E. Trends in diabetes treatment and control in US adults, 1999–2018. N. Engl. J. Med. 384, 2219–2228 (2021).
Kiyohara, Y. Epidemiology of dementia: the Hisayama study. Nihon Rinsho 72, 601–606 (2014).
Livingston, G. et al. Dementia prevention, intervention, and care: 2020 report of the Lancet Commission. Lancet 396, 413–446 (2020).
Mukadam, N., Sommerlad, A., Huntley, J. & Livingston, G. Population attributable fractions for risk factors for dementia in low-income and middle-income countries: an analysis using cross-sectional survey data. Lancet Glob. Health 7, e596–e603 (2019).
Suemoto, C. K. et al. Risk factors for dementia in Brazil: differences by region and race. Alzheimers Dement. https://doi.org/10.1002/alz.12820 (2022).
Surkalim, D. L. et al. The prevalence of loneliness across 113 countries: systematic review and meta-analysis. BMJ 376, e067068 (2022).
Serrat, R., Scharf, T., Villar, F. & Gómez, C. Fifty-five years of research into older people’s civic participation: recent trends, future directions. Gerontologist 60, e38–e51 (2020).
World Health Organization. Risk Reduction of Cognitive Decline and Dementia: WHO Guidelines (2019).
Ang, S. Life course social connectedness: age-cohort trends in social participation. Adv. Life Course Res. 39, 13–22 (2019).
Dahlberg, L., Agahi, N. & Lennartsson, C. Lonelier than ever? Loneliness of older people over two decades. Arch. Gerontol. Geriatrics 75, 96–103 (2018).
Sommerlad, A. et al. Social relationships and depression during the COVID-19 lockdown: longitudinal analysis of the COVID-19 Social Study. Psychol. Med. 52, 3381–3390 (2022).
Suárez-González, A., Rajagopalan, J., Livingston, G. & Alladi, S. The effect of COVID-19 isolation measures on the cognition and mental health of people living with dementia: a rapid systematic review of one year of quantitative evidence. EClinicalMedicine 39, 101047 (2021).
Levasseur, M. et al. Scoping study of definitions of social participation: update and co-construction of an interdisciplinary consensual definition. Age Ageing 51, afab215 (2022).
Berkman, L. F. & Syme, S. L. Social networks, host resistance, and mortality: a nine-year follow-up study of Alameda County residents. Am. J. Epidemiol. 109, 186–204 (1979).
Russell, D., Peplau, L. A. & Cutrona, C. E. The revised UCLA Loneliness Scale: concurrent and discriminant validity evidence. J. Pers. Soc. Psychol. 39, 472–480 (1980).
Yates, L. A., Ziser, S., Spector, A. & Orrell, M. Cognitive leisure activities and future risk of cognitive impairment and dementia: systematic review and meta-analysis. Int. Psychogeriatr. 28, 1791–1806 (2016).
Zhou, Z., Wang, P. & Fang, Y. Social engagement and its change are associated with dementia risk among Chinese older adults: a longitudinal study. Sci. Rep. 8, 1551 (2018).
Fancourt, D., Steptoe, A. & Cadar, D. Community engagement and dementia risk: time-to-event analyses from a national cohort study. J. Epidemiol. Community Health 74, 71–77 (2020).
Duffner, L. et al. The role of cognitive and social leisure activities in dementia risk: assessing longitudinal associations of modifiable and on-modifiable risk factors. Epidemiol. Psychiatr. Sci. 31, e5 (2022).
Amieva, H. et al. Prodromal Alzheimer’s disease: successive emergence of the clinical symptoms. Ann. Neurol. 64, 492–498 (2008).
Jack, C. R. Jr. et al. Hypothetical model of dynamic biomarkers of the Alzheimer’s pathological cascade. Lancet Neurol. 9, 119–128 (2010).
Almeida-Meza, P., Steptoe, A. & Cadar, D. Is engagement in intellectual and social leisure activities protective against dementia risk? Evidence from the English longitudinal study of ageing. J. Alzheimer’s Dis. 80, 555–565 (2021).
