Abstract
The link between spatio-temporal resource patterns and animal movement behaviour is a key ecological process, however, limited experimental support for this connection has been produced at the home range scale. In this study, we analysed the spatial responses of a resident large herbivore (roe deer Capreolus capreolus) using an in situ manipulation of a concentrated food resource. Specifically, we experimentally altered feeding site accessibility to roe deer and recorded (for 25 animal-years) individual responses by GPS tracking. We found that, following the loss of their preferred resource, roe deer actively tracked resource dynamics leading to more exploratory movements, and larger, spatially-shifted home ranges. Then, we showed, for the first time experimentally, the importance of site fidelity in the maintenance of large mammal home ranges by demonstrating the return of individuals to their familiar, preferred resource despite the presence of alternate, equally-valuable food resources. This behaviour was modulated at the individual level, where roe deer characterised by a high preference for feeding sites exhibited more pronounced behavioural adjustments during the manipulation. Together, our results establish the connections between herbivore movements, space-use, individual preference, and the spatio-temporal pattern of resources in home ranging behaviour.
Similar content being viewed by others
Introduction
Animals move to change the environmental conditions they experience1 such as the presence of predators and competitors, and the availability of resources. Because foraging efficiency can be linked to individual fitness2, food acquisition is thought to be a primary driver underlying animal movements3. Consequently, space-use represents the geographic realization of optimizing fitness as a function of resource availability and acquisition costs4.
Food resources are usually dynamic in both space and time5. In the case of herbivores, animals typically feed on vegetation distributed in patches, which are characterized by important temporal variations in quantity and quality6. In this context, strong spatio-temporal gradients in resource availability, at either landscape or regional scales, appear to drive migration and nomadism tactics7. In many herbivore populations, however, individuals show a high year-round fidelity to a spatially-localized home range. It has been suggested that the foraging benefits of site familiarity, where resources are constant or predictable, are responsible for the formation of a stable home range (see Fagan et al.8 for a review). While the home range has traditionally been perceived as a relatively static space-use tactic, recent evidence suggests that animals have sub-seasonal home ranges9 i.e., focus their movements into particular areas in response to seasonal variation in local resource availability. For example, sub-seasonal home ranges are a ubiquitous behavioural tactic across a wide ecological gradient in roe deer (Capreolus capreolus)10.
The link between animal movement behaviour and resource dynamics is less clear when observing home ranging behaviour than migration or nomadism3 because of the difficulty to quantify spatio-temporal variability in resource heterogeneity at small spatial scales10. In this study, we address this issue by experimentally manipulating the spatio-temporal patterns of food availability within home ranges. In situ food manipulation experiments have a long history in the study of population dynamics (e.g., understanding the numerical response of a population to food supplementation)11, and of animal communities12. Although these field experiments have provided fundamental insights in animal ecology, they have seldom been combined with the emerging technological capabilities of animal tracking13 to investigate the implications of food availability on individual movements and space-use at larger spatial scales. In a rare example of field experiment in large herbivores, white-tailed deer (Odocoileus virginianus) shifted their core-area i.e., familiar areas of use, in response to novel food supplementation14. In turn, great tits (Parus major) showed personality-dependent variability in responses to an alteration of resource distribution15.
Our research builds upon these two studies14,15 by investigating the spatial responses of a large herbivore, roe deer, to an experimental in situ manipulation of a high-quality, concentrated food resource in relation to both individual resource preferences and site familiarity. Roe deer, as solitary browsers with limited fat reserves16, exhibit a tight association between movement and resource dynamics17 with a strong plasticity to adapt its resource acquisition at different spatio-temporal scales18,19,20,21. In contrast to group-living ungulates, their foraging decisions are expected to be clearly expressed at the level of individuals.
