Abstract
The objective of this study was to identify the prognostic factors and to propose a new risk model in metastatic castration-resistant prostate cancer (mCRPC) patients. The clinical data were retrospectively obtained for 102 mCRPC patients who received cancer treatment between 2005 and 2018 at the University of Tokyo Hospital. We investigated clinical and pathological parameters, including prostate-specific antigen (PSA) kinetic profiles under androgen deprivation treatment, and identified predictors of overall survival (OS). The median age and PSA were 73 (Interquartile range [IQR], 68–79) years and 5.00 (IQR, 2.77–13.6) ng/ml. The median follow-up was 34 (IQR, 17–56) months. In univariate analysis, ‘lymph node metastasis’, ‘Hemoglobin (Hb)’, ‘Time to nadir PSA (TNPSA)’, ‘PSA doubling time (PSADT)’, ‘Time to CRPC’, and ‘presence of pain’ were prognostic factors. Multivariate analysis identified ‘Hb < 11 g/dL’, ‘TNPSA < 7 months’ and ‘PSADT < 5 months’ as independent prognostic factors of OS. The high-risk group (patients with two or three factors) demonstrated shorter OS (23 vs. 50 months) with an increased risk of death (HR = 2.997; 95% CI 1.632–5.506; P = 0.0004). The proposed risk stratification model may contribute to the prediction of survival and provide supportive information in treatment decision-making.
Similar content being viewed by others
Introduction
Prostate cancer is the second most commonly diagnosed type of cancer1, and the number of newly diagnosed cases was estimated at 1,414,000 patients in the fiscal year 20202. Prostate-specific antigen (PSA) screening increased the rates of localized-prostate cancer patients, but also detected metastatic prostate cancer in more than 10% of the screened patients3.
Historically, metastatic castration-sensitive prostate cancers (mCSPC) are treated with androgen deprivation therapy (ADT) by gonadotropin-releasing hormone agonists, antagonists, or orchiectomy. However, most of the ADT-treated mCSPC patients develop castration-resistant prostate cancers (CRPC) within three years4. CRPC is a condition in which response to ADT treatment can no longer be expected, and the entity is associated with a very poor prognosis5. Metastatic CRPC (mCRPC) is characterized by the rapid growth of cancer, and the median survival rate was reported as 13.2 months6. Although mCRPC is a lethal disease, the treatment response differs among individual cases, and risk stratification model may be useful for patient counseling at diagnosis and design of prospective trials.
In 2004, docetaxel was approved for the treatment of mCRPC patients based on the randomized control trials7,8. Consequently, many prognostic factors of overall survival (OS) of the mCRPC patients who received docetaxel were reported, such as visceral metastasis9, the number of metastatic sites9, presence of pain9,10, performance status (PS)9,11, progression type9, Gleason score (GS)9, PSA9, alkaline phosphate (ALP)9,11, hemoglobin (Hb)9,11, lactate dehydrogenase (LDH)10, albumin (Alb)10, PSA doubling time (PSADT)11, cycles of chemotherapy11,12, time to castration-resistant prostate cancer (TTCRPC)11.
New therapeutic options, including androgen receptor-axis targeted therapies (ARAT), were approved for mCRPC treatment in Japan based on randomized phase 3 trials in recent years: abiraterone acetate13, enzalutamide14, and cabazitaxel15. In mCRPC patients who received these new therapeutic agents, many prognostic factors of OS have been identified. Such factors include liver metastasis16, PS16, duration of ADT treatment16, time from prostate cancer diagnosis17, presence of pain17, PSA18,19, LDH16,17, Alb16, ALP17, Hb18, cycles of chemotherapy18, response to prior chemotherapy18, PSA decline after CRPC treatment20,21, age19, GS19, nadir PSA22, and time to nadir PSA (TNPSA)22.
Among these cohorts, some prognostic factors can only be obtained after CRPC treatment, such as cycles of chemotherapy, response to chemotherapy, and PSA decline after CRPC treatment11,12,18,20,21. Although these factors might reflect OS well, we cannot apply these factors for the OS prediction or treatment decision-making at the time of CRPC diagnosis. Therefore, we aimed to investigate the OS prognostic factors of mCRPC patients among clinical factors obtained before CRPC treatment.
