Abstract
Scarce fossil tetrapod burrows have been recorded in Cretaceous rocks, which is probably linked to the dominant equable climates that existed for most of this period. The occurrence of Cretaceous tetrapod burrows from Patagonia (Chubut Province, Argentina) dated between 118 and 115 million years ago, gives insights into their paleoecology and paleoenvironment. The rocks containing the tetrapod burrows are of pyroclastic origin and represent eolian dunes and ash-fall deposits, some reworked by fluvial currents and others showing soil development. Fossil burrow casts preserved in a paleosol are composed by a ramp with a slightly curved or straight path in plan-view and lacking bifurcation, a rounded termination with no enlargement, showing a reniform cross-section, and are assigned to the ichnospecies Reniformichnus katikatii. The strongly flattened cross-sectional shape of the burrow casts and comparison with modern lizard burrows suggest that the producers were lepidosaurs (body mass = 50–323 g). Among Cretaceous fossorial lepidosaurs from Patagonia, the best candidate is an eilenodontine sphenodontian. Sphenodontians burrowed in the fossil soils where also arthropods, earthworms and shrubby plants thrived. The rare occurrence of tetrapod burrows in Cretaceous rocks is linked to stressing conditions related to frequent arrival of volcanic ash and a semiarid seasonal climate.
Similar content being viewed by others
Introduction
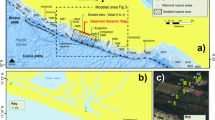
Tetrapods excavate for different purposes, including escaping of dehydration and extreme surface temperatures (thermoregulation), for food storage, breeding, hibernation, to avoid predation and/or to escape fires1,2. Although burrowing in other ecoregions occur, an underground cool and humid shelter, with stable temperature is especially critical to survive in arid and semi-arid zones3. The scarce record of Cretaceous tetrapod burrows4 can be a reflection of the dominant equable climates that existed for most of this period5,6. The only Early Cretaceous records of tetrapod burrows are possible mammal or reptile burrows from the Hauterivian of Korea7 and putative dinosaur burrows from the Albian of Australia8. Published records of Late Cretaceous tetrapod burrows are currently restricted to USA and Brazil, including the first and well-documented ornithopod dinosaur den containing the remains of its producer from the Cenomanian of USA9,10. Late Cretaceous examples are purported mammal burrows from the Campanian of USA11,12 and two examples from the Maastrichtian of Brazil, one assigned to freshwater turtles13 and the remaining to a notosuchian producer14. In this context, the finding of Early Cretaceous burrow casts from Patagonia give insights on the paleoecology and paleoenvironment where these structures were excavated, in a period with scarce evidence for tetrapod burrowing. The Los Chivos Hill area is located in the north-western part of Chubut Province, Patagonia, Argentina (Fig. 1b) and contains exposures of the Aptian Puesto La Paloma Member of the Cerro Barcino Formation from the Jurassic-Paleogene Somuncurá-Cañadón Asfalto basin15,16 (Fig. S1). At the study area, the Puesto La Paloma Member comprises a ~ 31 m thick sub-horizontal succession (Fig. 1a,c,d) mostly composed of sheet-like tuffaceous sandstones interbedded with scarce tuff, mudstone and breccia17,18. It records a pyroclastic-rich non-channelized fluvial environment associated with eolian dunes, ash-fall strata and development of paleosols17,18. According to radiometric dates from tuffaceous strata (206Pb/238U method on zircon), deposition of the Puesto La Paloma Member is largely restricted to the Aptian stage. In particular, the studied tetrapod burrows are bracketed by two ages: 118.497 ± 0.063 Ma and 115.508 ± 0.039 Ma18 (Fig. S1). The local sedimentary sequence includes well-sorted cross-bedded tuffaceous sandstone interpreted as transverse eolian dunes developed by dominant winds blowing from the northwest (Fig. S2, Table S1). The rest of the sequence represent ash-fall deposits from a distant western volcanic source, which were reworked by fluvial currents after rains and colonized by plants, with the consequent development of soils (Table S1). Semiarid and seasonal climatic conditions prevailed during deposition of the analyzed sequence. This is inferred from the coexistence of deposits of unconfined streams, alkaline carbonate lakes, and eolian dunes, as well as from poorly developed paleosols having indications of waterlogging and calcification18,19,20. Mean annual precipitation obtained using geochemical data from paleosols yielded estimates of ~ 200–700 mm/year20. The Los Chivos paleosol is the topmost and better developed soil were tetrapods burrowed (Fig. 1c).
Location, stratigraphic section and exposure of the Puesto La Paloma Member of the Cerro Barcino Formation. (a) General stratigraphic section of the Puesto La Paloma Member at the study area, showing the dated level18 and the Los Chivos paleosol (LCP) containing the tetrapod fossil burrows. (b) Location map. (c,d) Outcrop view of the intermediate and lowermost section of the Puesto La Paloma Member at the study locality. The arrow in (c) indicates the Los Chivos paleosol. Abbreviations in c and d refer to facies associations. DI: dry interdune. WI: wet interdune. ED: eolian dune. See Table S1 and Fig. S2.