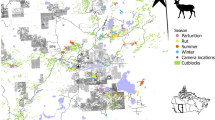
We collared roe deer in the Eastern Italian Alps with GPS units and followed their movements during transitory alterations of food availability (high-nutritional corn) at supplemental feeding sites (FS) i.e., discrete resource patches with an identifiable resource value distinguishable from the vegetation matrix4,5. The 6-week experiment consisted of three two-week phases—pre-closure, closure and post-closure. During the closure phase, we physically restricted the accessibility of food at the most attended feeding site of each individual (hereafter referred to as Manipulated, M, and considered as most familiar; Fig. 1). Throughout the experiment, roe deer had access to two alternative resources within the broader landscape: alternate feeding sites (A), whose food provisioning was held constant, and natural vegetation (V). The experiment was conducted during winter, when food scarcity limits roe deer foraging performance, and individuals are most inclined to adjust their spatial behaviour to continue meeting their energy requirements22. The experiment therefore mimics—on free-ranging animals—the variation in the availability of concentrated, high-reward resources akin to watering holes for savannah ungulates23 and feral horses (Equus ferus)24, or fruit trees for hornbills (Ceratogymna atrata and C. cylindricus)25 and frugivorous primates26,27.
Schematic representation of the experiment. (a) The manipulation consists of a transitory alteration of resource accessibility at a manipulated (M) feeding site (FS). (b) The experiment was expected to lead to spatial responses in the monitored roe deer, and in particular in a shift of use from M (green/red dot, change of colour denoting the alteration of accessibility) towards alternative resources—alternate FS (A; black triangles) or the natural vegetation (V; underlying matrix). In particular, this can lead to spatio-temporal dynamics in space-use (utilization distribution: colour gradient; 95% and 50% contour lines: thick and thin white lines, respectively; data from roe deer F5-2018).
Our initial hypothesis stated that individuals altered their movement behaviours and consequently space-use patterns to track dynamics in resource availability (H1; Table 1). We predicted that the loss of a key foraging resource should lead to larger (P1.1) and spatially-shifted (P1.2) home ranges, resulting from more explorative movements (P1.3). Furthermore, we predicted that roe deer reduced the intensity of use of the manipulated, familiar FS (M) when food accessibility was prevented (P1.4a) and compensated for this loss by using alternate, accessible FS (A; P1.4b).
We further hypothesized that the behavioural adjustments to changes in resource availability would vary between individuals (H2; Table 1). In particular, because roe deer males have been shown to maintain a high year-round fidelity to their summer territory28, we predicted that they would respond less markedly to the experiment than females (P2.1). We also predicted the responsiveness of roe deer to be positively influenced by the individual’s prior preference for FS (P2.2).
If the spatial patterns of roe deer home ranges result from the benefits of site familiarity29,30, animals should strive to use familiar areas and resources when accessible (H3; Table 1). Accordingly, we predicted that when initial conditions of food accessibility are re-established after perturbation, the initial space-use patterns would be restored (P3.1), following a return to high use of the familiar FS (M; P3.2).
Results
Space-use and movement responses to alteration of resource availability
Roe deer space-use changed significantly during the experiment: the size of both home ranges (95% UD isopleth; Fig. 2a; Supplementary Information S3: Table S1) and core areas (50% isopleth; Fig. 2b; Supplementary Information S3: Table S2) increased significantly during the experimental closure (P1.1). On average, home range size increased from 27.21 ha (\(\sigma\)=11.37) during pre-closure to 34.68 ha (\(\sigma\)=10.33) during closure, and settled to 30.55 ha (\(\sigma\)=9.10) during post-closure. Core area size followed a similar trend with averages of 3.98 ha (\(\sigma\)=2.29), 5.66 ha (\(\sigma\)=2.34) and 5.04 ha (\(\sigma\)=2.07), respectively.
