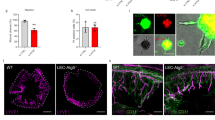
Abstract
Lymphatic vessels (LVs), lined by lymphatic endothelial cells (LECs), are indispensable for life1. However, the role of metabolism in LECs has been incompletely elucidated. In the present study, it is reported that LEC-specific loss of OXCT1, a key enzyme of ketone body oxidation2, reduces LEC proliferation, migration and vessel sprouting in vitro and impairs lymphangiogenesis in development and disease in Prox1ΔOXCT1 mice. Mechanistically, OXCT1 silencing lowers acetyl-CoA levels, tricarboxylic acid cycle metabolite pools, and nucleotide precursor and deoxynucleotide triphosphate levels required for LEC proliferation. Ketone body supplementation to LECs induces the opposite effects. Notably, elevation of lymph ketone body levels by a high-fat, low-carbohydrate ketogenic diet or by administration of the ketone body β-hydroxybutyrate increases lymphangiogenesis after corneal injury and myocardial infarction. Intriguingly, in a mouse model of microsurgical ablation of LVs in the tail, which repeats features of acquired lymphoedema in humans, the ketogenic diet improves LV function and growth, reduces infiltration of anti-lymphangiogenic immune cells and decreases oedema, suggesting a novel dietary therapeutic opportunity.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article (and its Supplementary Information). The raw data that support the findings of this study are available from the corresponding author on reasonable request. Supplementary Figs. 1, 5 and 6 have associated raw data (uncropped blots) in Supplementary Fig. 7. Figure 3 and Supplementary Fig. 4 have associated raw data (Excel files) with metabolite abundances.
References
Alitalo, K. The lymphatic vasculature in disease. Nat. Med. 17, 1371–1380 (2011).
Puchalska, P. & Crawford, P. A. Multi-dimensional roles of ketone bodies in fuel metabolism, signaling, and therapeutics. Cell Metab. 25, 262–284 (2017).
Dayan, J. H., Ly, C. L., Kataru, R. P. & Mehrara, B. J. Lymphedema: pathogenesis and novel therapies. Annu. Rev. Med. 69, 263–276 (2017).
Avraham, T. et al. Th2 differentiation is necessary for soft tissue fibrosis and lymphatic dysfunction resulting from lymphedema. FASEB J. 27, 1114–1126 (2013).
Tabibiazar, R. et al. Inflammatory manifestations of experimental lymphatic insufficiency. PLoS Med. 3, e254 (2006).
Cormier, J. N. et al. Lymphedema beyond breast cancer: a systematic review and meta-analysis of cancer-related secondary lymphedema. Cancer 116, 5138–5149 (2010).
Rockson, S. G. & Rivera, K. K. Estimating the population burden of lymphedema. Ann. N.Y. Acad. Sci. 1131, 147–154 (2008).
Wong, B. W. et al. The role of fatty acid beta-oxidation in lymphangiogenesis. Nature 542, 49–54 (2017).
Yu, P. et al. FGF-dependent metabolic control of vascular development. Nature 545, 224–228 (2017).
Cotter, D. G., d’Avignon, D. A., Wentz, A. E., Weber, M. L. & Crawford, P. A. Obligate role for ketone body oxidation in neonatal metabolic homeostasis. J. Biol. Chem. 286, 6902–6910 (2011).
Bazigou, E. et al. Genes regulating lymphangiogenesis control venous valve formation and maintenance in mice. J. Clin. Invest. 121, 2984–2992 (2011).
Detry, B. et al. Sunitinib inhibits inflammatory corneal lymphangiogenesis. Invest. Ophthalmol. Vis. Sci. 54, 3082–3093 (2013).
Schoors, S. et al. Fatty acid carbon is essential for dNTP synthesis in endothelial cells. Nature 520, 192–197 (2015).
Pinto, A., Bonucci, A., Maggi, E., Corsi, M. & Businaro, R. Anti-oxidant and anti-inflammatory activity of ketogenic diet: new perspectives for neuroprotection in Alzheimer’s disease. Antioxidants 7, 63 (2018).
Yamanashi, T. et al. Beta-hydroxybutyrate, an endogenic NLRP3 inflammasome inhibitor, attenuates stress-induced behavioral and inflammatory responses. Sci. Rep. 7, 7677 (2017).
Goldberg, E. L. et al. β-Hydroxybutyrate deactivates neutrophil NLRP3 inflammasome to relieve gout flares. Cell Rep. 18, 2077–2087 (2017).
Henry, R. A., Kuo, Y. M. & Andrews, A. J. Differences in specificity and selectivity between CBP and p300 acetylation of histone H3 and H3/H4. Biochemistry 52, 5746–5759 (2013).
Xu, Y. M., Du, J. Y. & Lau, A. T. Posttranslational modifications of human histone H3: an update. Proteomics 14, 2047–2060 (2014).
Peters, A. H. et al. Partitioning and plasticity of repressive histone methylation states in mammalian chromatin. Mol. Cell 12, 1577–1589 (2003).
Zhang, X., Wen, H. & Shi, X. Lysine methylation: beyond histones. Acta Biochim. Biophys. Sin. 44, 14–27 (2012).