Results and discussion
Tetrapod burrow casts
Burrow casts are composed by a ramp (inclined tunnel) with a slightly curved or straight path in plan-view and lacking bifurcation, a rounded termination with no enlargement, and a reniform cross-section (Figs. 2a–c, 3a–c). These morphological attributes allow assignation to the ichnospecies Reniformichnus katikatii Krummeck and Bordy21 (Table S2 discuss the ichnotaxonomic assignation). Ramp inclination is higher in the proximal end (up to 20°) and tend to subhorizontal at the distal end. Burrow casts occur with apparent orientation and are locally abundant (Fig. 2d,e). Average burrow horizontal diameter is 63.34 ± 2.07 mm, average burrow vertical diameter is 34.26 ± 2.25 mm, and the maximum preserved length is 512 mm (Table 1). Burrow cross-section is consistently elliptical flattened with an average vertical diameter/horizontal diameter ratio of 0.53 ± 0.02. The roof of burrow fills is convex upward and the bottom is commonly bilobed, although some specimens are also convex downward (Fig. 4a–g). In bilobed specimens, the height of the nearly central furrow is up to 20% of the vertical diameter.
Field occurrence of tetrapod burrow casts. (a,b) Plan view of curved tetrapod burrow casts. (c) Vertical exposure of upper part of the Los Chivos paleosol with two tetrapod burrow casts (arrowed). (d,e) Plan view of fallen block of top of Los Chivos paleosol with several burrow casts (red) and rhizoliths (gray) and interpretative diagram. The inset in (b) is a rose diagram of the dip azimuth of the burrow casts in the block.
Cross-section shape of R. katikatii and inferred body mass of the producer. (a) MPEF-IC 4310. (b) Fs#14. (c) MPEF-IC 4314. (d) MPEF-IC 4315. (e) Fs#11. (f) MPEF-IC 4318. The arrow points to an invertebrate burrow. (g) MPEF-IC 4312. (h) Histogram of inferred body mass obtained using the formula by Wu et al.32.
Most burrow casts display groups of three parallel millimeter-thick ridges that are interpreted as claw trace sets (Figs. 3a–c, 5a–d). Claw traces are sharper and better developed in the roof and lateral side of the burrow casts, where the average width of the sets of claw traces is 8.89 ± 0.55 mm. The pattern of claw traces in the roof is typically arcuate, with the sets of traces starting at the midline and converging in the lateral surface of the burrow cast (Fig. 5a,b). In the bottom, the sets of claw traces average 8.48 ± 0.47 mm in width and compose a low angle to chevron pattern (Fig. 5f,h). Individual claw traces display a consistent width in roof and bottom averaging 1.34 ± 0.05 mm (Table 1). The contrasting claw trace pattern in roof and bottom are tentatively linked to scratch-digging with the forelimbs (arcuate traces in roof) and pushing back loose sediment with the hindlimbs (chevron traces in bottom), respectively. Sparse subcircular and smaller cylindrical burrows having a diameter ranging from 4.4 to 7.5 mm cross-cut the tetrapod burrows (Fig. 5a–c,f). The fill of tetrapod burrows is massive cemented volcanic ash with abundant subspherical concentric structures interpreted as ash aggregates (accretionary lapilli) and vesiculated ash (pumice) clasts, both with concretionary growth (Figs. 4a–h, 6a,b). Computed tomography images of R. katikatii allow to identify vertical millimeter-thick burrows with rounded and enlarged terminus that postdate tetrapod burrow abandonment and filling by sediments (Fig. 6c,d). Presence of a bilobed bottom is a feature typical of some fossil and extant tetrapod burrows21,22,23,24,25,26,27. This feature was interpreted as reflecting protracted occupation of a burrow and repeated passage of the occupant, thus producing a differential compaction of the sides of burrow bottom27,28.
Surface ornamentation of R. katikatii. (a–c) Plan view, interpretative diagram and oblique lateral view of MPEF-IC 4310. White arrows and grey areas (in b) indicate invertebrate burrows. Bracket in (c) indicates a set of claw traces. (d,e) Claw traces on roof and smooth bilobed bottom in MPEF-IC 4314. (f) Nearly flat bottom with subtle claw traces (white arrow) and invertebrate burrow (yellow arrow) in MPEF-IC 4318. (g) Smooth bilobed bottom in MPEF-IC 4317. (h) Bilobed bottom with claw traces forming a chevron pattern (arrows) in MPEF-IC 4315.
Massive fill of R. katikatii. (a,b) Polished cross-section of burrow fill and interpretative drawing. Note accretionary lapilli (white arrow) and pumice clasts (yellow arrow) both surrounded by concretionary cementation. (c) Tridimensional model from CT of MPEF-IC 4312 (oblique lateral view) with transparent outline and orange bodies interpreted as denser parts product of cementation. The arrow indicates the subvertical burrow of (d). (d) Detail of quasi-spiral submillimetric burrow with a rounded and enlarged end.
The burrows were left open by the occupant, as suggested by the massive fill, and passively received input of volcanic ash, either by settling from ash clouds or reworked by currents. The former origin is favored by the presence of accretionary lapilli, that is typically formed subaerially and commonly cannot be reworked29. The average ratio between the average width of the sets of claw traces (a proxy for the autopodium size as each trace can be linked to the middle three digits) and the horizontal diameter (an indication of the producer size) is 0.15 (Table 1), which is indicative of a tetrapod origin30. The presence of sets of claw traces is suggestive of scratch-digging mechanism, which is employed by a large variety of limbed tetrapods31. Using the allometric relationship with the cross-sectional area of burrow casts32, the body mass of the producer was estimated as ranging between 50 and 323 g (Fig. 4h).
