Abstract
Incidental meningiomas (IMs) are the most common intracranial neoplasms, especially in perimenopausal women. There is ongoing debate on whether their incidence is increased by hormone replacement therapy. Meningiomas often express estrogen receptors, which were linked to higher proliferative activity according to some reports. Consequently, there is a theoretical risk of estrogen-based HRT (e-HRT) leading to an increase in tumor growth and thus altering the natural history of IMs. However, clinical data is lacking to support this notion. To identify differences in the natural history of IM after e-HRT exposure. We queried the NorthShore Meningioma Database for patients with ≥ 6 months of e-HRT. They were compared with age-matched IM controls. Forty patients were included in the e-HRT group (mean age 62.1 ± 12.0 years; mean duration of HRT 5.3 ± 4.5 years) and 80 in the no-HRT group (mean age 62.2 ± 12 years). Radiographic appearance was similar between groups. The average 2D tumor diameter was 35% lower in the e-HRT group (p = 0.02), with an absolute growth-rate of half of the no-HRT group (p = 0.02). Radiographic and clinical progression-free survival were 1.2 years and 3.3 years longer in the e-HRT group, respectively. These preliminary results suggest that e-HRT may be safe in incidental meningiomas.
Similar content being viewed by others
Introduction
Meningiomas are the most common primary intracranial neoplasm, accounting for more than half of all benign tumors of the central nervous system1. According to CBTRUS estimates, nearly 30,000 meningiomas are discovered annually in the United States. The diagnosis is usually incidental, and since these tumors are typically indolent, most require no immediate intervention. By definition, incidental meningiomas are usually small and not associated with neurologic symptoms at the time of discovery. Nonetheless, their growth over time may lead to significant morbidity and mortality. Some incidental meningiomas will eventually require treatment, which typically consists of surgery or radiotherapy. Meningiomas are two-to-three-fold more common in women compared to men. Their incidence shows a sharp rise in the 4th decade of life overlapping with the perimenopausal period in women1,2,3. Consequently, it is common for women diagnosed with meningiomas to be concurrently receiving or considering initiation of hormone replacement therapy (HRT).
Most meningiomas are known to express progesterone receptors (PR) and are often positive for estrogen receptors (ER) as well4,5,6,7,8. Unlike progesterone, estradiol was implicated as a potent enhancer of meningioma cell proliferation in vitro4,9, and ER-positive meningiomas were associated with a higher proliferative index than their ER-negative counterparts7. Therefore, there is a theoretical concern for accelerated tumor growth in women receiving estrogen HRT (e-HRT). This notion seems to be supported by a number of epidemiology studies that found increased meningioma incidence in women who received e-HRT3,10,11. Nonetheless, epidemiologic data on incidental meningiomas provide little guidance as to whether closer monitoring or a more aggressive approach to treatment is warranted if there is a history of prior HRT, or whether it is safe for a patient with these tumors to start or continue HRT. Unfortunately, in the absence of studies that assess the association of HRT with in vivo meningioma growth rate and clinical outcomes, clinicians have to rely on anecdotal evidence and expert opinion on how to manage HRT and incidental meningiomas concurrently12.
The present study aimed to provide clinically translatable evidence through a retrospective comparison of tumor growth patterns and clinical outcomes in meningioma patients who received estrogen hormone replacement therapy (e-HRT), as compared to those without a history of female sex hormone replacement therapy (no-HRT).
Methods
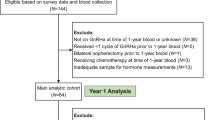
We conducted a single-center retrospective case–control study of female patients monitored for probable meningiomas within the NorthShore University HealthSystem Neurology and Neuro-Oncology practice. We included women with radiographic findings consistent with an incidentally found (i.e., asymptomatic) meningioma and with six or more documented months of clinical and radiographic follow-up.
All eligible patients were 18 years of age or older and were treated with e-HRT for at least 6 months, initiated within 5 years before or during their meningioma surveillance period. Treatment status was determined based on retrospective review of medication records and clinic notes. We compared each e-HRT patient with two age-matched female controls with a diagnosis of incidental meningioma, and no history of HRT, all of which were recruited from the same meningioma cohort (Fig. 1). This study was approved by the NorthShore University HealthSystem Institutional Review Board and all methods were carried out in accordance with relevant guidelines and regulations. The Informed Consent procedure was waived by the Northshore HealthSystem Institutional Review Board, based on retrospective nature of the study and no more than minimal risk to the subjects.
