Abstract
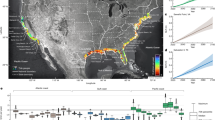
Conservation interventions are central strategies for achieving sustainable development goals given the inextricable dependence of humanity on nature. Current debate centres on whether interventions such as marine protected areas (MPAs) promote co-benefits or trade-offs among multiple goals such as poverty alleviation, food security and protection of marine resources. Resolving this question is hindered by a lack of quantitative impact evaluations of concurrent ecological and social co-benefits of MPAs. Here we use a statistical matching approach to examine whether MPAs are associated with co-benefits or trade-offs between reef fish abundances and measures of human well-being, including income, diet and food security in the Mesoamerican region. We find that highly protected areas (HPAs) with stringent fishing restrictions tend to support high mean abundances and stable or increasing trends in fish abundances compared with unprotected sites and ‘general use zones’ of MPAs. At the same time, indicators of income and food security were elevated in communities near MPAs, especially HPAs, compared with communities far from MPAs. Finally, proximity to MPAs and to reefs with high fish abundance were both positively associated with well-being across space. Together, these results provide quantitative evidence of co-benefits for fish and people associated with MPAs, highlighting the potential value of MPAs in achieving multiple sustainable development goals.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All data used in this study were obtained from open sources listed in Supplementary Tables 16 and 17. Ecological data are available on www.agrra.org, with some years also displayed in healthyreefs.org. Human well-being indicators are freely available from www.dhsprogram.com/data.
Code availability
The R and JAGS code used for analyses are available upon reasonable request to the corresponding author.
References
Transforming Our World: the 2030 Agenda for Sustainable Development (United Nations, 2015).
Hopkins, S. R. et al. How to identify win–win interventions that benefit human health and conservation. Nat. Sustain. 4, 298–304 (2021).
Nilsson, M., Griggs, D. & Visbeck, M. Policy: map the interactions between Sustainable Development Goals. Nature 534, 320–322 (2016).
Singh, G. G. et al. A rapid assessment of co-benefits and trade-offs among Sustainable Development Goals. Mar. Policy 93, 223–231 (2018).
McElwee, P. et al. The impact of interventions in the global land and agri-food sectors on Nature’s Contributions to People and the UN Sustainable Development Goals. Glob. Change Biol. 26, 4691–4721 (2020).
Baylis, K. et al. Mainstreaming impact evaluation in nature conservation. Conserv. Lett. 9, 58–64 (2016).
Gill, D. A. et al. Social synergies, tradeoffs, and equity in marine conservation impacts. Annu. Rev. Environ. Resour. 44, 347–372 (2019).
Sims, K. R. E. & Alix-Garcia, J. M. Parks versus PES: evaluating direct and incentive-based land conservation in Mexico. J. Environ. Econ. Manage. 86, 8–28 (2017).
Ferraro, P. J. et al. Estimating the impacts of conservation on ecosystem services and poverty by integrating modeling and evaluation. Proc. Natl Acad. Sci. USA 112, 7420–7425 (2015).
Global Indicator Framework for the Sustainable Development Goals and Targets of the 2030 Agenda for Sustainable Development (United Nations, 2018).
Protected Planet Live Report (UNEP-WCMC, IUCN and NGS, 2021).
Kunming-Montreal Global biodiversity Framework: Draft Decision Submitted by the President (Convention on Biological Diversity, 2022).
Halpern, B. S. & Warner, R. R. Marine reserves have rapid and lasting effects. Ecol. Lett. 5, 361–366 (2002).
Cinner, J. E. et al. Meeting fisheries, ecosystem function, and biodiversity goals in a human-dominated world. Science 368, 307–311 (2020).
Claudet, J. et al. Marine reserves: size and age do matter. Ecol. Lett. 11, 481–489 (2008).
Edgar, G. J. et al. Global conservation outcomes depend on marine protected areas with five key features. Nature 506, 216–220 (2014).
Gill, D. A. et al. Capacity shortfalls hinder the performance of marine protected areas globally. Nature 543, 665–669 (2017).
Turnbull, J. W., Johnston, E. L. & Clark, G. F. Evaluating the social and ecological effectiveness of partially protected marine areas. Conserv. Biol. 35, 921–932 (2021).