Sphenodontians as burrow producers
The burrow casts exhibit a diameter, cross-section shape, overall architecture, including the low vertical diameter/horizontal diameter ratio, that are indicative of a producer with a sprawling posture as typical of lepidosaurs33. Extant Liolaemus sp. (Squamata: Liolaemidae) burrows from central Argentina (Fig. S3) share with the studied tetrapod fossil burrows overall architecture, cross sectional shape (average vertical diameter/horizontal diameter ratio = 0.56 ± 0.01), bilobed bottom and surface ornamentation. Casted Liolaemus burrows consists of a simple ramp with a constant horizontal and vertical diameter, having a rounded end without enlargement, with an “L” shaped outline in plan view (Fig. 7a,b,e,f). The bottom of Liolaemus casts is bilobed (Fig. 7c,d) as observed in the fossil example, probably indicating a protracted usage of the burrow. The surface ornamentation displays an arcuate pattern (Fig. 7g–j) that is strongly similar to that exhibited by the fossil tetrapod casts (Fig. 5a,b), suggesting a similar excavation mechanism.
Modern Liolaemus sp. plaster burrow casts (a,c,e,i, and j belong to GHUNLPam 29090; and b,d,f–h to GHUNLPam 29091). (a,b) Side view. The dashed line marks the terrain surface (see also Fig. S3). Arrows indicate the excess plaster poured in the surface to mark the ground surface. (c,d) Bilobed bottom, note that the distal end is smooth. (e,f) Plan view, note distal curvature. Arrows indicate the excess plaster. (g–j) Surface ornamentation and interpretative drawing of the distal part of burrow casts.
Considering the Cretaceous tetrapod fossil record of South America, the candidate for producer of R. katikatii are lepidosaurs or, less likely, basal mammals. Some basal mammals displayed a sprawling posture, in some cases related to arboreal habits34,35, which can be thus discarded. Most of the remains of Cretaceous mammals in South America correspond to cranial fragments and isolated teeth36,37,38, for which is difficult to estimate their body mass (Table S3). A single well-documented mammal taxon is Vincelestes nuequenianus (Mammalia, Cladotheria) from Barremian-Aptian deposits of Patagonia, but it can be discarded as producer because of larger size (body mass ranging from 619 to 1228 g) and an inferred scansorial-arboreal habit39,40. The Cenomanian taxon Cronopio dentiacutus (Mammalia, Dryolestidae) is diminutive (skull width about 10 mm) and is only known from an incomplete skull41. A mammalian digger for R. katikatii can be dismissed considering the cross-sectional shape of the burrows (height/width ~ 0.5) and that the Cretaceous fossil record from Patagonia lacks a candidate with fossorial habits that match the inferred body mass of the producer.
Cretaceous lepidosaurs from South America belong mostly to Squamata in the north and southeast of Brazil, while Sphenodontia is restricted to more southern latitudes, in the south of Brazil and Argentina42. The record of Mesozoic Squamata in Patagonia is incomplete and discontinuous, restricted to the Late Cretaceous and mostly composed by unidentified Iguania and Scincomorpha (Table S3). Snakes are not considered potential producers because of their large size and lack of appendages or only presence of vestigial posterior appendages43,44,45,46. Squamata records are from the Cenomanian–Turonian to early Campanian and belong to fragmentary cranial remains from diminutive individuals (Table S3), which are considered much smaller than the presumed producer. Sphenodontians are known in Patagonia from the Early Cretaceous to the Paleocene (Table S3), most belong to eilenodontines47,48,49 that were gregarious and herbivorous reptiles that lived in burrows excavated using their powerful beaks and hoofed claws48, and there is a single small-sized sphenodontine that likely preyed on insects and small vertebrates50. There are three potential sphenodontian candidates for the producer of the burrow casts. The only Early Cretaceous record is the eilenodontine Kaikaifilusaurus (Priosphenodon) minimus that was recovered from the same formation than the burrow casts48 and the skull length of a subadult specimen is 20 mm, with a inferred body mass below the range for the fossil burrows (Table S3). Kaikaifilusaurus minimus is characterized by a flat skull, sharp beak that is slightly procumbent in its anterior portion improving both the excavation and cutting of vegetation, along with the well-developed adductor musculature, tall and robust jaw, wide teeth with low crowns. These features suggest fossorial habits48, making K. minimus a good candidate for producer of the burrow casts. Another candidate of adequate size is the medium sized sphenodontine Tika giacchinoi from Cenomanian beds of northern Patagonia, however, there is no positive evidence that might suggest a burrowing behavior50. The third candidate is the eilenodontine Patagosphenos watuku recovered from Turonian levels that presents a similar bone microstructure to that of the living Sphenodon with medullar cavity reduction of long bones49, this feature is interpreted as an adaptation for fossoriality in living mammals51. Although the recovered remains are fragmentary, the small incomplete dentary (Table S3) indicates a very small individual to be considered the producer of these burrows. The remaining Late Cretaceous records of sphenodontians from Patagonia are considerably larger or younger than the burrow-bearing unit (Table S3). To summarize, a lepidosaurian origin for the producer of R. katikatii is indicated by the cross-sectional shape and by comparison with modern lepidosaur burrows (Liolaemus sp.). Patagonian Cretaceous Squamata are much smaller than the inferred body mass of the producer and are thus not considered a likely tracemaker. Among the Cretaceous sphenodontians from Patagonia, the most likely burrow digger is K. minimus (recovered from the same lithostratigraphic unit that the fossil burrows) because of fossorial habits and similar body mass.