Evaluation of growth rate
Baseline radiographic characteristics on MRI were compared between the two groups based on previously established parameters predictive of meningioma behavior, such as intensity on T2-weighted MRI, presence of calcification on CT, or on susceptibility-weighted MRI sequences, and the presence of perilesional edema13,14. Size measurements were performed using the contrast-enhancing regions on MRI, whereas calcification was assessed using susceptibility-weighted MRI, or CT scans acquired within a year from the initial MRI. We established size as the (a) single largest perpendicular dimension (1D) and (b) the largest biperpendicular diameter as described by McDonald et al. (2D)15. For both 1D and 2D measures, absolute annual growth rates (AGR: [final size − initial size]/follow up time in years) and proportional annual growth rates (PGR: AGR/initial size * 100%) were estimated. Growth trends were visualized on spaghetti plots and were qualitatively analyzed to identify differences in growth patterns. Meningioma size at diagnosis and end of follow-up, as well as growth rates, were compared between the e-HRT and no-HRT groups. Group comparisons for nominal parameters were performed using Fisher’s exact test. Continuous variables were first tested for normality; those with a normal distribution were compared using Student’s independent sample two-tailed T-tests, whereas non-normal variables were compared using the Mann–Whitney U-test.
Evaluation of outcomes
Progression-free survival (PFS) was assessed radiographically, and defined by: (a) ≥ 5 mm AND ≥ 20% 1D increase as compared to the initial scan (RECIST 1.1)16; (b) ≥ 25% increase in 2D size compared to the first scan (and ≥ 3 mm increase in largest diameter if largest biperpendicular diameters < 10 mm at diagnosis, following RANO meningioma criteria)15,17; (c) time to clinical progression, defined as development of symptoms attributable to the meningioma or initiation of radiotherapy or resection. If multiple meningiomas were present, only the dominant (i.e., largest) lesion was measured. PFS group differences were assessed via the log-rank test and visualized using Kaplan–Meier curves. Cox regression analyses were performed to assess the relationship between PFS (radiographic and clinical) and the duration of hormone therapy, timing relative to diagnosis, HRT route, and BMI.
We repeated the same analyses for the following subgroups: (1) e-HRT via oral/subcutaneous/transdermal route only, (2) vaginal estrogen only, (3) e-HRT for at least one year before diagnosis, and (4) e-HRT for at least one year after diagnosis. The threshold for statistical significance was p < 0.05. Statistical analysis was carried out using IBM SPSS 25.0 (Armonk, NY).
Results
Patient characteristics
Forty e-HRT patients met all the eligibility criteria; 11 completed e-HRT before the meningioma diagnosis, 11 started e-HRT before and continued during the surveillance period, and 18 patients started e-HRT during surveillance (Table 1). Their mean age was 62.1 years (± 1.9). The mean duration of e-HRT was 5.0 years (± 0.7). Most patients (62.5%) received oral or transdermal products, whereas 30% used vaginal e-HRT, and 7.5% received e-HRT through more than one route. The age-matched control group consisted of 80 patients. Age at the end of follow up was similar between the groups, despite the e-HRT group had a non-significant trend toward having longer follow up (5.3 ± 0.4 vs. 6.8 ± 0.6 years, p = 0.061). Average BMI was significantly lower in the e-HRT group (26.3 ± 0.9 vs. 29.1 ± 0.9 kg/m2; p = 0.043). There was no difference in parity between the groups. Sixty-three percent of the e-HRT group had a history of hysterectomy.
Radiographic characteristics
Radiographic appearance, including the presence of calcification, edema, or T2-weighted intensity, were also similar between the e-HRT and control group (p ≥ 0.3). Meningiomas in the e-HRT group were smaller at diagnosis (13.0 mm vs. 16.1 mm) and remained smaller at the end of follow-up (15.1 vs. 18.7 mm; p = 0.03 for both comparisons; Table 2). The 2D measurements revealed similar group differences. Although both absolute and proportional growth rates were 1.5–2.3 times higher in the no-HRT group across 1D and 2D measures, only the 2D AGR difference was statistically significant (28.0 mm2/year vs. 12.1 mm2/year; p = 0.023). Visualized growth patterns were similar between the two groups (Supplemental Fig. 1).