Changes in roe deer space-use patterns—home range size (y-axis, a), core area size (y-axis, b) and space-use overlap (y-axis, c)—as a function of preference for feeding sites (x-axis) and experimental phase (colour; a, b) and phase contrast (colour; c). Observations are represented as dots (females) and squares (males; points are jittered slightly), and model predictions as solid lines (95% confidence intervals: ribbons). Parameter estimates and associated significance can be found in Supplementary Information S3: Tables S1, S2 and S3.
Home range and core area sizes were influenced by individual preference for FS (\({h}_{FS}\)) and there was an interaction between \({h}_{FS}\) and experimental phase: individuals with a high \({h}_{FS}\) tended to have smaller home ranges during the pre-closure, smaller core areas overall, and stronger increases in both home range and core area sizes following the experimental closure (Fig. 2a,b; Supplementary Information S3: Tables S1, S2; P2.2). There was no significant effect of sex or interactions between sex and experimental phase on home range size (P2.1 not supported). Overall, the models quantifying the changes in observed home range and core area sizes accounted for a high proportion of the total variance (conditional coefficient of determination, cR2: 0.62 and 0.51, respectively).
Alongside home range size, roe deer home range shifted dramatically following the experimental closure (Fig. 2c): the degree of space-use overlap between pre-closure and closure phases was significantly lower (mean = 0.370, CI = 0.301–0.405; P1.2) than the overlap between the temporally-separated pre- and post-closure phases (mean = 0.535, CI = 0.475–0.594; P3.1). Space-use overlap was significantly affected by \({h}_{FS}\) (Fig. 2c; P2.2), with higher \({h}_{FS}\) being associated to larger space-use shifts following closure (Supplementary Information S3: Table S3). However, there was no apparent influence of sex in the observed space-use patterns (P2.1 not supported). The fitted model accounted for an important proportion of the variance in space-use overlap (cR2 = 0.50).
Marked changes in roe deer movement behaviour during the experiment underpinned the observed modifications in space-use patterns. Average hourly step length during the pre-closure phase was 60.32 m (\(\sigma\)=85.79); during the closure phase it increased to 74.26 m (\(\sigma\)=108.11); and during post-closure it decreased to 68.18 m (\(\sigma\)=96.61, P1.3). In general, males (Supplementary Information S4: Fig. S1, right-hand panels, Table S1; P2.1), and individuals associated with high \({h}_{FS}\) values (Supplementary Information S4: Fig. S1, top panels, Table S1; P2.2) exhibited stronger increases in step length during the closure phase. In addition, roe deer movements were more persistent during the closure phase, as shown by a significant decrease in the mean absolute turning angle for males with a high \({h}_{FS}\) (Supplementary Information S4: Fig. S2, top-right panel, Table S2; P1.3, P2.1, P2.2).
Resource use responses to alteration of resource availability
The spatio-temporal dynamics of resource availability during the experiment led to important shifts in resource use (Fig. 3; Supplementary Information S5: Table S1). On average, the proportion of use of the manipulated FS (M) dropped from 31% during the pre-closure phase to 4% during closure (P1.4a), and then rebounded to 18% in the post-closure phase (P3.2). This decrease in the use of M during the closure phase was partially compensated by elevated use of the alternate FS (A)—which increased from 2 to 16% following closure (P1.4b), and an increase of the use of vegetation (V) from 67 to 80% following closure. During the post closure, use of A and V declined to 8% and 73%, respectively. The shifts in resource use were very consistent among roe deer for M and A but were more variable for V (Fig. 3, top panels).
Roe deer shifts in resource use during the experiment—manipulated feeding site (M, left column), alternate feeding sites (A, central column) and vegetation (V, right column). Top row: mean proportional use (dots and lines) as a function of the experiment phase (x-axis) and preference for feeding sites (colour scale). Bottom row: predictions of the resource use models (\({u}_{M,t}\), \({u}_{A,t}\) and \({u}_{V,t}\); estimate: solid lines; 95% confidence interval: ribbon) and mean relative use (females: dots; males: squares) as a function of the experiment phase (colour) and preference for feeding sites (x-axis). The model predictions do not consider resource lags at 1, 2 and 24 h nor the influence of sex (although selected in the final model for \({u}_{A,t})\) for clarity and conciseness. Parameter estimates and associated significance can be found in Supplementary Information S5: Table S1.