The sphenodontians, very diverse and widely distributed during the Mesozoic, nowadays only live in New Zealand, constituting a relict population. Sphenodon lives in burrows of various morphologies, from a simple ramp with a terminal nest to complex systems with several entrances and a nest52,53. Simple Sphenodon ramps are 110 mm to 500 mm long, have an average height of 45 mm and average width of 73 mm53. The ratio between height and width of the cross-section of modern Sphenodon burrows is 0.61, indicating an elliptical flattened cross section. The size and overall morphology of Sphenodon burrows is similar to those described above for Early Cretaceous burrow casts (Table 1).
Considering the known Cretaceous fossorial lepidosaurs from Patagonia, the inferred size of the of the tetrapod remains (although most are incomplete) and its close relationship with the size of its burrows54, in addition to the similarity with the tuatara burrows morphometry, it is suggested that the most likely producers of the burrows described here are the sphenodontians, which were abundant in Patagonia during the Cretaceous (Table S3). In particular, an adult K. minimus could be the best candidate considering that was recovered from the same formation and the skull features suggesting a fossorial habit. These are the first documented fossil sphenodontian burrows.
Los Chivos paleosol and ichnological expression of soil biota
The Los Chivos paleosol contains three horizons having transitional boundaries (Fig. S4). The upper horizon is a 0.75 m thick, light pinkish white massive fine-grained tuff with millimetric Fe–Mn nodules. Sphenodontian burrows occur in this horizon, along with vertical meniscate (Taenidium barretti) or massive (Skolithos linearis) invertebrate burrows and rhizoliths, which are locally abundant. The middle horizon is a 0.60 m thick fine-grained tuff with a coarse granular structure, pinkish white to white in color, showing diffuse parallel lamination. The lower horizon is a 110 cm thick, light grey massive very fine-grained tuff also showing coarse structure (Fig. S4). In addition to the tetrapod burrows, the soil biota is reflected in biogenic structures attributed to earthworms, unidentified arthropods and sparse shrubby plants. Biogenic structures produced by earthworms includes subvertical cylindrical burrows (averaging 7 mm wide) and globose swellings with pelletal filling (ichnospecies Edaphichnium lumbricatum) (Fig. 8a–c) occasionally associated with meniscate burrows (ichnospecies Taenidium barretti) (Fig. 8e,f). Fecal pellets are yellowish and rounded to elliptical, with an average diameter of 0.87 ± 0.05 mm. Edaphichnium lumbricatum has been reported typically in the Cenozoic55,56,57,58, although also occur in Late Jurassic and Late Cretaceous paleosols59,60,61. Arthropod domiciles are represented by subvertical burrows with massive fill and rounded end (ichnospecies Skolithos linearis) that occur profusely in the uppermost part of the Los Chivos paleosol, locally with high density (up to 290 burrows/m2) (Fig. 8d). These structures average 8.39 ± 0.22 mm in diameter and can reach 0.10 m in length. Skolithos linearis were likely produced by insects or arachnids62,63,64. Evidence about the plant community that thrived in this paleosol is provided by root-generated structures or rhizoliths. Identified rhizoliths are mostly siliceous rhizocretions and ferruginous root casts65. Rhizocretions are common and display a concentric internal structure, downward branching and consequent reduction in diameter (Fig. 8g–j). Maximum preserved length is 0.17 m and average diameter is 14.2 ± 1.5 mm (n = 8). Ferruginous rhizoliths are vertical, up to 0.4 m long, with horizontal branching and a central, 20 mm wide, roughly cylindrical, internal brown root cast and a 30–40 mm wide light brown external zone (Fig. 8k). The central tubular zone also displays submillimetric root traces. The size of root structures suggests a sparse shrubby vegetation by comparison with modern analogues66,67.
Trace fossils associated with R. katikatii. (a–c) Edaphichnium lumbricatum composing subvertical burrows (a,b) and a swelling in (c) (black arrows). Note bifurcation (white arrow in a) and meniscate fill (white arrow in b). (d) Skolithos linearis (black arrows) with rounded end (white arrow). (e,f) Taenidium barretti (margin of burrow arrowed). (g–j) Siliceous rhizocretions (black arrows). Note secondary bifurcation (white arrows in g and j) and concentric rings (in h and i). (g) and (h) are plan views, (i) is a polished section and (j) is a subvertical exposure. (k) Subvertical ferruginous rhizolith, including root cast (white arrow) and lighter halo (black arrows).
The development of the Los Chivos paleosol occurred under a semiarid and seasonal climate, in a flat area between eolian dunes (interdune) where arrived frequent ash clouds from distant volcanoes, which settled subaerially and were reworked by wind and rainwater (Table S1). A period of prolonged stability (at least 2000–3000 years68) with minimum or no arrival of new ash clouds permitted plant rooting and soil development with subsequent establishment of a soil community. The weak alteration degree of the tuffaceous parent material, the moderate distinction of horizons, scarce microscopic features for soil development, and the preservation of the original bedding are characteristics of a moderate to weakly developed paleosol. Consequently, it is best compared with modern andisols or, less probably, andic entisols. The presence of calcite and Fe–Mn oxide coatings suggest a seasonal climate69. The frequent arrival of volcanic ash produced barren and xeric landscapes70 that probably enhanced the semiarid and seasonal climatic conditions inferred from sedimentary facies and paleosol geochemistry. Although tetrapod burrows are commonly multi-purpose structures, R. katikatii from the Early Cretaceous of Patagonia was primarily used for shelter to ameliorate seasonal climatic variations and to keep uniform temperature and humidity. We cannot discard the use for breeding and to avoid predation, but there is no evidence supporting food storage, hibernation, or escape from fires.