Costello, M. J. & Ballantine, B. Biodiversity conservation should focus on no-take Marine Reserves: 94% of Marine Protected Areas allow fishing. Trends Ecol. Evol. 30, 507–509 (2015).
Pollnac, R. & Seara, T. Factors influencing success of marine protected areas in the Visayas, Philippines as related to increasing protected area coverage. Environ. Manage. 47, 584–592 (2011).
Ban, N. C. et al. Well-being outcomes of marine protected areas. Nat. Sustain. 2, 524–532 (2019).
Mascia, M. B., Claus, C. A. & Naidoo, R. Impacts of marine protected areas on fishing communities. Conserv. Biol. 24, 1424–1429 (2010).
Bennett, N. J. & Dearden, P. From measuring outcomes to providing inputs: governance, management, and local development for more effective marine protected areas. Mar. Policy 50, 96–110 (2014).
Mascia, M. B. & Claus, C. A. A property rights approach to understanding human displacement from protected areas: the case of marine protected areas. Conserv. Biol. 23, 16–23 (2009).
McKinnon, M. C. et al. What are the effects of nature conservation on human well-being? A systematic map of empirical evidence from developing countries. Environ. Evid. 5, 8 (2016).
Conservation Effectiveness. Visualizing the Effectiveness of Conservation Strategies https://www.conservationeffectiveness.org/ (2021).
Kramer, P. A. & Kramer, P. R. Ecoregional Conservation Planning for the Mesoamerican Caribbean Reef (WWF, 2002).
Lang, J. C., Marks, K. W., Kramer, P. A., Kramer, P. R. & Ginsburg, R. N. AGRRA Protocols Version 5.4 (Atlantic and Gulf Rapid Reef Assessment Program, 2010).
Demographic and Health Survey Interviewer’s Manual (ICF, 2020).
Ahmadia, G. N. et al. Integrating impact evaluation in the design and implementation of monitoring marine protected areas. Phil. Trans. R. Soc. B 370, 20140275 (2015).
Babcock, R. C. et al. Decadal trends in marine reserves reveal differential rates of change in direct and indirect effects. Proc. Natl Acad. Sci. USA 107, 18256–18261 (2010).
Reducing Stunting in Children: Equity Considerations for Achieving the Global Nutrition Targets 2025 (World Health Organization, 2018).
Weiss, D. J. et al. A global map of travel time to cities to assess inequalities in accessibility in 2015. Nature 553, 333–336 (2018).
Okwi, P. O. et al. Spatial determinants of poverty in rural Kenya. Proc. Natl Acad. Sci. USA 104, 16769–16774 (2007).
Canty, S. W. J. & Deichmann, J. L. Do small-scale fisheries have the capacity to provide food security to coastal populations? Fish Fish. 23, 708–718 (2022).
Hicks, C. C. et al. Harnessing global fisheries to tackle micronutrient deficiencies. Nature 574, 95–98 (2019).
The State of World Fisheries and Aquaculture 2018: Meeting the Sustainable Development Goals (FAO, 2018).
Hruschka, D. J., Gerkey, D. & Hadley, C. Estimating the absolute wealth of households. Bull. World Health Organ. 93, 483–490 (2015).
Garcia, S. M., Charles, A., Sanders, J. & Westlund, L. in Marine Protected Areas: Interactions with Fishery Livelihoods and Food Security (FAO, 2018).
Canty, S. et al. The hidden value of artisanal fisheries in Honduras. Fish. Manage. Ecol. 26, 249–259 (2019).
Chollett, I., Canty, S. W. J., Box, S. J. & Mumby, P. J. Adapting to the impacts of global change on an artisanal coral reef fishery. Ecol. Econ. 102, 118–125 (2014).
Grorud-Colvert, K. et al. The MPA Guide: a framework to achieve global goals for the ocean. Science 373, eabf0861 (2021).
Naidoo, R. et al. Evaluating the impacts of protected areas on human well-being across the developing world. Sci. Adv. 5, eaav3006 (2019).
Healthy Reefs Initiative. Eco-audit 2021 https://eco-audits.healthyreefs.org/ (2021).
Venter, O. et al. Global terrestrial Human Footprint maps for 1993 and 2009. Sci. Data 3, 160067 (2016).