Nemoto, Y. et al. An additive effect of leading role in the organization between social participation and dementia onset among Japanese older adults: the AGES cohort study. BMC Geriatr. 17, 297 (2017).
Sommerlad, A. et al. Leisure activity participation and risk of dementia: an 18-year follow-up of the Whitehall II Study. Neurology 95, e2803–e2815 (2020).
Floud, S. et al. Cognitive and social activities and long-term dementia risk: the prospective UK Million Women Study. Lancet Public Health 6, e116–e123 (2021).
Kuiper, J. S. et al. Social relationships and risk of dementia: a systematic review and meta-analysis of longitudinal cohort studies. Ageing Res. Rev. 22, 39–57 (2015).
Penninkilampi, R., Casey, A.-N., Singh, M. F. & Brodaty, H. The association between social engagement, loneliness, and risk of dementia: a systematic review and meta-analysis. J. Alzheimer’s Dis. 66, 1619–1633 (2018).
Sommerlad, A., Ruegger, J., Singh-Manoux, A., Lewis, G. & Livingston, G. Marriage and risk of dementia: systematic review and meta-analysis of observational studies. J. Neurol. Neurosurg. Psychiatry 89, 231–238 (2018).
Sundström, A., Westerlund, O., Mousavi-Nasab, H., Adolfsson, R. & Nilsson, L.-G. The relationship between marital and parental status and the risk of dementia. Int. Psychogeriatr. 26, 749–757 (2014).
Sommerlad, A., Sabia, S., Singh-Manoux, A., Lewis, G. & Livingston, G. Association of social contact with dementia and cognition: 28-year follow-up of the Whitehall II cohort study. PLoS Med. 16, e1002862 (2019).
Elovainio, M. et al. Association of social isolation, loneliness and genetic risk with incidence of dementia: UK Biobank Cohort Study. BMJ Open 12, e053936 (2022).
Shen, C. et al. Associations of social isolation and loneliness with later dementia. Neurology 99, e164–e175 (2022).
Lara, E. et al. Does loneliness contribute to mild cognitive impairment and dementia? A systematic review and meta-analysis of longitudinal studies. Ageing Res. Rev. 52, 7–16 (2019).
Akhter‐Khan, S. C. et al. Associations of loneliness with risk of Alzheimer’s disease dementia in the Framingham Heart Study. Alzheimer’s Dement. 17, 1619–1627 (2021).
Sutin, A. R., Stephan, Y., Luchetti, M. & Terracciano, A. Loneliness and risk of dementia. J. Gerontol. B Psychol. Sci. Soc. Sci. 75, 1414–1422 (2020).
Salinas, J. et al. Association of loneliness with 10-year dementia risk and early markers of vulnerability for neurocognitive decline. Neurology 98, e1337–e1348 (2022).
Freak-Poli, R. et al. Loneliness, not social support, is associated with cognitive decline and dementia across two longitudinal population-based cohorts. J. Alzheimer’s Dis. 85, 295–308 (2022).
Shibata, M. et al. Emotional loneliness is associated with a risk of dementia in a general Japanese older population: the Hisayama study. J. Gerontol. B Psychol. Sci. Soc. Sci. 76, 1756–1766 (2021).
Akhter-Khan, S. et al. Associations of loneliness with risk of Alzheimer’s disease dementia in the Framingham Heart Study. Alzheimers Dement. 17, 1619–1627 (2021).
Oh, D. J. et al. Association of low emotional and tangible support with risk of dementia among adults 60 years and older in South Korea. JAMA Netw. Open 5, e2226260–e2226260 (2022).
Gardener, H. et al. Social connectivity is related to mild cognitive impairment and dementia. J. Alzheimer’s Dis. 84, 1811–1820 (2021).
Weuve, J. et al. Guidelines for reporting methodological challenges and evaluating potential bias in dementia research. Alzheimer’s Dement. 11, 1098–1109 (2015).
Kuiper, J. S. et al. Social relationships and cognitive decline: a systematic review and meta-analysis of longitudinal cohort studies. Int. J. Epidemiol. 45, 1169–1206 (2016).