Roe deer preference for FS significantly influenced how animals used the three resource types and, in particular, interacted with experimental phase for M and A (Fig. 3, bottom panels; Supplementary Information S5: Table S1). Roe deer characterized by a high \({h}_{FS}\) had significantly higher use of M during pre-closure (by definition) and post-closure, as well as consistently lower use of V. High \({h}_{FS}\) animals were associated with stronger decreases in use of M and larger increases in the use of A during closure (P2.2). This compensation for A during closure was stronger for females (Supplementary Information S5: Fig. S1, Table S1; P2.1). However, sex did not influence the use of M or V (P2.1 not supported). Overall, the fitted models accounted for a high proportion of the variance in resource use (cR2: 0.35, 0.21 and 0.31 for M, A and V, respectively).
Discussion
The results of this field resource manipulation experiment provide direct evidence for the tight coupling between the spatio-temporal distribution of resources and consequently spatially-restricted movements of a resident large herbivore. Specifically, we showed that roe deer tracked resource dynamics (Fig. 3; H1), which led to changes in their space-use (Fig. 2) and underlying movement behaviour (Supplementary Information S4: Figs. S1, S2), and that individual traits, especially resource preference, mediated these behavioural adjustments (H2). In addition, we showed that roe deer exhibited a high attraction to familiar locations, a process which led to site fidelity (H3). As far as we are aware, this is the first experimental demonstration of these interdependencies in a large mammalian herbivore.
The experimental alterations of food availability led to larger, spatially-shifted home ranges (Fig. 3), and more exploratory movements by roe deer (Supplementary Information S4: Figs. S1, S2), thereby directly establishing the connections between movement, space-use and the spatio-temporal patterns of resources.
In a previous observational study, elk were shown to alternate between two movement modes: a low speed and high sinuosity mode thought to be within-patch area-restricted search, and a high speed and low sinuosity mode between resource patches31. In our experimental study, we can directly link these movement modes to changing resources: the exploratory movements of roe deer (high velocity and low sinuosity) observed during the closure phase (P1.3) suggested that the animals were motivated to find alternate resource patches when their familiar feeding site (FS) became inaccessible, thereby increasing (P1.1), but also shifting (P1.2), their home range. While changes in home range size and location following resource manipulation have been found in studies of lizards32, birds15 and voles33, to date, there have been few experimental investigations of the connections between space-use and the spatio-temporal distribution of resources in large mammals.
In an earlier experimental study, white-tailed deer shifted their home range core towards the vicinity of newly deployed FS14. Our study builds upon these results by demonstrating multiple, successive responses to resource manipulation. Specifically, we link measured changes in underlying fine-scale movement behaviour of individuals to resulting patterns of space-use that indicate dynamic resource tracking behaviour by roe deer (H1). Although roe deer increased their use of the vegetation matrix during the closure phase, individuals compensated the loss of their manipulated FS (M) to a large degree by shifts in their movements and space-use towards alternate FS (A; Figs. 3; P1.4a–b). Consequently, individuals maintained a high overall use of FS throughout the experiment.
While resource tracking behaviour may be expected under optimal foraging theory, in many free-ranging animals, social fences arising from territoriality or density-dependent resource competition constrain the movement responses of individuals to changes in the spatial distribution of resources34. In our experimental system, these concerns were largely alleviated because, first, roe deer do not generally defend territories (with the exception of adult males during spring and summer35—seasons not covered by our experiment), and consequently their spatial distribution can approximate that of an ideal free distribution36. Second, while intra- and inter-specific competition in herbivores is largely linked to resource depletion6, this density-dependent constraint of food availability was prevented by providing ad libitum forage at the FS.