Results
Patient characteristics
The clinical and histological features of the eligible patients are presented in Table 1. This retrospective cohort included 102 M1CRPC patients, with a median follow-up of 34 (Interquartile range [IQR], 17–56) months. The median age of patients at the CRPC diagnosis was 73 (IQR, 68–79) years. The majority (95%) of patients had metastasis at the initial diagnosis of prostate cancer, while 5 (4.9%) patients had no metastatic lesion at the time of prostate cancer diagnosis but had developed metastasis during ADT treatment. Seventy-eight out of 102 patients received combined androgen blockade therapy with anti-androgen at CRPC diagnosis. Notably, the patient characteristics based on the cutoffs of identified prognostic factors are also shown in Supplementary Table 2–4.
Treatment sequences
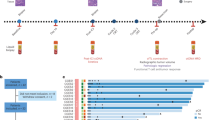
The treatment sequences for CRPC are shown in Fig. 1. Among the patients who received life-prolonging therapies, docetaxel monotherapy was the most administered regimen in the first-line treatment for CRPC (29.4%; 30 of 102 patients), followed by enzalutamide (12.7%) and abiraterone acetate (4.9%). Fifty-four (52.9%) patients received vintage therapies, including flutamide, chlormadinone, low-dose dexamethasone, or estramustine, of which 20 patients received docetaxel as a 2nd line treatment. Patients who received docetaxel as the first-line therapy received cabazitaxel or ARAT as the second-line treatment. On the other hand, patients who received ARAT as the first line received secondary ARAT agents, of which five patients received docetaxel as the third-line treatment.
Prognostic factors for OS and risk stratification
In the univariate analysis, six prognostic factors out of 19 clinical parameters were significantly associated with the OS of mCRPC patients (Table 2). The number of factors nominated for the multivariate analysis was based on the one in ten rule (one predictive variable can be studied for every ten events.) that allowed us to include 5 factors in the analysis23. ‘Time to CRPC < 20 months’ was excluded in the multivariate analysis as this factor may be reflected by the combination of ‘Time to nadir PSA’ and ‘PSA doubling time’.
Multivariate analysis further identified ‘Hb < 11 g/dL’, ‘TNPSA < 7 months’ and ‘PSADT < 5 months’ as independent predictors of OS. Risk stratification was performed based on these predictors (Fig. 2A). Patients with two or three factors were classified as the high-risk group, and patients with zero or one risk factor were classified as the low-risk group. The median OS was 23 months (95% Confidence interval [CI], 20 to 38 months) in the high-risk group and 50 months (95% CI 41 to 58 months) in the low-risk group, respectively (Fig. 2B). In addition, 1-year, 3-year, and 5-year survival rates in the high-risk and low-risk groups were 84.5% vs. 95.8%, 40.8% vs. 86.5%, and 24.5% vs. 63.0%, respectively (Fig. 2C). Patients in the high-risk group demonstrated an increased risk of death (HR = 2.997; 95% CI 1.632–5.506; P = 0.0004) compared to the patients in the low-risk group (Fig. 2D). Internal validation using a bootstrap method showed a C-index of 0.69, 0.66, 0.67 for the proposed risk models based on the entire cohort, chemotherapy group, and non-chemotherapy group, respectively (Figs. 2B, 3A,B).
A nomogram was developed for predicting 3-year OS based on the prognostic factors (Supplementary Fig. 1A). The calibration plots demonstrated good consistency between predicted and actual 3-year OS (Supplementary Fig. 1B).
Subgroup analysis
Due to the treatment variability in this cohort, patients were divided into groups according to the use of chemotherapy at any time during the treatment (Fig. 1). We further conducted survival analysis on these two groups to ascertain whether the risk classification would be applicable regardless of chemotherapy treatment.
Overall, 56 (54.9%) patients received chemotherapy (chemotherapy group), and 46 (45.1%) patients received ARAT or vintage therapies (non-chemotherapy group) (Supplementary Table 1). Between chemotherapy and non-chemotherapy group, age at prostate cancer and CRPC diagnosis, TTCRPC, and PSA reduction rate (PSARR), the value of PSA and Hb were statistically different.