Piolatto, M. et al. The effect of social relationships on cognitive decline in older adults: an updated systematic review and meta-analysis of longitudinal cohort studies. BMC Public Health 22, 278 (2022).
Kelly, M. E. et al. The impact of social activities, social networks, social support and social relationships on the cognitive functioning of healthy older adults: a systematic review. Syst. Rev. 6, 259 (2017).
Röhr, S. et al. Changes in social network size are associated with cognitive changes in the oldest-old. Front. Psychiatry 11, 330 (2020).
Saito, T. et al. Association between intra-individual changes in social network diversity and global cognition in older adults: does closeness to network members make a difference? J. Psychosom. Res. 151, 110658 (2021).
Roberts, B. A., Fuhrer, R., Marmot, M. & Richards, M. Does retirement influence cognitive performance? The Whitehall II Study. J. Epidemiol. Community Health 65, 958–963 (2011).
Meng, A., Nexø, M. A. & Borg, V. The impact of retirement on age related cognitive decline—a systematic review. BMC Geriatr. 17, 160 (2017).
Xue, B. et al. Effect of retirement on cognitive function: the Whitehall II cohort study. Eur. J. Epidemiol. 33, 989–1001 (2018).
Rohwedder, S. & Willis, R. J. Mental retirement. J. Econ. Perspect. 24, 119–138 (2010).
Clouston, S. A. & Denier, N. Mental retirement and health selection: analyses from the US Health and Retirement Study. Soc. Sci. Med. 178, 78–86 (2017).
Boss, L., Kang, D.-H. & Branson, S. Loneliness and cognitive function in the older adult: a systematic review. Int. Psychogeriatr. 27, 541–553 (2015).
Schnittger, R. I., Wherton, J., Prendergast, D. & Lawlor, B. A. Risk factors and mediating pathways of loneliness and social support in community-dwelling older adults. Aging Ment. Health 16, 335–346 (2012).
Shen, L.-X. et al. Social isolation, social interaction, and Alzheimer’s disease: a Mendelian randomization study. J. Alzheimer’s Dis. 80, 665–672 (2021).
Kivimäki, M. et al. IPD-Work consortium: pre-defined meta-analyses of individual-participant data strengthen evidence base for a link between psychosocial factors and health. Scand. J. Work Environ. Health 41, 312–321 (2015).
Fry, A. et al. Comparison of sociodemographic and health-related characteristics of UK Biobank participants with those of the general population. Am. J. Epidemiol. 186, 1026–1034 (2017).
Brayne, C. & Moffitt, T. E. The limitations of large-scale volunteer databases to address inequalities and global challenges in health and aging. Nat. Aging 2, 775–783 (2022).
Verghese, J. et al. Leisure activities and the risk of dementia in the elderly. N. Engl. J. Med. 348, 2508–2516 (2003).
Ma’u, E., Cullum, S., Cheung, G., Livingston, G. & Mukadam, N. Differences in the potential for dementia prevention between major ethnic groups within one country: a cross-sectional analysis of population attributable fraction of potentially modifiable risk factors in New Zealand. Lancet Reg. Health West. Pac. 13, 100191 (2021).
Dekhtyar, S. et al. Genetic risk of dementia mitigated by cognitive reserve: a cohort study. Ann. Neurol. 86, 68–78 (2019).
Stern, Y. et al. Whitepaper: Defining and investigating cognitive reserve, brain reserve, and brain maintenance. Alzheimer’s Dement. 16, 1305–1311 (2020).
Alvares Pereira, G., Silva Nunes, M. V., Alzola, P. & Contador, I. Cognitive reserve and brain maintenance in aging and dementia: an integrative review. Appl. Neuropsychol. Adult 29, 1615–1625 (2022).
Grande, G., Qiu, C. & Fratiglioni, L. Prevention of dementia in an ageing world: evidence and biological rationale. Ageing Res. Rev. 64, 101045 (2020).
Fratiglioni, L., Marseglia, A. & Dekhtyar, S. Ageing without dementia: can stimulating psychosocial and lifestyle experiences make a difference? Lancet Neurol. 19, 533–543 (2020).