In the subgroup analysis, e-HRT and control groups maintained the same distribution of most averages, as noted in the whole group analysis, but none of the comparisons reached statistical significance.
Outcome analysis
Mean PFS defined by RECIST 1.1, RANO, and clinical criteria were 11.2, 8.7, and 14.8 years in the e-HRT group versus 10.6, 8.4, and 11.5 years in the control group, respectively (Fig. 2). None of the differences were statistically significant (log-rank p > 0.1 in all analyses). Duration of e-HRT did not have an effect on either radiographic or clinical progression-free survival in the e-HRT group (HR = 1.00, p > 0.2; Supplemental Table 2).
Kaplan–Meier curves of progression-free survival (PFS), based on (a) the largest diameter (mm; RECIST 1.1), (b) the largest biperpendicular diameters (mm2, RANO), or (c) clinical criteria where progression is defined as new symptom attributable to the meningioma, or initiation of treatment. For patients with multiple meningiomas, only the dominant lesion was included in the measurement. Vertical lines represent censored cases.
The subgroup analyses showed consistently lower risk of progression in the e-HRT group, but none of these comparisons reached the level a statistical significance (Supplemental Table 2).
Discussion
In this retrospective study of incidental meningiomas, we found consistently smaller tumors in those receiving estrogen hormone replacement within five years of discovery of meningioma or during surveillance. The differences were most pronounced on 2D measurements, revealing 35% smaller tumors in the HRT group both at diagnosis and the at end of follow-up. Growth rates were about twofold slower in the hormone replacement group, but only the difference in absolute 2D growth rates was significant in this cohort. Clinical PFS was 3.3 years longer for the e-HRT group, whereas radiographic PFS was very similar to the no-HRT group. No additional subgroup differences were revealed based on HRT route, timing relative to diagnosis, duration of the regimen used.
Our findings add clinical nuance to the limited and often conflicting evidence regarding the effect of e-HRT on meningiomas and their growth-rates. On a cellular level, PR expression is nearly ubiquitous in grade I meningiomas. In contrast, the rate of ER positivity has been reported in a wide range from 1 to 48%, which is likely a result of variability in measurement methods, and is possibly underestimated due to the preoperative use of glucocorticoids that can inhibit sex hormone receptor expression4,6,7,8,18. Of note, ER expression has been associated with higher meningioma proliferative rates in some in vitro and ex vivo studies7,9. Yet, on the individual level, in human data is restricted to case series of meningioma progression during pregnancy and extrapolation of experience with other cancers, such as the higher rate of progression in previously treated breast cancer patients19,20,21,22. On a population level, epidemiology studies that assessed the overall risk of meningioma development in women using HRT also yielded inconsistent results11,23,24,25,26,27. One of the most extensive case–control studies involved over 1000 cases and controls and concluded no statistically significant risk for meningioma development with HRT23. In contrast, a meta-analysis found the pooled odds ratio to be 1.29 compared to the HRT naïve11. A more recent case–control study looking at an ethnically diverse population found an odds ratio of 1.09 for e-HRT27. However, to the best of our knowledge, none of these studies have reported meningioma growth-rates or any form of outcomes in patients receiving e-HRT.