Ha, N. T. T., Koike, K. & Nhuan, M. T. Improved accuracy of chlorophyll-a concentration estimates from MODIS imagery using a two-band ratio algorithm and geostatistics: as applied to the monitoring of eutrophication processes over Tien Yen Bay (Northern Vietnam). Remote Sens. 6, 421–442 (2014).
Skirving, W. et al. CoralTemp and the Coral Reef Watch Coral Bleaching Heat Stress Product Suite Version 3.1. Remote Sens. 12, 3856 (2020).
ESA. Land Cover CCI Product User Guide Version 2 maps.elie.ucl.ac.be/CCI/viewer/download/ESACCI-LC-Ph2-PUGv2_2.0.pdf (2017).
Muyanga, M. & Jayne, T. S. Effects of rising rural population density on smallholder agriculture in Kenya. Food Policy 48, 98–113 (2014).
Kuempel, C. D., Jones, K. R., Watson, J. E. M. & Possingham, H. P. Quantifying biases in marine-protected-area placement relative to abatable threats. Conserv. Biol. 33, 1350–1359 (2019).
Hallpern, B. S., Lester, S. E. & Kellner, J. B. Spillover from marine reserves and the replenishment of fished stocks. Environ. Conserv. 36, 268–276 (2009).
Stuart, E. A., King, G., Imai, K. & Ho, D. MatchIt: nonparametric preprocessing for parametric causal inference. J. Stat. Softw. 42, 1–28 (2011).
Bürkner, P.-C. brms: an R package for Bayesian multilevel models using Stan. J. Stat. Softw. 80, 1–28 (2017).
Protected Planet. The World Database on Protected Areas (WDPA) www.protectedplanet.net (UNEP-WCMC and IUCN, 2020).
Esri. Oceans Basemap https://www.arcgis.com/home/item.html?id=5ae9e138a17842688b0b79283a4353f6 (2020).
Esri. World Countries https://hub.arcgis.com/maps/esri::world-countries-generalized/about (2020).
Acknowledgements
We thank the many people who collected, processed, collated and made available the data underlying this study, particularly the 74 partner organizations within the Healthy Reefs Initiative (www.healthyreefs.org/partners). Primary funding for reef surveys was provided by Summit Foundation and Oak Foundation. S.W.J.C. and M.M. were supported by Summit Foundation. S.C. thanks the generous support received from Betty and Gordon Moore. A.J.N. was supported by a Smithsonian SI-CI Postdoctoral Fellowship. This is Smithsonian Marine Station contribution number 1191.
Author information
Authors and Affiliations
Contributions
A.J.N., S.W.J.C. and N.B. designed the study. S.W.J.C., C.C., A.V. and M.M. contributed to field data collection on reefs. M.M. oversaw ecological data acquisition, compilation., and funding. A.J.N. collated data, conducted analyses., and led manuscript preparation. A.J.N., S.W.J.C., N.B., C.C., A.V., J.L.D., T.S.A., S.E.B.-A., S.C. and M.M. all contributed to substantive revisions and edits.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Sustainability thanks Elliott Hazen, M. Aaron MacNeil and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Maps of monitored reef sites and Demographic and Health Survey (DHS) clusters in coastal areas.
(A) To examine the effects of marine protected areas (MPAs) on fish abundance, we analysed monitoring data from reef sites (n = 87) along coastal waters of Mexico, Belize, Guatemala and Honduras in open access waters (yellow) and in general use (GUZs; green) and highly protected areas (HPAs; purple) of MPAs. Unmatched sites are shown in grey. (B) To determine whether MPAs affect indicators of human well-being, we analysed survey responses for up to 2,117 individuals from 222 matched DHS clusters along coasts of Guatemala and Honduras that were far from MPAs (>10 km; yellow) or near GUZs (≤10 km; green) or HPAs (purple). (C) To evaluate potential tradeoffs or synergies between human well-being and fish assemblages across space, we analysed fish biomass and indicators of food security and income from nearest neighbour reef sites (triangles) and DHS clusters (squares) along the Atlantic coast of Guatemala and Honduras. Map data are from sources in refs. 28,29,56.
Extended Data Fig. 2 Mean abundance and trends in open access sites and general use (GUZs) and highly protected areas (HPAs) of marine protected areas (MPAs) (n = 4,336 transects sampled at 87 sites).