Methods
Collected fossil specimens are housed at the Ichnology Collection, Museo Paleontológico Egidio Feruglio (Trelew, Chubut, Argentina) under the acronym MPEF-IC. Plaster burrow casts of extant Squamata are kept at the Paleontological Collection of the Facultad de Ciencias Exactas y Naturales, Universidad Nacional de La Pampa, under the acronym GHUNLPam. Computed tomography (CT) scans of selected burrow casts were carried out at the FAERAC Foundation (Santa Rosa, La Pampa, Argentina) with a medical tomograph Toshiba Aquilon One 320 (MPEF-IC 4310, 4312) and Siemens SOMATOM go.Now (MPEF-IC 4311, 4318). CT scans generate slices in sagittal, coronal and axial views where each type of view has its own spacing ranging from 0.112 to 0.300 mm. The 3D Slicer software71 (https://www.slicer.org) was used for the analysis, processing and three-dimensional visualization of the CT scans. Tridimensional photogrammetric models of selected burrow casts were generated based on photographs taken with a Nikon Coolpix L830 camera and processed in the software Agisoft Metashape Pro™. The resulting models were exported in OBJ files to Adobe Photoshop CC™ and converted to U3D files (a standard format for 3D), to compose a PDF file for easier visualization.
Cross-sectional area of fossil burrow casts (Ab, cm2) was estimated using scaled photographs of the collected and field material with the software ImageJ (https://imagej.nih.gov). This value was then employed to estimate the body mass (Mb, g) of the producer using the allometric relationship32 Ab = 0.46 Mb0.74. Body mass of fossil Sphenodontia was estimated using the relationship between head length (HL, mm) and body mass for extant specimens of Sphenodon punctatus72. The data is herein fitted by equation Mb = 3.38990.081HL (R2 = 0.9106, n = 209).
Micromorphological descriptions of paleosol was conducted with a Nikon Eclipse E400 POL petrographic microscope following standard procedures73,74,75. Burrow cast measurements are expressed as average values ± standard error and indicating the number of measurements.
Data availability
The datasets generated and/or analyzed during the current study are available in the Supplementary Material and Figshare.com repository. CT and photogrammetric tridimensional model of specimen MPEF-IC 4310 https://figshare.com/s/ee80e2be44cd2148209c, photogrammetric tridimensional model of specimen MPEF-IC 4311 https://figshare.com/s/00407b3cdef2e38eadb9; photogrammetric tridimensional model of specimen MPEF-IC 4312 https://figshare.com/s/4c9495e49143371181eb.
References
Kinlaw, A. A review of burrowing by semi-fossorial vertebrates in arid environments. J. Arid Environ. 41, 127–145 (1999).
Reichman, O. J. & Smith, S. C. Burrows and burrowing behavior by mammals. Curr. Mammal. 2, 197–244 (1990).
Kinlaw, A. Burrows of semi-fossorial vertebrates in upland communities of central Florida: their architecture, dispersion and ecological consequences PhD thesis, University of Florida, (2006).
Cardonatto, M. C. & Melchor, R. N. Large mammal burrows in late Miocene calcic paleosols from central Argentina: paleoenvironment, taphonomy and producers. PeerJ 6, e4787. https://doi.org/10.7717/peerj.4787 (2018).
Barron, E. J. A warm, equable Cretaceous: The nature of the problem. Earth Sci. Rev. 19, 305–338. https://doi.org/10.1016/0012-8252(83)90001-6 (1983).
Huber, B. T., MacLeod, K. G., Watkins, D. K. & Coffin, M. F. The rise and fall of the Cretaceous Hot Greenhouse climate. Glob. Planet. Change 167, 1–23. https://doi.org/10.1016/j.gloplacha.2018.04.004 (2018).
Paik, I. S., Kim, H. J. & Lee, H. Unique burrows in the Cretaceous Hasandong Formation, Hadong, Gyeongsangnam-do, Korea: Occurrences, origin, and paleoecological implications. J. Geol. Soc. Korea, 141–155 (2015).
Martin, A. J. Dinosaur burrows in the Otway Group (Albian) of Victoria, Australia, and their relation to Cretaceous polar environments. Cretac. Res. 30, 1223–1237. https://doi.org/10.1016/j.cretres.2009.06.003 (2009).
Varricchio, D. J., Martin, A. J. & Katsura, Y. First trace and body fossil evidence of a burrowing, denning dinosaur. Proc. R. Soc. B Biol. Sci. 274, 1361–1368 (2007).
Krumenacker, L. J., Varricchio, D. J., Wilson, J. P., Martin, A. & Ferguson, A. Taphonomy of and new burrows from Oryctodromeus cubicularis, a burrowing neornithischian dinosaur, from the mid-Cretaceous (Albian-Cenomanian) of Idaho and Montana U.S.A. Palaeogeogr. Palaeocl. 530, 300–311. https://doi.org/10.1016/j.palaeo.2019.05.047 (2019).
Martin, A. J. & Varricchio, D. J. Paleoecological utility of insect trace fossils in dinosaur nesting sites of the Two Medicine Formation (Campanian), Choteau, Montana. Hist. Biol. 23, 15–25 (2011).
Simpson, E. L. et al. Predatory digging behavior by dinosaurs. Geology 38, 699–702. https://doi.org/10.1130/G31019.1 (2010).