Turkstra, L. S. Conversation-based assessment of social cognition in adults with traumatic brain injury. Brain Inj. 22, 397–409 (2008).
Lanooij, S. D., Eisel, U. L., Drinkenburg, W. H., van der Zee, E. A. & Kas, M. J. Influencing cognitive performance via social interactions: a novel therapeutic approach for brain disorders based on neuroanatomical mapping? Mol. Psychiatry 28, 28–33 (2022).
Scarmeas, N. & Stern, Y. Cognitive reserve and lifestyle. J. Clin. Exp. Neuropsychol. 25, 625–633 (2003).
Bennett, D. A., Schneider, J. A., Tang, Y., Arnold, S. E. & Wilson, R. S. The effect of social networks on the relation between Alzheimer’s disease pathology and level of cognitive function in old people: a longitudinal cohort study. Lancet Neurol. 5, 406–412 (2006).
Marseglia, A., Wang, H.-X., Rizzuto, D., Fratiglioni, L. & Xu, W. Participating in mental, social, and physical leisure activities and having a rich social network reduce the incidence of diabetes-related dementia in a cohort of Swedish older adults. Diabetes Care 42, 232–239 (2019).
van der Velpen, I. F. et al. Social health is associated with structural brain changes in older adults: the Rotterdam Study. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 7, 659–668 (2022).
Maguire, E. A. et al. Navigation-related structural change in the hippocampi of taxi drivers. Proc. Natl Acad. Sci. USA 97, 4398–4403 (2000).
Kivimäki, M. et al. Cognitive stimulation in the workplace, plasma proteins, and risk of dementia: three analyses of population cohort studies. BMJ 374, n1804 (2021).
Elovainio, M. et al. Contribution of risk factors to excess mortality in isolated and lonely individuals: an analysis of data from the UK Biobank cohort study. Lancet Public Health 2, e260–e266 (2017).
Kelly, S., Olanrewaju, O., Cowan, A., Brayne, C. & Lafortune, L. Alcohol and older people: a systematic review of barriers, facilitators and context of drinking in older people and implications for intervention design. PLoS ONE 13, e0191189 (2018).
Holt-Lunstad, J., Smith, T. B., Baker, M., Harris, T. & Stephenson, D. Loneliness and social isolation as risk factors for mortality: a meta-analytic review. Perspect. Psychol. Sci. 10, 227–237 (2015).
Larson, E. B. et al. Exercise is associated with reduced risk for incident dementia among persons 65 years of age and older. Ann. Intern. Med. 144, 73–81 (2006).
Barberger-Gateau, P. et al. Dietary patterns and risk of dementia The Three-City cohort study. Neurology 69, 1921–1930 (2007).
Del Brutto, O. H., Mera, R. M., Recalde, B. Y., Rumbea, D. A. & Del Brutto, V. J. High social risk influence progression of white matter hyperintensities of presumed vascular origin: a prospective study in community-dwelling older adults. Stroke 53, 2577–2584 (2022).
Cacioppo, J. T., Cacioppo, S., Capitanio, J. P. & Cole, S. W. The neuroendocrinology of social isolation. Annu. Rev. Psychol. 66, 733–767 (2015).
Rothman, S. M. & Mattson, M. P. Adverse stress, hippocampal networks, and Alzheimer’s disease. Neuromolecular Med. 12, 56–70 (2010).
Johansson, L. et al. Common psychosocial stressors in middle-aged women related to longstanding distress and increased risk of Alzheimer’s disease: a 38-year longitudinal population study. BMJ Open 3, e003142 (2013).
Stafford, M., Gardner, M., Kumari, M., Kuh, D. & Ben-Shlomo, Y. Social isolation and diurnal cortisol patterns in an ageing cohort. Psychoneuroendocrinology 38, 2737–2745 (2013).
Friedler, B., Crapser, J. & McCullough, L. One is the deadliest number: the detrimental effects of social isolation on cerebrovascular diseases and cognition. Acta Neuropathol. 129, 493–509 (2015).