The majority of meningiomas are discovered incidentally and have a slow growth-rate. Most only require surgical intervention years or decades after the initial diagnosis, if any at all. Given these intrinsic tumor characteristics, studies assessing outcomes in meningiomas are unable to use overall survival as an appropriate outcome measure, and radiographic outcome measures are still under development17. In our study, we assessed radiographic (1D and 2D) as well as clinical PFS as surrogate measures for tumor behavior. Although semi-automated 3D volumetric analysis may be more sensitive to detect subtle tumor growth and can be especially useful for monitoring en plaque meningiomas in a research setting, it requires advanced image processing that is not widely available in the clinical practice, neither is it standardized17. Instead, the present study is intended to emulate information gathering in the day-to-day clinical setting. 1D and 2D measurements, as well as clinical data, can be quickly performed in the clinical setting. Some may argue that the association between growth rate and HRT exposure may only be evident after a long treatment period, given the slow progression of these tumors. However, a recent study of incidental meningiomas found a plateauing of growth-rate in these tumors after five years. 14 Thus, the almost seven years of follow up and five years of e-HRT should be sufficient to reveal significant growth acceleration. Both the statistically significant and non-significant differences found in the present study may suggest the opposite: on average, patients in the e-HRT group completed their follow up with smaller meningiomas, as opposed to controls. Therefore, there is no clear trend to indicate a possible masked effect due to short follow-up time. Also, the duration of hormone replacement was not a significant risk factor for radiographic or clinical progression in the main group nor the subgroup analysis. Lastly, it is unclear whether this group difference is due to chance due to the lower BMI in the e-HRT group. Higher BMI had been previously connected to a higher risk to develop meningiomas23,27.
Limitations
The most significant limitations of this study are due to the relatively low sample size and the retrospective design. The latter evokes the potential risk of differential reporting on medication use in cases vs. controls. In order to improve the accuracy of data collection, our study selected patients based on records of medication prescription, as opposed to reported use.
Another limitation of our study is the non-uniform timing of e-HRT in relation to diagnosis and monitoring of the meningiomas; 28% of patients in this cohort had already completed e-HRT by the time of their meningioma diagnosis. Measuring the diameters on the last MRI available should still allow size comparison as it designates the end of exposure for all the patients included.
This analysis was performed in a progestin agnostic fashion as the majority of e-HRT patients had a history of hysterectomy, and thus the low number of patients receiving progestins was insufficient for a meaningful analysis.
Lastly, the focus on incidental meningiomas may have introduced selection bias through automatic exclusion of cases with a symptomatic presentation. Among these could theoretically be a group of estrogen “super sensitive” tumors with a highly accelerated growth-rate after e-HRT is started. However, the existence of such group is not supported by our data from patients who started e-HRT during the surveillance period.
Conclusion
In conclusion, this is the first study to our knowledge that reports on growth-rate of meningiomas in patients receiving estrogen HRT. We found a similar to lower meningioma growth-rate in women exposed to estrogen HRT during the surveillance period, and up to 5-years prior to diagnosis compared to women not receiving estrogen HRT. Our study has the potential for changing practice in suggesting it may be safe for women to continue estrogen-based HRT after diagnosis of a meningioma; however, the role of progestins in HRT is not addressed here. Additionally, our study suggests that women with meningiomas taking estrogen HRT do not require more aggressive monitoring than other patients. A larger prospective case–control study is necessary provide further support for our findings.
References
Ostrom, Q. T. et al. CBTRUS Statistical Report: primary brain and other central nervous system tumors diagnosed in the United States in 2011–2015. Neuro-Oncology 20, iv1–iv86 (2018).
Nakasu, S., Hirano, A., Shimura, T. & Llena, J. F. Incidental meningiomas in autopsy study. Surg. Neurol. 27, 319–322 (1987).
Christensen, H. C., Kosteljanetz, M. & Johansen, C. Incidences of gliomas and meningiomas in Denmark, 1943 to 1997. Neurosurgery 52, 1327–1334 (2003).
Blankenstein, M. A., van der Meulen-Dijk, C. & Thijssen, J. H. H. Effect of steroids and antisteroids on human meningioma cells in primary culture. J. Steroid Biochem. 34, 419–421 (1989).
Koehorst, S. G. A. & Koehorst, S. G. A. Wild type and alternatively spliced estrogen receptor messenger RNA in human meningioma tissue and MCF7 breast cancer cells. J. Steroid Biochem. Mol. Biol. 45, 227–233 (1993).
Speirs, V., Boyle-Walsh, E. & Fraser, W. D. Constitutive co-expression of estrogen and progesterone receptor mRNA in human meningiomas by RT-PCR and response of in vitro cell cultures to steroid hormones. Int. J. Cancer 72, 714–719 (1997).
Korhonen, K. et al. Female predominance in meningiomascan not be explained by differences in progesterone, estrogen, or androgen receptor expression. J. Neurooncol. 80, 1–7 (2006).