Silva, G. T. G., Nascimento, D. L., Batezelli, A., Ladeira, F. S. B. & Silva, M. L. Cretaceous (Maastrichtian) chelonian burrows preserved in floodplain deposits in the Bauru Basin of Brazil: Evidence for the fossorial origin of turtle shells. Palaeogeogr. Palaeoclimatol. Palaeoecol. 596, 110994. https://doi.org/10.1016/j.palaeo.2022.110994 (2022).
Martinelli, A. G. et al. Palaeoecological implications of an Upper Cretaceous tetrapod burrow (Bauru Basin; Peirópolis, Minas Gerais, Brazil). Palaeogeogr. Palaeoclimatol. Palaeoecol. 528, 147–159. https://doi.org/10.1016/j.palaeo.2019.05.015 (2019).
Figari, E. G., Scasso, R. A., Cúneo, R. N. & Escapa, I. Estratigrafía y evolución geológica de la cuenca de Cañadón Asfalto, provincia del Chubut, Argentina, Latin American. J. Sedimentol. Basin Anal. 22, 135–169 (2015).
Allard, J. O. et al. Geología y Recursos Naturales de la Provincia del Chubut. Relatorio del 21° Congreso Geológico Argentino (ed Giacosa, R. E.) Ch. B.5., 187–265 (Asociación Geológica Argentina, 2022).
Villegas, P. M., Visconti, G. & Umazano, A. M. 14° Reunión Argentina de Sedimentología 295–296 (Asociación Argentina de Sedimentología, 2014).
Krause, J. M. et al. High-resolution chronostratigraphy of the Cerro Barcino Formation (Patagonia): Paleobiologic implications for the mid-Cretaceous dinosaur-rich fauna of South America. Gondwana Res. 80, 33–49. https://doi.org/10.1016/j.gr.2019.10.005 (2020).
Umazano, A. M. et al. Changing fluvial styles in volcaniclastic successions: A Cretaceous example from the Cerro Barcino Formation, Patagonia. J. S. Am. Earth Sci. 77, 185–205. https://doi.org/10.1016/j.jsames.2017.05.005 (2017).
Krause, J. M., Umazano, A. M., Bellosi, E. S. & White, T. S. XIV Reunión Argentina de Sedimentología 146–147 (Asociación Argentina de Sedimentología, 2014).
Krummeck, W. D. & Bordy, E. M. Reniformichnus katikatii (new ichnogenus and ichnospecies): Continental vertebrate burrows from the Lower Triassic, main Karoo Basin, South Africa. Ichnos 25, 138–149. https://doi.org/10.1080/10420940.2017.1292909 (2018).
Damiani, R., Modesto, S., Yates, A. & Neveling, J. Earliest evidence of cynodont burrowing. Proc. R. Soc. Lond. B Biol. Sci. 270, 1747–1751 (2003).
Sidor, C. A., Miller, M. F. & Isbell, J. L. Tetrapod burrows from the Triassic of Antarctica. J. Vertebr. Paleontol. 28, 277–284 (2008).
Xing, L. et al. Middle Jurassic tetrapod burrows preserved in association with the large sauropod Omeisaurus jiaoi from the Sichuan Basin, China. Hist. Biol. 29, 931–936. https://doi.org/10.1080/08912963.2016.1263627 (2017).
Bordy, E. M., Sztanó, O., Rampersadh, A., Almond, J. & Choiniere, J. N. Vertebrate scratch traces from the Middle Triassic Burgersdorp Formation of the main Karoo Basin, South Africa: Sedimentological and ichnological assessment. J. Afr. Earth Sci. https://doi.org/10.1016/j.jafrearsci.2019.103594 (2019).
Smith, R. M. H., Angielczyk, K. D., Benoit, J. & Fernandez, V. Neonate aggregation in the Permian dicynodont Diictodon (Therapsida, Anomodontia): Evidence for a reproductive function for burrows?. Palaeogeogr. Palaeoclimatol. Palaeoecol. 569, 110311. https://doi.org/10.1016/j.palaeo.2021.110311 (2021).
Cardonatto, M. C. & Melchor, R. N. Environmental influence on burrow system features of a colonial and fossorial rodent. Implications for interpreting fossil tetrapod burrows. Palaios 36, 225–245. https://doi.org/10.2110/palo.2020.065 (2021).
Cardonatto, M. C. & Melchor, R. N. Recognition of fossil nebkha deposits: Clues from neoichnology and sedimentology. Palaios 35, 277–291. https://doi.org/10.2110/palo.2020.024 (2020).
Schumacher, R. & Schmincke, H.-U. Internal structure and occurrence of accretionary lapilli—a case study at Laacher See Volcano. Bull. Volcanol. 53, 612–634. https://doi.org/10.1007/BF00493689 (1991).
Melchor, R. N. et al. Large striated burrows from fluvial deposits of the Neogene Vinchina Formation, La Rioja, Argentina: A crab origin suggested by neoichnology and sedimentology. Palaeogeogr. Palaeoclimatol. Palaeoecol. 291, 400–418 (2010).
Hildebrand, M. Functional Vertebrate Morphology (eds Hildebrand, M. et al.) Ch. 6, 89–109 (Harvard University Press, 1985).
Wu, N. C., Alton, L. A., Clemente, C. J., Kearney, M. R. & White, C. R. Morphology and burrowing energetics of semi-fossorial skinks (Liopholis spp). J. Exp. Biol. 218, 2416–2426. https://doi.org/10.1242/jeb.113803 (2015).
Bakker, R. T. Dinosaur physiology and the origin of mammals. Evolution 25, 636–658. https://doi.org/10.2307/2406945 (1971).