Patients were categorized into two risk groups based on the total points that were calculated by the number of risk factors (Supplementary Table 5). After subgrouping, a statistically significant difference in OS was observed between risk groups for patients regardless of chemotherapy treatment (Fig. 3A,B). The 3-year survival rates for the patients with and without chemotherapy treatment were 90.2% and 83.2% in the low-risk group and 44.7% and 32.7% in the high-risk group, respectively (Fig. 3C,D). Multivariate analysis was performed using the three prognostic factors for the subgroups divided by the presence of chemotherapy use (Supplementary Table 6). ‘Hb < 11 g/dL’ was statistically significant in the non-chemotherapy group, while ‘TNPSA < 7 months’ and ‘PSA doubling time < 5 months’ were significant prognostic factors of OS in the chemotherapy group.
Discussion
We identified Hb < 11 g/dL, TNPSA < 7 months and PSADT < 5 months as independent prognostic factors in mCRPC patients. Based on our new risk model, patients with a higher risk showed worse survival rates regardless of the use of chemotherapy treatment. Additionally, the nomogram that was developed in the present study showed optimal agreement between predicted and actual observation regarding 3-year OS.
Our cohort defined OS as the time from CRPC diagnosis until death, though other mCRPC cohorts calculated OS from the time point of the treatment randomization9,16 or treatment initiation17,20. Several cohorts assessed OS prediction factors by the same OS definition19,22 as the present study and demonstrated that age19, GS19, PSA19, nadir PSA22, and TNPSA22 were independent OS prognostic factors of mCRPC patients. We identified ‘Hb < 11 g/dL’ as a new prognostic factor of OS in non-chemotherapy treated patients. Notably, this factor was not assessed in the previous cohorts19,22.
Several CRPC cohorts reported a low Hb level as an OS prognostic factor, both for patients receiving any treatment9, and for patients who received chemotherapy11,18. However, a subanalysis of the impact of low Hb levels in patients who did not receive chemotherapy has not been conducted previously. Given this background, we further divided patients according to the presence of chemotherapy treatment and identified ‘Hb < 11 g/dL’ as a prognostic factor of the patients who never received chemotherapy treatment as well. Anemia worsens the prognosis of patients in other cancer patient cohorts24,25. Our cohort consists of the patients diagnosed as CRPC after ADT treatment for metastatic prostate cancer treatment, and these patients were assumed to be in chronic and progressive disease status. Several factors might induce anemia, such as bone marrow replacement by metastatic cells26, chronic inflammation27, and cytokine-mediated disorder28. The low Hb level might reflect these factors and impact the OS consequently.
In the present study, ‘PSADT < 5 months’ was a prognostic factor of OS in mCRPC patients. PSADT was previously reported as a predictor of OS in nonmetastatic CRPC29. In mCRPC patients, shorter PSADT increased the risk of overall mortality in the TAX327 cohort9. However, the PSADT used in the study was basically calculated at the point of treatment initiation, in which CRPC treatment was initiated after a PSA level of 100 ng/mL. In our cohort, PSADT predicted OS using much lower PSA levels measured at the time of CRPC diagnosis.
In this cohort, the cut-off value of PSADT was five months, which was longer than the 46.3 days that was previously reported in the CRPC cohort with docetaxel treatment by Qu et al.11. Suggested cut-off values vary greatly among publications depending on the method of calculation, mainly because of the difference in time-points of PSA measurement. Qu Y.Y. et al. calculated PSADT using two PSA values at the time of CRPC diagnosis and the most recent PSA level before the CRPC diagnosis11. By contrast, we used all PSA values within 12 months according to the recommendation of PSA Working Group30. However, the disadvantage of this method is that the calculation becomes complicated when using three or more PSA values.
Our cohort has several limitations. First, clinical data were retrieved retrospectively. To confirm the utility of the identified prognostic factors, prospective studies are necessary. Second, several cohorts showed PS as a prognostic factor of OS9,11,16. However, this cohort did not assess PS due to missing data. Third, treatment strategy was not consistent in this cohort, so while this study may reflect real-world clinical situations, there may be a bias concerning the selection of treatment. Fourth, as many as 45% of the patients in this study have never received chemotherapy, due to mental and physical intolerance to chemotherapy. Similar results were observed in a previous study in which 61% of the patients received docetaxel among the mCRPC patients diagnosed between 2010–201331. Another study by George et al. reported that only 8% of the patients received docetaxel treatment as 2nd line treatment among the patients who received abiraterone or enzalutamide as 1st line. Fifth, the sample size of this study was limited and patients were treated with various types of treatment. Therefore, future studies with external validation may be required to determine the generalizability of the risk stratification that was presented in this study.