Carroll, J. E., Roux, A. V. D., Fitzpatrick, A. L. & Seeman, T. Low social support is associated with shorter leukocyte telomere length in late life: multi-ethnic study of atherosclerosis (MESA). Psychosom. Med. 75, 171–177 (2013).
Mitchell, A. M., Kowalsky, J. M., Epel, E. S., Lin, J. & Christian, L. M. Childhood adversity, social support, and telomere length among perinatal women. Psychoneuroendocrinology 87, 43–52 (2018).
Wilson, S. J. et al. Loneliness and telomere length: immune and parasympathetic function in associations with accelerated aging. Ann. Behav. Med. 53, 541–550 (2019).
Smith, K. J., Gavey, S., RIddell, N. E., Kontari, P. & Victor, C. The association between loneliness, social isolation and inflammation: a systematic review and meta-analysis. Neurosci. Biobehav. Rev. 112, 519–541 (2020).
Walker, K. A. et al. Systemic inflammation during midlife and cognitive change over 20 years: the ARIC study. Neurology 92, e1256–e1267 (2019).
Sweeney, M. D., Zhao, Z., Montagne, A., Nelson, A. R. & Zlokovic, B. V. Blood–brain barrier: from physiology to disease and back. Physiol. Rev. 99, 21–78 (2019).
Walker, K. A. et al. Large-scale plasma proteomic analysis identifies proteins and pathways associated with dementia risk. Nat. Aging 1, 473–489 (2021).
Lindbohm, J. V. et al. Immune system wide biomarker analyses provide causal evidence for autoimmunity in dementia causing diseases. Nat. Aging 2, 956–972 (2022).
Sipilä, P. N. et al. Hospital-treated infectious diseases and the risk of dementia: a large, multicohort, observational study with a replication cohort. Lancet Infect. Dis. 21, 1557–1567 (2021).
Sun, J. et al. Hospital-treated infections in early-and mid-life and risk of Alzheimer’s disease, Parkinson’s disease, and amyotrophic lateral sclerosis: a nationwide nested case-control study in Sweden. PLoS Med. 19, e1004092 (2022).
Bu, F., Philip, K. & Fancourt, D. Social isolation and loneliness as risk factors for hospital admissions for respiratory disease among older adults. Thorax 75, 597–599 (2020).
Scharf, I. et al. Social isolation causes downregulation of immune and stress response genes and behavioural changes in a social insect. Mol. Ecol. 30, 2378–2389 (2021).
Elovainio, M. et al. Association of social isolation and loneliness with future risk of hospital-treated infections: a prospective multicohort study. Lancet Public Health 8, e109–e118 (2023).
Byers, A. L. & Yaffe, K. Depression and risk of developing dementia. Nat. Rev. Neurol. 7, 323–331 (2011).
Singh-Manoux, A. et al. Trajectories of depressive symptoms before diagnosis of dementia: a 28-year follow-up study. JAMA Psychiatry 74, 712–718 (2017).
Young, S. N. The neurobiology of human social behaviour: an important but neglected topic. J. Psychiatry Neurosci. 33, 391–392 (2008).
Watts, D. J., Dodds, P. S. & Newman, M. E. Identity and search in social networks. Science 296, 1302–1305 (2002).
Leary, M. R., Herbst, K. C. & McCrary, F. Finding pleasure in solitary activities: desire for aloneness or disinterest in social contact? Pers. Individ. Dif. 35, 59–68 (2003).
Chevallier, C., Kohls, G., Troiani, V., Brodkin, E. S. & Schultz, R. T. The social motivation theory of autism. Trends Cogn. Sci. 16, 231–239 (2012).
Livingston, J. D. & Boyd, J. E. Correlates and consequences of internalized stigma for people living with mental illness: a systematic review and meta-analysis. Soc. Sci. Med. 71, 2150–2161 (2010).
Rosen, W. G., Mohs, R. C. & Davis, K. L. A new rating scale for Alzheimer’s disease. Am. J. Psychiatry 141, 1356–1364 (1984).