Chen, M. & Luo, Z.-X. Postcranial skeleton of the Cretaceous mammal Akidolestes cifellii and its locomotor adaptations. J. Mamm. Evol. 20, 159–189. https://doi.org/10.1007/s10914-012-9199-9 (2013).
Chen, M., Luo, Z.-X. & Wilson, G. P. The postcranial skeleton of Yanoconodon allini from the Early Cretaceous of Hebei, China, and its implications for locomotor adaptation in eutriconodontan mammals. J. Vertebr. Paleontol. 37, e1315425. https://doi.org/10.1080/02724634.2017.1315425 (2017).
Bonaparte, J. F. & Migale, L. Protomamíferos mesozoicos de América del Sur (Fundación de Historia Natural Félix de Azara, 2015).
Rougier, G. W., Martinelli, A. & Forasiepi, A. Mesozoic Mammals from South America and Their Forerunners (Springer Nature Switzerland, 2021).
Forasiepi, A. M., Coria, R. A., Hurum, J. & Currie, P. J. First Dryolestoid (Mammalia, Dryolestoidea, Meridiolestida) from the Coniacian of Patagonia and new evidence on their early radiation in South America. Ameghiniana 49, 497–504 (2012).
Rougier, G. W. Vincelestes neuquenianus Bonaparte (Mammalia, Theria), un primitivo mamífero del Cretácico Inferior de la Cuenca Neuquina PhD thesis, Universidad de Buenos Aires, (1993).
Macrini, T. E., Rougier, G. W. & Rowe, T. Description of a cranial endocast from the fossil mammal Vincelestes neuquenianus (Theriiformes) and its relevance to the evolution of endocranial characters in therians. Anat. Rec. 290, 875–892. https://doi.org/10.1002/ar.20551 (2007).
Rougier, G. W., Apesteguía, S. & Gaetano, L. C. Highly specialized mammalian skulls from the Late Cretaceous of South America. Nature 479, 98–102. https://doi.org/10.1038/nature10591 (2011).
Simões, T. R., Caldwell, M. W., Weinschütz, L. C., Wilner, E. & Kellner, A. W. A. Mesozoic lizards from Brazil and their role in early squamate evolution in South America. J. Herpetol. 51, 307–315. https://doi.org/10.1670/16-007 (2017).
Albino, A. & Brizuela, S. Avances en el conocimiento de los reptiles escamosos fósiles continentales de América del Sur. Publicación Electrónica de la Asociación Paleontológica Argentina 15, 31–39. https://doi.org/10.5710/PEAPA.10.09.2015.97 (2015).
Caldwell, M. W. & Calvo, J. Details of a new skull and articulated cervical column of Dinilysia patagonica Woodward, 1901. J. Vertebr. Paleontol. 28, 349–362. https://doi.org/10.1671/0272-4634(2008)28[349:DOANSA]2.0.CO;2 (2008).
Zaher, H., Apesteguía, S. & Scanferla, C. A. The anatomy of the Upper Cretaceous snake Najash rionegrina Apesteguía & Zaher, 2006, and the evolution of limblessness in snakes. Zool. J. Linn. Soc. 156, 801–826. https://doi.org/10.1111/j.1096-3642.2009.00511.x (2009).
Garberoglio, F. F. et al. New skulls and skeletons of the Cretaceous legged snake Najash, and the evolution of the modern snake body plan. Sci. Adv. 5, eaax5833. https://doi.org/10.1126/sciadv.aax5833 (2019).
Apesteguía, S. & Novas, F. E. Large Cretaceous sphenodontian from Patagonia provides insight into lepidosaur evolution in Gondwana. Nature 425, 609–612. https://doi.org/10.1038/nature01995 (2003).
Apesteguía, S. & Carballido, J. L. A new eilenodontine (Lepidosauria, Sphenodontidae) from the Lower Cretaceous of central Patagonia. J. Vertebr. Paleontol. 34, 303–317. https://doi.org/10.1080/02724634.2013.803974 (2014).
Gentil, A. R., Agnolin, F. L., Garcia Marsà, J. A., Motta, M. J. & Novas, F. E. Bridging the gap: Sphenodont remains from the Turonian (Upper Cretaceous) of Patagonia. Palaeobiological inferences. Cretaceous Res. 98, 72–83. https://doi.org/10.1016/j.cretres.2019.01.016 (2019).
Apesteguía, S., Garberoglio, F. F. & Gómez, R. O. Earliest Tuatara relative (Lepidosauria: Sphenodontinae) from southern continents. Ameghiniana 58, 416–441. https://doi.org/10.5710/AMGH.13.07.2021.3442 (2021).
Montoya-Sanhueza, G. & Chinsamy, A. Long bone histology of the subterranean rodent Bathyergus suillus (Bathyergidae): Ontogenetic pattern of cortical bone thickening. J. Anat. 230, 203–233. https://doi.org/10.1111/joa.12547 (2017).
Newman, D. G. Burrow use and population densities of Tuatara (Sphenodon punctatus) and how they are influenced by fairy prions (Pachyptila turtur) on Stephens Island, New Zealand. Herpetologica 43, 336–344 (1987).
Thompson, M. B., Packard, G. C., Packard, M. J. & Rose, B. Analysis of the nest environment of tuatara Sphenodon punctatus. J. Zool. 238, 239–251. https://doi.org/10.1111/j.1469-7998.1996.tb05392.x (1996).
Andersen, D. C. Below-ground herbivory: The adaptive value of geomyid burrows. Am. Nat. 119, 18–28 (1982).