Carey, B. W., Finley, L. W., Cross, J. R., Allis, C. D. & Thompson, C. B. Intracellular alpha-ketoglutarate maintains the pluripotency of embryonic stem cells. Nature 518, 413–416 (2015).
TeSlaa, T. et al. α-Ketoglutarate accelerates the initial differentiation of primed human pluripotent stem cells. Cell Metab. 24, 485–493 (2016).
Tian, W. et al. Leukotriene B4 antagonism ameliorates experimental lymphedema. Sci. Transl. Med. 9, eaal3920 (2017).
Zampell, J. C. et al. CD4+ cells regulate fibrosis and lymphangiogenesis in response to lymphatic fluid stasis. PLoS ONE 7, e49940 (2012).
Gousopoulos, E., Proulx, S. T., Scholl, J., Uecker, M. & Detmar, M. Prominent lymphatic vessel hyperplasia with progressive dysfunction and distinct immune cell infiltration in lymphedema. Am. J. Pathol. 186, 2193–2203 (2016).
Keith, L., Rowsemitt, C. & Richards, L. G. Lifestyle modification group for lymphedema and obesity results in significant health outcomes. Am. J. Lifestyle Med. 0, 1559827617742108 (2017).
Hu, X., Jiang, Z. & Liu, N. A novel approach for harvesting lymphatic endothelial cells from human foreskin dermis. Lymphat. Res. Biol. 4, 191–198 (2006).
Vandekeere, S. et al. Serine synthesis via PHGDH Is essential for heme production in endothelial cells. Cell Metab. 28, P573–P587 (2018).
Muzumdar, M. D., Tasic, B., Miyamichi, K., Li, L. & Luo, L. A global double-fluorescent Cre reporter mouse. Genesis 45, 593–605 (2007).
Kalucka, J. et al. Quiescent endothelial cells upregulate fatty acid beta-oxidation for vasculoprotection via redox homeostasis. Cell Metab. 28, P881–P894 (2018).
Acknowledgements
We thank P. Crawford and T. Mäkinen for providing Oxct1lox/lox and Prox1-creERT2 mice, respectively. We thank G. Bogaert for providing human foreskins. We also thank S.M. Fendt for discussion and advice. This work was supported by fellowships from LE&RN/FDRS (A.Z.), and supporting grants from IUAP P7/03 (P.C.), Methusalem funding by the Flemish government (P.C.), FWO (G.0598.12, G.0532.10, G.0817.11, G.0834.13, to P.C.), Leducq Transatlantic Network Artemis (P.C.), AXA Research Fund (no. 1465, to P.C.), Foundation against Cancer (P.C.), Fund for Translation Biomedical Research (to P.C.), ERC Advanced Research Grant (EU-ERC269073, to P.C.). We thank A. Van Nuffelen, A. Carton, A. Manderveld, K. Brepoels, K. Peeters, N. Dai, M. Rifaad, M. Parys, I. Betz, C. De Legher, S. Wyns, P.J. Coolen, M. Nijs, P. Vanwesemael, B. Verherstraeten, G. Dubois, E. Van Dyck, A. Acosta Sanchez and D. Verdegem for their technical assistance, and various laboratory members for their feedback and discussions.
Author information
Authors and Affiliations
Contributions
M.G.C., A.Z., J.S., A.-C.K.T., L.-A.T., W.V., A.B., R.M.P., S.V., I.C. and B.G. performed research and/or analysed the data. P.d.Z. managed the collaboration with UZ Leuven, wrote the documentation for the Medical Ethical Committee agreement and the informed consent for HDLEC subjects. M.G.C., A.Z., R.M.P., M.M., G.E., M.D. and P.C. designed experiments. M.G.C., A.Z. and P.C. wrote the paper. P.C. conceptualized and supervised the study. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial or non-financial interests in relation to the work described.
Additional information
Peer review information: Primary Handling Editor: Pooja Jha.
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–7
Supplementary Table 1
Metabolite abundances in control and OXCT1KD HDLECs measured by MS. Raw metabolite abundances are expressed as arbitrary units normalized to micrograms of protein content
Supplementary Table 2
Metabolite abundances in control and OXCT1KD2 HDLECs measured by MS. Raw metabolite abundances are expressed as arbitrary units normalized to micrograms of protein content
Supplementary Table 3
Metabolite abundances in control and supplemented HDLECs measured by MS. Raw metabolite abundances are expressed as arbitrary units normalized to micrograms of protein content
Rights and permissions
About this article
Cite this article
García-Caballero, M., Zecchin, A., Souffreau, J. et al. Role and therapeutic potential of dietary ketone bodies in lymph vessel growth. Nat Metab 1, 666–675 (2019). https://doi.org/10.1038/s42255-019-0087-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s42255-019-0087-y
This article is cited by
-
Ketones and the cardiovascular system
Nature Cardiovascular Research (2023)
-
Metabolic Messengers: ketone bodies
Nature Metabolism (2023)
-
Molecular and metabolic orchestration of the lymphatic vasculature in physiology and pathology
Nature Communications (2023)
-
Effects of the ketogenic diet in mice with hind limb ischemia
Nutrition & Metabolism (2022)
-
Lipid droplet degradation by autophagy connects mitochondria metabolism to Prox1-driven expression of lymphatic genes and lymphangiogenesis
Nature Communications (2022)