To assess the effects of fishing restrictions associated with MPAs on assemblages, we used a quasi-experimental approach wherein we first matched survey sites based on site characteristics and then quantified effects of fishing restrictions (GUZs versus HPAs) while controlling for remaining variation in matching variables in Bayesian hierarchical models. Plots show estimated effects of fishing restrictions on means and trends in counts for three commercially important fish families. Error bars represent 95% Bayesian credible intervals, and letters indicate difference among groups that have statistical support – that is, 95% BCIs for contrasts exclude zero.
Extended Data Fig. 3 Interactive effects of fishing restrictions and age of marine protected areas (MPAs) on mean total fish biomass (n = 2,596 transects sampled at 61 sites).
Mean predictions (lines) and 95% credible bands (shaded areas) are from Bayesian hierarchical models fit with interaction terms for MPA age and fishing restrictions while controlling for leftover variation in matching variables. MPA age is centreed and scaled (z-score).
Extended Data Fig. 4 Interactive effects of fishing restrictions and age of marine protected areas (MPAs) on trends in total fish biomass (n = 3,102 transects sampled at 61 sites).
Mean predictions (lines) and 95% credible bands (shaded areas) are from Bayesian hierarchical models fit with interaction terms among MPA age, fishing restrictions, and year while controlling for leftover variation in matching variables. Year is centred and scaled (z-score).
Extended Data Fig. 5 Effects of size of marine protected areas (MPAs) on trends in total fish biomass (n = 3,102 transects sampled at 61 sites).
Mean predictions (lines) and 95% credible bands (shaded areas) are from Bayesian hierarchical models fit with interaction terms for MPA area and year while controlling for leftover variation in matching variables. Year is centred and scaled (z-score).
Extended Data Fig. 6 The probability of stunting (n = 1,880 individuals) is lower near marine protected areas (MPAs) than areas far from MPAs.
Plot shows model coefficients that represent the effects of MPA proximity on probability of stunting, here while controlling for wealth index. To assess the effects of MPA proximity on dimensions of human wellbeing, we used a quasi-experimental approach wherein we first matched survey clusters near and far from MPAs based on site characteristics and then quantified effects of MPA proximity while controlling for remaining variation in matching variables using Bayesian hierarchical models. Error bars represent 80% (thick) and 95% (thin) Bayesian credible intervals (BCI), and asterisks indicate mean effects with statistical support – 95% BCIs exclude zero.
Extended Data Fig. 7 Effects of proximity to general use zones (GUZ) and highly protected areas (HPA) on well-being indicators are qualitatively similar under different choices of threshold distances for defining clusters near and far from these zones of marine protected areas (MPAs).
Model coefficients are from Bayesian hierarchical models fit as in Fig. 3, while defining threshold distance as 5, 10, 15, or 20 km. Coefficients represent mean differences relative to the reference category of clusters far from MPAs. Error bars represent 95% Bayesian credible intervals. Sample sizes varied across datasets when applying different distance thresholds. For the stunting outcome, n for 5 km = 1251, 10 km = 1919, 15 km = 2611, and 20 km = 2843. For the fish diet outcome, n for 5 km = 796, 10 km = 1305, 15 km = 1801, and 20 km = 2012. For the wealth index outcome, n for 5 km = 1382, 10 km = 2117, 15 km = 2866, and 20 km = 3121.
Extended Data Fig. 8 Association between fish biomass and probability of fish consumption (A) and stunting (B) in nearby communities.
Colours indicate communities near general use zones (green) and highly protected areas (purple) of marine protected areas (MPAs) and those far from MPAs (yellow). Heavy grey line represents the mean posterior effect of fish biomass on human well-being indicators, and light grey lines are 100 random samples from the posterior distribution. For the fish diet outcome, n = 56 pairs of reef sites and survey clusters. For the stunting outcome, n = 58 pairs of reef sites and survey clusters.
Supplementary information
Supplementary Information
Supplementary Methods and Tables 1–24.
Rights and permissions
About this article
Cite this article
Nowakowski, A.J., Canty, S.W.J., Bennett, N.J. et al. Co-benefits of marine protected areas for nature and people. Nat Sustain 6, 1210–1218 (2023). https://doi.org/10.1038/s41893-023-01150-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41893-023-01150-4