Bown, T. M. & Kraus, M. J. Ichnofossils of the alluvial Willwood Formation (lower Eocene), Bighorn Basin, northwest Wyoming, USA. Palaeogeogr. Palaeoclimatol. Palaeoecol. 43, 95–128 (1983).
Smith, J. J., Hasiotis, S. T., Kraus, M. J. & Woody, D. T. Relationship of floodplain ichnocoenoses to paleopedology, paleohydrology, and paleoclimate in the Willwood Formation, Wyoming, during the Paleocene-Eocene thermal maximum. Palaios 23, 683–699. https://doi.org/10.2110/palo.2007.p07-080r (2008).
Retallack, G. J. Late Oligocene bunch grassland and early Miocene sod grassland paleosols from central Oregon, USA. Palaeogeogr. Palaeoclimatol. Palaeoecol. 207, 203–237 (2004).
Laza, J. Ichnology of the Lowlands of South America. Paleoichnological Studies in Continental Cenozoic Rocks (Springer, 2020).
Melchor, R. N., de Valais, S. & Genise, J. E. Ichnia 2004: First International Congress on Ichnology, Field Trip Guidebook (eds Bellosi, E. & Melchor, R.) 47–63 (Museo Paleontológico Egidio Feruglio, 2004).
Genise, J. F. Ichnoentomology: Insect Traces in Soils and Paleosols Vol. 37, 695 (Springer International Publishing, 2016).
Weaver, L. N. et al. Early mammalian social behaviour revealed by multituberculates from a dinosaur nesting site. Nat. Ecol. Evol. 5, 32–37. https://doi.org/10.1038/s41559-020-01325-8 (2021).
Ratcliffe, B. C. & Fagerstrom, J. A. Invertebrate Lebensspuren of Holocene flood plains: their morphology, origin and paleoecological significance. J. Paleontol. 54, 614–630 (1980).
Schlirf, M., Uchman, A. & Kümmel, M. Upper Triassic (Keuper) non-marine trace fossils from the Haßberge area (Franconia, south-eastern Germany). Paläontol. Z. 75, 71–96 (2001).
Mendoza Belmontes, F., Melchor, R. N. & Piacentini, L. N. Wolf spider burrows from a modern saline sandflat in central Argentina: morphology, taphonomy and clues for recognition of fossil examples. PeerJ 6, e5054. https://doi.org/10.7717/peerj.5054 (2018).
Klappa, C. F. Rhizoliths in terrestrial carbonates: classification, recognition, genesis and significance. Sedimentology 27, 613–629 (1980).
Sarjeant, W. A. S. The Study of Trace Fossils (ed Frey, R. W.) 163–179 (Springer, 1975).
Pfefferkorn, H. W. & Fuchs, K. A field classification of fossil plant substrate interactions. Neues Jahrbuch für Geologie Paläontologie, Abhandlungen 183, 17–36 (1991).
McDaniel, P. A., Lowe, D. J., Arnalds, O. & Ping, C.-L. Handbook of Soil Sciences: Properties and Processes (eds Huang, P. M. et al.) 29–48 (CRC Press, 2012).
Tabor, N. J. & Myers, T. S. Paleosols as indicators of paleoenvironment and paleoclimate. Annu. Rev. Earth Planet. Sci. 43, 333–361. https://doi.org/10.1146/annurev-earth-060614-105355 (2015).
Harris, J. & Van Couvering, J. Mock aridity and the paleoecology of volcanically influenced ecosystems. Geology 23, 593–596. https://doi.org/10.1130/0091-7613(1995)023%3c0593:MAATPO%3e2.3.CO;2 (1995).
Fedorov, A. et al. 3D Slicer as an image computing platform for the Quantitative Imaging Network. Magn. Reson. Imaging 30, 1323–1341. https://doi.org/10.1016/j.mri.2012.05.001 (2012).
Herrel, A., Schaerlaeken, V., Moravec, J. & Ross, C. F. Sexual shape dimorphism in tuatara. Copeia 2009, 727–731 (2009).
Bullock, P., Fedoroff, N., Jongerius, A., Stoops, G. & Tursina, T. Handbook for Soil Thin Section Description (Wain Publications, 1985).
Stoops, G. Guidelines for Analysis and Description of Soil and Regolith Thin Sections (Soil Science Society of America, 2003).
Stoops, G., Marcelino, V. & Mees, F. Interpretation of Micromorphological Features of Soils and Regoliths (Elsevier, 2010).
Acknowledgements
This work was funded by project PICT 2019-114 from the Agencia Nacional de Promoción Científica y Tecnológica of Argentina, and project PIP–146 from the Consejo Nacional de Investigaciones Científicas y Técnicas to RM. Anthony Herrel kindly shared its database on modern Sphenodon used in the estimation of body mass for fossil material. We would like to thank to all the colleagues who have collaborated in the field campaigns.
Author information
Authors and Affiliations
Contributions
The individual contributions of each co-author to the article are as follows: R.M. participated of the field work, coordinated the manuscript and participated in the data analysis. M.P., P.V., A.U. participated in the field work, manuscript and data analysis. N.E. and M.C.C. provided contextual data, data analysis. and participated of the manuscript. All authors discussed and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Melchor, R., Perez, M., Villegas, P. et al. Early Cretaceous lepidosaur (sphenodontian?) burrows. Sci Rep 13, 10209 (2023). https://doi.org/10.1038/s41598-023-37385-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-37385-6
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.