Despite these limitations, our analysis showed the impact of TNPSA, PSADT, and Hb on the prognosis of mCRPC patients. Since the factors analyzed were limited to those which could be generated at the time of mCRPC, this risk stratification may be used to predict the prognosis of mCRPC patients during routine patient treatment. This finding requires further validation by prospective cohorts.
Materials and methods
Study design
This retrospective cohort included patients diagnosed as mCRPC between 2005 and 2018 at the University of Tokyo Hospital. This study was performed according to the provisions of the Declaration of Helsinki and approved by the ‘Ethics Committee of the Tokyo University Hospital’ (approval number 3124). Regarding the present study, the ‘Ethics Committee of the Tokyo University Hospital’ waived the requirement of the written informed consent.
Patient selection
Clinical data of patients that showed metastatic lesions at CRPC diagnosis were retrospectively extracted from medical records at the University of Tokyo Hospital. Metastatic lesions were identified by conventional imaging techniques such as bone scans and computed tomography of the chest, abdomen, and pelvis. CRPC was defined as having PSA or radiographic progression under castrated status. ‘Castrated status’ was defined as ‘serum total testosterone < 50 ng/dL’ or ‘condition in which ADT treatment is performed either by surgical orchiectomy or luteinizing hormone-releasing agonist/antagonist’32. PSA progression was defined as an increase in PSA level of ≥ 25% and ≥ 2 ng/mL above the nadir32. Radiographic progression was defined as two or more new metastatic lesions detected on the bone scans33, or one lesion detected and confirmed as adenocarcinoma by biopsy of the metastatic lesion.
A total of 13 patients were excluded due to insufficient information. Two patients were considered intolerable for CRPC treatment due to poor general condition, and were excluded for the present study. Two patients had other concomitant aggressive malignancies and were excluded because of inability to evaluate metastatic lesions. In total, 102 patients were selected for the final analysis.
Data retrieval
Clinical parameters were reviewed from the clinical records, including age, prostate biopsy GS before treatment, presence of lymph node metastasis, medications, complications, blood tests at the time of CRPC diagnosis, the use of local therapy, and the treatment sequences for CRPC. The extent of disease was determined by the radiographic image by bone scans34. TTCRPC was defined as the duration between the start of ADT treatment and CRPC diagnosis. The presence of symptomatic pain was decided by whether analgesic agents were prescribed at the time of CRPC diagnosis. Charlson comorbidity index was calculated according to the definition developed by Charlson et al.35. All PSA results obtained during a 12 month time span leading up to CRPC diagnosis were collected, and used for the PSADT calculation by the web-based calculator from Memorial Sloan Kettering Cancer Center (https://www.mskcc.org/nomograms/prostate/psa_doubling_time). PSA measurements under 0.2 ng/mL were not used in the calculation of PSADT30. PSARR was calculated using 1 − (nadir PSA)/(PSA value at prostate cancer diagnosis). TNPSA was defined as the time interval between ADT initiation and nadir PSA during ADT treatment before CRPC diagnosis. OS was defined as the time from CRPC diagnosis until death from any cause or the last follow-up.
Statistical analysis
Clinical data were shown by the frequency and the percentage of categorical variables, or the median value attached with the IQR for continuous variables. Regarding continuous variables, the cutoff values were determined by the closest point to the upper left corner of the ROC curve analysis (Youden index) and rounded to the clinically significant value. Univariate and multivariate analyses were performed using Cox proportional hazards regression model to identify independent factors predicting OS. Associations between continuous variables were compared by the Mann–Whitney U test, and the difference of categorical values was compared by Fisher’s exact test or Pearson’s chi-square test. All P values with < 0.05 were considered statistically significant. Statistically significant prognostic factors in the univariate analysis were included in the multivariate analysis. Patients were categorized into high and low-risk groups based on the aforementioned identified factors. The Kaplan–Meier curves of OS in each risk group were analyzed by log-rank test to evaluate the difference in OS among risk groups. The internal validation was carried out by calculating C-index of the risk model based on 1,000 bootstrap samples. A nomogram was developed using a Cox regression model based on the independent prognostic factors identified in the multivariate analysis. Calibration curves of the nomogram for 3-year OS were generated to determine the deviation of the observed probability from the predicted probability of survival using 200 bootstrap resamples. JMP 16.1.0 (SAS Institute Inc., Cary, NC, USA) was used for statistical analysis except for the calculation of the C-index, development of the nomogram, and construction of the calibration curves in which R Version 3.6.0 (Comprehensive R Archive Network) was used.