This study demonstrates that inter-individual variation in preference for FS strongly mediated the responses of roe deer movement patterns, space-use and resource use to changes in the spatio-temporal distribution of resources (H2). During the closure phase, the changes in all measured variables were larger for individuals associated with a high FS preference (P2.1). The influence of FS preference was particularly striking in the shifts of space-use (Fig. 2c) and in the compensating use of alternate FS following the loss of the familiar resource (Fig. 3). The observed inter-individual differences in FS preference (Supplementary Information S2: Table S1) may be linked to either the environment the individuals were exposed to, or a property of the individuals themselves. Preference can therefore be considered a dynamic variable37 that we evaluated at the individual level over a short period of relative stability (pre-closure phase in each winter). We considered the temporal extent of our experiment (ca 6 weeks) short enough to consider FS preference for each animal to be relatively constant, because in this time period the physiological conditions and vegetation nutritional value would not vary substantially or consistently.
Animals attending FS benefit from exploiting a forage-rich location, however, they may risk elevated intra- and inter-specific competition, disturbance from human presence, and an increased susceptibility to predation and hunting. In roe deer, individual behavioural profile (e.g., body temperature at capture) correlates with the use of risky but profitable habitats such as open areas, suggesting that variations in personality could lead to individual differences in habitat use38. By analogy, FS preference could be associated to bold or risk-taking personalities. Interestingly, preference for FS tended to correlate with individual body temperature at capture (Pearson’s r = − 0.37 p value = 0.084; Supplementary Information S7), with bolder individuals (lower temperature) using FS more intensely. Personality may not only lead individuals to use resources to a different extent, but also condition their tendency to track spatio-temporal resource dynamics39, and explore novel environments40. In our study, roe deer with higher preference for FS (i.e., bolder) were those responding most markedly to the experiment, consistently with patterns observed in great tits15.
During the post-closure phase of the experiment, roe deer increased their use of familiar FS (M), whose food accessibility had been restored after a transitory restriction (Fig. 3, left-hand panels; P3.1), and home ranges shifted back to pre-closure patterns, as suggested by the high spatial overlap between temporally-disjointed pre- and post-closure space-use (Fig. 2c; P3.2). The restoration of these pre-manipulation patterns supports the hypothesis that site familiarity provides inherent benefits to animals maintaining a home range (H3)41. These results are coherent with published literature demonstrating that ungulates tend to select for previously visited locations i.e., site familiarity42,43. Indeed, the systematic return of roe deer to M during the post-closure phase, when both alternative resources were accessible, suggests that roe deer were actively selecting for familiar areas and that site familiarity had an inherent value.
In observational studies of animal movement, a spurious familiarity effect can occur when an important factor influencing animal behaviour is not considered, and the re-visitation of particular locations is interpreted as an evidence for site familiarity selection44. However, this confounding effect is unlikely to affect the results of this experiment. First, corn was delivered ad libitum across all FS (M or A) i.e., homogeneous foraging value. Second, the FS were located in comparable environments and especially in relation to cover, a factor that largely influences roe deer movements and space-use45,46. Third, and most importantly, the specific identities of M and A varied interchangeably between individuals (see Supplementary Information S1: Table S1). Hence, we conclude that the return to pre-closure patterns of foraging behaviour and space-use are unlikely to be result of variations in the characteristics of specific FS, but rather of an inherent familiarity effect. For roe deer, site familiarity could allow a more profitable exploitation of natural forage, as seen for bison (Bison bison)43 or reduce intraspecific competition for such resource (see Riotte-Lambert et al.29 for a theoretical argument). In our study area, there were no natural predators. However, in presence of predation risk the attraction to familiar areas could be related to an avoidance tactic47.