Leães, C. G. S. et al. Immunohistochemical expression of aromatase and estrogen, androgen and progesterone receptors in normal and neoplastic human meningeal cells. Neuropathology 30, 44–49 (2010).
Jay, J. R., MacLaughlin, D. T., Riley, K. R. & Martuza, R. L. Modulation of meningioma cell growth by sex steroid hormones in vitro. J. Neurosurg. 62, 757–762 (1985).
Qi, Z.-Y. et al. Reproductive and exogenous hormone factors in relation to risk of meningioma in women: a meta-analysis. PLoS ONE 8, e83261 (2013).
Fan, Z.-X. et al. Hormone replacement therapy and risk of meningioma in women: a meta-analysis. Cancer Causes Control CCC 24, 1517–1525 (2013).
Wahab, M. & Al-Azzawi, F. Meningioma and hormonal influences. Climacteric 6, 285–292 (2003).
Lee, E. J. et al. A novel weighted scoring system for estimating the risk of rapid growth in untreated intracranial meningiomas. J. Neurosurg. 127, 971–980 (2017).
Islim, A. I. et al. A prognostic model to personalize monitoring regimes for patients with incidental asymptomatic meningiomas. Neuro-Oncology https://doi.org/10.1093/neuonc/noz160 (2019).
Macdonald, D. R., Cascino, T. L., Schold, S. C. Jr. & Cairncross, J. G. Response criteria for phase II studies of supratentorial malignant glioma. J. Clin. Oncol. 8, 1277–1280 (1990).
Eisenhauer, E. A. et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 11). Eur. J. Cancer Oxf. Engl. 1990(45), 228–247 (2009).
Huang, R. Y. et al. Proposed response assessment and endpoints for meningioma clinical trials: report from the Response Assessment in Neuro-Oncology Working Group. Neuro-Oncology 21, 26–36 (2018).
Cahill, D. W. et al. Estrogen and progesterone receptors in meningiomas. J. Neurosurg. 60, 985–993 (1984).
Roelvink, N. C., Kamphorst, W., van Alphen, H. A. M. & Rao, B. R. Pregnancy-related primary brain and spinal tumors. Arch. Neurol. 44, 209–215 (1987).
Lusis, E. A. et al. Meningiomas in pregnancy. Neurosurgery 71, 951–961 (2012).
Holmberg, L. & Anderson, H. HABITS (hormonal replacement therapy after breast cancer—is it safe?), a randomised comparison: trial stopped. The Lancet 363, 453–455 (2004).
Fahlén, M. et al. Hormone replacement therapy after breast cancer: 10 year follow up of the Stockholm randomised trial. Eur. J. Cancer 49, 52–59 (2013).
Claus, E. B. et al. Exogenous hormone use, reproductive factors, and risk of intracranial meningioma in females. J. Neurosurg. 118, 649–656 (2013).
Hatch, E. E. et al. Reproductive and hormonal factors and risk of brain tumors in adult females. Int. J. Cancer 114, 797–805 (2005).
Wigertz, A. et al. Risk of brain tumors associated with exposure to exogenous female sex hormones. Am. J. Epidemiol. 164, 629–636 (2006).
Custer, B., Longstreth, W., Phillips, L. E., Koepsell, T. D. & Van Belle, G. Hormonal exposures and the risk of intracranial meningioma in women: a population-based case-control study. BMC Cancer 6, 152 (2006).
Muskens, I. S. et al. Body mass index, comorbidities, and hormonal factors in relation to meningioma in an ethnically diverse population: the Multiethnic Cohort. Neuro-Oncology 21, 498–507 (2019).
Author information
Authors and Affiliations
Contributions
L.D., R.M. and D.K. wrote the main manuscript text, and D.K. prepared the figures. L.D. and R.M. contributed equally to the main manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Dresser, L., Yuen, C.A., Wilmington, A. et al. Estrogen hormone replacement therapy in incidental intracranial meningioma: a growth-rate analysis. Sci Rep 10, 17960 (2020). https://doi.org/10.1038/s41598-020-74344-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-74344-x
This article is cited by
-
Nomogram for predicting cardiovascular disease mortality in patients with meningioma: a competing risk analysis
Neurosurgical Review (2023)
-
Hormone therapies in meningioma-where are we?
Journal of Neuro-Oncology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.