Data availability
The dataset used in the present study is not publicly available due to the ongoing clinical studies based on the same dataset. However, it can be used by a reasonable request to the corresponding author.
References
Rawla, P. Epidemiology of prostate cancer. World J. Oncol. 10, 63–89 (2019).
Gandaglia, G. et al. Epidemiology and prevention of prostate cancer. Eur. Urol. Oncol. 4, 877–892 (2021).
Saito, E. et al. Long-term trends in prostate cancer incidence by stage at diagnosis in Japan using the multiple imputation approach, 1993–2014. Cancer Epidemiol. Biomark. Prev. 29, 1222–1228 (2020).
Lin, T. T. et al. Risk factors for progression to castration-resistant prostate cancer in metastatic prostate cancer patients. J. Cancer 10, 5608–5613 (2019).
Chandrasekar, T., Yang, J. C., Gao, A. C. & Evans, C. P. Mechanisms of resistance in castration-resistant prostate cancer (CRPC). Transl. Androl. Urol. 4, 365–380 (2015).
Aly, M. et al. Survival in patients diagnosed with castration-resistant prostate cancer: A population-based observational study in Sweden. Scand. J. Urol. 54, 115–121 (2020).
Petrylak, D. P. et al. Docetaxel and estramustine compared with mitoxantrone and prednisone for advanced refractory prostate cancer. N. Engl. J. Med. 351, 1513–1520 (2004).
Tannock, I. F. et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N. Engl. J. Med. 351, 1502–1512 (2004).
Armstrong, A. J. et al. A contemporary prognostic nomogram for men with hormone-refractory metastatic prostate cancer: a TAX327 study analysis. Clin. Cancer Res. 13, 6396–6403 (2007).
Halabi, S. et al. Updated prognostic model for predicting overall survival in first-line chemotherapy for patients with metastatic castration-resistant prostate cancer. J. Clin. Oncol. 32, 671–677 (2014).
Qu, Y. Y. et al. Prognostic factors in Chinese patients with metastatic castration-resistant prostate cancer treated with docetaxel-based chemotherapy. Asian J. Androl. 15, 110–115 (2013).
Kawahara, T. et al. Risk factors for metastatic castration-resistant prostate cancer (CRPC) predict long-term treatment with docetaxel. PLoS ONE 7, e48186 (2012).
Ryan, C. J. et al. Abiraterone acetate plus prednisone versus placebo plus prednisone in chemotherapy-naive men with metastatic castration-resistant prostate cancer (COU-AA-302): final overall survival analysis of a randomised, double-blind, placebo-controlled phase 3 study. Lancet Oncol. 16, 152–160 (2015).
Beer, T. M. et al. Enzalutamide in metastatic prostate cancer before chemotherapy. N. Engl. J. Med. 371, 424–433 (2014).
Oudard, S. TROPIC: Phase III trial of cabazitaxel for the treatment of metastatic castration-resistant prostate cancer. Future Oncol. 7, 497–506 (2011).
Chi, K. N. et al. A prognostic index model for predicting overall survival in patients with metastatic castration-resistant prostate cancer treated with abiraterone acetate after docetaxel. Ann. Oncol. 27, 454–460 (2016).
Jimenez Romero, M. E., Gallego, M. C., Navarro Serrato, J. C., Sanchez Hurtado, M. A. & Santotoribio, J. D. Treatment of metastatic castration-resistant prostate cancer patients with abiraterone acetate and prednisone and corresponding survival prognostic factors. Anticancer Res. 41, 3955–3968 (2021).
Yasuoka, S. et al. Risk factors for poor survival in metastatic castration-resistant prostate cancer treated with cabazitaxel in Japan. Anticancer Res. 39, 5803–5809 (2019).
Valero, J., Peleteiro, P., Henriquez, I., Conde, A., Piquer, T., & Lozano, A., et al. Age, Gleason Score, and PSA are important prognostic factors for survival in metastatic castration-resistant prostate cancer. Results of The Uroncor Group (Uro-Oncological Tumors) of the Spanish Society of Radiation Oncology (SEOR). Clin. Transl. Oncol. 22, 1378–89 (2020).
Bosso, D. et al. PSA declines and survival in patients with metastatic castration-resistant prostate cancer treated with enzalutamide: A retrospective case-report study. Medicine (Baltimore) 96, e6817 (2017).