Our results imply that when resource patterns are changing, individual behavioural decisions likely reflect both site familiarity and resource tracking. Species with little fat reserves need a constant, high-nutritional intake, and may hence be required to rapidly adjust their movements away from their most familiar areas to track spatio-temporal resource dynamics, as seen in roe deer during the closure phase of this experiment or in great tits15. It would be interesting to investigate whether capital breeding species, with greater capacity to buffer transitory shortages of food availability, are more reluctant to abandon familiar locations.
Ultimately, site familiarity is the manifestation of an animal’s ability to acquire spatial information, in particular by means of spatial memory8. Large herbivores are capable of memorizing the location and profitability of resources43. In this study, it is likely that the variations in roe deer responses to resource changes that are not explained by preference may be the result of individual prior experience and knowledge of the status and distribution of concentrated resources. An interesting avenue for further studies will be to evaluate the role of these cognitive processes on individual foraging decisions.
Methods
Study area
The study area is located in the north-eastern Italian Alps (Argentario range, in Val di Cembra and Valsugana; Autonomous Province of Trento), covers c. 16 km2 and ranges between 500 and 1,000 m a.s.l. The topography is generally mild, but steeper slopes (> 30°) occur in the northern portion. The climate is continental and characterized by a mean temperature of 1.0 °C in January and 21.0 °C in July, and a mean annual rainfall of 966 mm (average 2000–2018; https://www.meteotrentino.it). There is occasional snow cover between December and March, although the soil is mostly frozen at night. The study area is covered by 80.0% forest, mostly as relatively homogeneous secondary growth stands interspersed with small pastures. The forests are dominated by Pinus sylvestris with abundant shrub undergrowth, and by mixed stands of Fagus sylvatica, Picea abies and Abies alba and, to a lower extent, by Quercus petraea stands.
Roe deer is the primary large herbivore in the study area (6–9 individuals km−2; ref. values from Autonomous Province of Trento Wildlife Office). Adult roe deer do not have natural predators in this landscape, but young fawns may be predated by red fox (Vulpes vulpes). The fine-scale food selection of roe deer in the Alps has been described as mainly dependent on shrubs or regeneration of tree species as well as a diversity of herbaceous plants from the undergrowth from spring to fall, switching between items according to the temporal trends of availability48. In the winter time, roe deer strongly select for forested environments and opportunistically for supplemental food where available22.
Supplemental feeding management of roe deer is conducted at > 50 distinct feeding sites within the study area (FS; Supplementary Information S1: Fig. S1) and authorized year-round within a larger zone of c. 45 km2 (official authorization: “Autonomous Province of Trento order n. 2852/2013”). FS are typically shaped as wooden hopper dispensers that provide a continuous supply of corn accessible through a tray (Fig. 1). They have been deployed and provided continuously with food (at least in fall and winter) for many years (i.e., for longer that the average lifespan of roe deer in our study area). They are managed by private hunters for roe deer but are also attended sporadically by red deer (Cervus elaphus), as well as non-target mammals (Meles meles, Sciurus vulgaris, Apodemus sp., Microtus sp.) and birds (Garrulus glandarius, Columba palumbus).
Experimental design
We took advantage of roe deer use of a focal, identifiable resource—the FS—to design an in situ experimental manipulation of resource availability. We created three successive experimental phases based on the availability of this resource—pre-closure, closure and post-closure—by physically managing the accessibility of food at the FS. During the closure phase, access to forage at FS was transitorily restricted by placing wooden boards obstructing the tray; boards were then removed again in the post-closure phase (Fig. 1).
The experiment was conducted between January and April, when the use of high-nutritional supplemental feed (i.e., corn) by roe deer is the most intense17, for three consecutive winters (2017, 2018 and 2019). We implemented the experiment on 18 individuals, of which seven could be manipulated in two consecutive years—five individuals were recaptured and two collar deployments spanned two winters—leading to a total of 25 individual winter trajectories i.e., “animal-years” (21 adults: 15 females, 6 males; 4 yearlings: 2 females, 2 males; sample size n = 4, 11 and 10 in 2017, 2018 and 2019 respectively; see Supplementary Information S1 for details). Because roe deer captures at middle to low density in Alpine, heavily forested environments are rare events that have to rely on low-efficiency techniques such as box traps and because we had to account for stakeholder acceptance, repeating the experiment on single individuals in consecutive years allowed us to take full advantage of our sample.