Pitkala, K. H., Routasalo, P., Kautiainen, H., Sintonen, H. & Tilvis, R. S. Effects of socially stimulating group intervention on lonely, older people’s cognition: a randomized, controlled trial. Am. J. Geriatr. Psychiatry 19, 654–663 (2011).
Mortimer, J. A. et al. Changes in brain volume and cognition in a randomized trial of exercise and social interaction in a community-based sample of non-demented Chinese elders. J. Alzheimer’s Dis. 30, 757–766 (2012).
Park, D. C. et al. The impact of sustained engagement on cognitive function in older adults: the Synapse Project. Psychological Sci. 25, 103–112 (2014).
O’Rourke, H. M., Collins, L. & Sidani, S. Interventions to address social connectedness and loneliness for older adults: a scoping review. BMC Geriatr. 18, 214 (2018).
Cohen-Mansfield, J. & Perach, R. Interventions for alleviating loneliness among older persons: a critical review. Am. J. Health Promot. 29, e109–e125 (2015).
Hafdi, M., Hoevenaar-Blom, M. P. & Richard, E. Multi‐domain interventions for the prevention of dementia and cognitive decline. Cochrane Database Syst. Rev. 11, CD013572 (2021).
Ngandu, T. et al. A 2 year multidomain intervention of diet, exercise, cognitive training, and vascular risk monitoring versus control to prevent cognitive decline in at-risk elderly people (FINGER): a randomised controlled trial. Lancet 385, 2255–2263 (2015).
Lee, K. S. et al. Effects of a multidomain lifestyle modification on cognitive function in older adults: an eighteen-month community-based cluster randomized controlled trial. Psychother. Psychosom. 83, 270–278 (2014).
Rosenberg, A. et al. Multidomain lifestyle intervention benefits a large elderly population at risk for cognitive decline and dementia regardless of baseline characteristics: the FINGER trial. Alzheimer’s Dement. 14, 263–270 (2018).
Park, H. K. et al. South Korean study to prevent cognitive impairment and protect brain health through lifestyle intervention in at-risk elderly people: protocol of a multicenter, randomized controlled feasibility trial. J. Clin. Neurol. 16, 292–303 (2020).
Moon, S. Y. et al. Facility-based and home-based multidomain interventions including cognitive training, exercise, diet, vascular risk management, and motivation for older adults: a randomized controlled feasibility trial. Aging 13, 15898–15916 (2021).
Moon, S. Y. et al. Impact of a multidomain lifestyle intervention on regional spontaneous brain activity. Front. Aging Neurosci. 14, 926077 (2022).
Zülke, A. et al. AgeWell.de—study protocol of a pragmatic multi-center cluster-randomized controlled prevention trial against cognitive decline in older primary care patients. BMC Geriatr. 19, 203 (2019).
Wang, Y. et al. MIND China (China) World‐wide FINGERS network: the first global network of multidomain dementia prevention trials. Alzheimer’s Dement. 16, e046957 (2020).
Baker, L. D. et al. US POINTER (USA) World‐wide FINGERS network: the first global network of multidomain dementia prevention trials. Alzheimer’s Dement. 16, e046951 (2020).
Ingram, J., Hand, C. J. & Maciejewski, G. Social isolation during COVID‐19 lockdown impairs cognitive function. Appl. Cogn. Psychol. 35, 935–947 (2021).
Menze, I., Mueller, P., Mueller, N. G. & Schmicker, M. Age-related cognitive effects of the COVID-19 pandemic restrictions and associated mental health changes in Germans. Sci. Rep. 12, 8172 (2022).
Shen, F., Min, C., Lu, Y. & Chu, Y. The effect of cognition and affect on preventive behaviors during the COVID-19 pandemic: a cross-sectional study in China. BMC Public Health 21, 722 (2021).
Xu, E., Xie, Y. & Al-Aly, Z. Long-term neurologic outcomes of COVID-19. Nat. Med. 28, 2406–2415 (2022).
Karr, J. E., Graham, R. B., Hofer, S. M. & Muniz-Terrera, G. When does cognitive decline begin? A systematic review of change point studies on accelerated decline in cognitive and neurological outcomes preceding mild cognitive impairment, dementia, and death. Psychol. Aging 33, 195–218 (2018).