Schiff, J. P. et al. Early prostate-specific antigen response post-abiraterone as predictor of overall survival in metastatic castrate-resistant prostate cancer. BMC Cancer 19, 524 (2019).
Hamano, I. et al. Impact of nadir PSA level and time to nadir during initial androgen deprivation therapy on prognosis in patients with metastatic castration-resistant prostate cancer. World J. Urol. 37, 2365–2373 (2019).
Harrell, F. E. Jr., Lee, K. L. & Mark, D. B. Multivariable prognostic models: Issues in developing models, evaluating assumptions and adequacy, and measuring and reducing errors. Stat. Med. 15, 361–387 (1996).
Wei, Q. et al. Correlation between hemoglobin levels and the prognosis of first-line chemotherapy in patients with advanced gastric cancer. Cancer Manag Res. 12, 7009–7019 (2020).
Wan, S. et al. Post-diagnosis hemoglobin change associates with overall survival of multiple malignancies—Results from a 14-year hospital-based cohort of lung, breast, colorectal, and liver cancers. BMC Cancer 13, 340 (2013).
Mercadante, S., Gebbia, V., Marrazzo, A. & Filosto, S. Anaemia in cancer: Pathophysiology and treatment. Cancer Treat Rev. 26, 303–311 (2000).
Cash, J. M. & Sears, D. A. The anemia of chronic disease: spectrum of associated diseases in a series of unselected hospitalized patients. Am. J. Med. 87, 638–644 (1989).
Means, R. T. Jr. & Krantz, S. B. Progress in understanding the pathogenesis of the anemia of chronic disease. Blood 80, 1639–1647 (1992).
Smith, M. R. et al. Natural history of rising serum prostate-specific antigen in men with castrate nonmetastatic prostate cancer. J. Clin. Oncol. 23, 2918–2925 (2005).
Arlen, P. M., Bianco, F., Dahut, W. L., D'Amico, A., Figg, W. D. & Freedland, S. J., et al. Prostate Specific Antigen Working Group guidelines on prostate specific antigen doubling time. J. Urol. 179, 2181–5; discussion 5–6 (2008).
Francini, E. et al. Impact of new systemic therapies on overall survival of patients with metastatic castration-resistant prostate cancer in a hospital-based registry. Prostate Cancer Prostatic Dis. 22, 420–427 (2019).
McGregor, B. et al. Bone targeted therapy and skeletal related events in the era of enzalutamide and abiraterone acetate for castration resistant prostate cancer with bone metastases. Prostate Cancer Prostatic Dis. 24, 341–348 (2021).
Scher, H. I. et al. Design and end points of clinical trials for patients with progressive prostate cancer and castrate levels of testosterone: recommendations of the Prostate Cancer Clinical Trials Working Group. J. Clin. Oncol. 26, 1148–1159 (2008).
Soloway, M. S. et al. Stratification of patients with metastatic prostate cancer based on extent of disease on initial bone scan. Cancer 61, 195–202 (1988).
Charlson, M. E., Pompei, P., Ales, K. L. & MacKenzie, C. R. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J. Chronic Dis. 40, 373–383 (1987).
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
Conceptualization: Y.H., Y.Y., T.K.; Data collection: Y.H., T.K., M.N., T.I., T.T., Y.K., D.Y., M.S.; methodology: Y.Y., Y.T.; formal analysis: Y.H.; supervision: H.K.; writing—original draft; Y.H., Y.Y., Y.T.; writing—review and editing: Y.Y., S.T., T.K, Y.A., Y.S., D.Y., M.S., H.K. All authors have read and agreed to the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hakozaki, Y., Yamada, Y., Takeshima, Y. et al. Low hemoglobin and PSA kinetics are prognostic factors of overall survival in metastatic castration-resistant prostate cancer patients. Sci Rep 13, 2672 (2023). https://doi.org/10.1038/s41598-023-29634-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-29634-5
This article is cited by
-
Increase of prostate-specific antigen doubling time predicts survival in metastatic castration-resistant prostate cancer patients undergoing radium therapy
Annals of Nuclear Medicine (2024)
-
Flare phenomenon visualized by 99mTc-bone scintigraphy has prognostic value for patients with metastatic castration-resistant prostate cancer
Annals of Nuclear Medicine (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.