Yu, J. C., Hlávka, J. P., Joe, E., Richmond, F. J. & Lakdawalla, D. N. Impact of non‐binding FDA guidances on primary endpoint selection in Alzheimer’s disease trials. Alzheimer’s Dement. 8, e12280 (2022).
AGE Platform Europe. The Right to Adequate Housing of Older Persons; Age Platform Europe Contribution (2022).
World Health Organization. Global Age-Friendly Cities: A Guide (World Health Organization, 2007).
Holt-Lunstad, J., Smith, T. B. & Layton, J. B. Social relationships and mortality risk: a meta-analytic review. PLoS Med. 7, e1000316 (2010).
World Health Organization. Optimizing Brain Health Across the Life Course: WHO Position Paper (2022).
National Academies of Sciences, E. & Medicine. Evidence-Based Practice for Public Health Emergency Preparedness and Response (National Academies Press, 2020).
Röhr, S. et al. Social factors and the prevalence of social isolation in a population-based adult cohort. Soc. Psychiatry Psychiatr. Epidemiol 57, 1959–1968 (2021).
Valenzuela, M. J. & Sachdev, P. Brain reserve and dementia: a systematic review. Psychol. Med. 36, 441–454 (2006).
Berkman, L. F., Glass, T., Brissette, I. & Seeman, T. E. From social integration to health: Durkheim in the new millennium. Soc. Sci. Med. 51, 843–857 (2000).
Leedahl, S. N., Sellon, A. & Chapin, R. K. Assessment of multiple constructs of social integration for older adults living in nursing homes. J. Gerontol. Soc. Work 61, 526–548 (2018).
Zavaleta, D., Samuel, K. & Mills, C. Social Isolation: A Conceptual and Measurement Proposal. OPHI Working Papers 67. Report no. 1907194541 (2014).
Perlman, D. & Peplau, L. A. in Preventing the Harmful Consequences of Severe and Persistent Loneliness vol. 13 (eds. L. A. Peplau & S. Goldston) 13–46 (US Government Printing Office, 1984).
Stringhini, S. et al. Socioeconomic status, structural and functional measures of social support, and mortality: The British Whitehall II Cohort Study, 1985–2009. Am. J. Epidemiol. 175, 1275–1283 (2012).
Acknowledgements
This research was funded in whole, or in part, by the Wellcome Trust (222932/Z/21/Z). For the purpose of open access, the author has applied a CC BY public copyright license to any author-accepted manuscript version arising from this submission. A.S. and G.L. are supported by the University College London Hospitals National Institute for Health Research (NIHR) Biomedical Research Centre and funded by the NIHR ENHANCE study (NIHR203670). M.K. was supported by the Wellcome Trust (221854/Z/20/Z), the UK Medical Research Council (MRC; S011676/1, R024227/1), the US National Institute on Aging (R01AG056477) and the Academy of Finland (350426). E.B.L. is supported by research grants from the US National Institute on Aging (UO1AG0006781, RO1AG). S.R. is a Global Atlantic Fellow for Equity in Brain Health and supported by the Global Brain Health Institute. A.S.-M. is supported by grants from the National Institute on Aging, National Institutes of Health (R01AG056477, RF1AG062553). G.L. is supported by NIHR ARC and is an NIHR senior investigator.
Author information
Authors and Affiliations
Contributions
A.S. and G.L. conceived the review. A.S. reviewed the literature and drafted the article. M.K., E.B.L., S.R., K.S. and A.S.-M. contributed to the redrafting of the article and approved the final version for submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Aging thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sommerlad, A., Kivimäki, M., Larson, E.B. et al. Social participation and risk of developing dementia. Nat Aging 3, 532–545 (2023). https://doi.org/10.1038/s43587-023-00387-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s43587-023-00387-0
This article is cited by
-
“May the Force (and Size) Be with You”: Muscle Mass and Function Are Important Risk Factors for Cognitive Decline and Dementia
The Journal of nutrition, health and aging (2023)