Roe deer were captured using baited box traps (n = 21 capture events) or net drives (n = 2), and were fitted with GPS-GSM radio collars programmed to acquire hourly GPS locations for a year, after which they were released via a drop-off mechanism. Captures and marking were performed complying with ethical and welfare rules, under authorization of the Wildlife Committee of the Autonomous Province of Trento (Resolution of the Provincial Government n. 602, under approval of the Wildlife Committee of 20/09/2011, and successive integration approved on the 23/04/2015); all methods and experiments were carried out in accordance with the relevant guidelines and regulations. Radio-collared roe deer moved an average of 61.2 m per hour. This value of the average hourly movement distance (l) was subsequently utilized in the analyses described below.
For all captured animals, we assumed a post-capture response in ranging behaviour. We therefore considered the first re-visitation of the capture location as a likely sign of resettlement in the original range and we used this time as onset of the experimental pre-closure phase. Although not all the individuals were manipulated at the same time, we avoided interference between capture operations and FS manipulations, and between co-occurring different manipulation phases (i.e., ensuring that co-occurring manipulations occurred in separate areas).
During the pre-closure phase, we ensured a continuous supply of food at all managed FS—i.e., that were provisioned at least once in the month prior to the experiment—located within 500 m of each roe deer locations (known through twice-daily download of GSM-transmitted GPS relocations). At the end of the pre-closure phase, we identified the “manipulated” FS (M) for each individual as the managed FS with the largest number of locations within a radius \(l\) during this initial phase, and considered it as the FS to which an individual is most familiar. All other managed FS were considered as “alternate” (A) FS. During the closure phase, corn was made inaccessible at M for a duration of approximately 15 days, depending on personnel availabilities (min = 14.0 days, max = 18.1, mean = 15.5). M was then re-opened, thereby initiating the post-closure phase. During both pre- and post-closure phases, corn was available ad libitum at M. All A FS had corn available ad libitum throughout the duration of the experiment. To ensure a continuous supply of food during the experiment, field personnel visited and replenished the FS every third day. Across the experimental manipulations, we used a total of twelve distinct FS as M, and 23 distinct FS as A (mean = 4.04 A sites per animal-year, \(\sigma \hspace{0.17em}\)= 1.43; of these, an average of 1.76, \(\sigma \hspace{0.17em}\)= 1.13, were actually used by roe deer; see Supplementary Information S1: Table S1 for details on the identity of M and A for all animal-years). M sites were separate from A sites by an average distance of 702.5 m (\(\sigma \hspace{0.17em}\)= 310.5), and M and used A sites by an averaged distance of 567.5 m (\(\sigma \hspace{0.17em}\)= 235.7).
Data preparation
To ensure meaningful comparisons between animal-years, we homogenized the durations of each experimental phase to the minimum length of the closure phase in our sample (i.e., 14 days). Specifically, we truncated the movement data by removing initial excess positions for the pre-closure and closure phases, and terminal excess positions for the post-closure phase. GPS acquisition success was extremely high (99.57% during the experiment) and we did not interpolate missing fixes in the collected data.
The analyses of space-use and movement behaviour were based on spatially-explicit, raw movement trajectories. The analyses of resource use, instead, relied on spatially-implicit, state time series derived from the underlying movement data. To this end, we created an initial time series, for each animal-year, by intersecting the relocations with three spatial domains: vegetation (the matrix; V), manipulated FS (M) and alternate FS (A). We converted FS locations (M and A) into areas by buffering them. To investigate the sensitivity of buffer choice we considered six buffer sizes: l (i.e., 61.2 m) multiplied by 0.5, 1, 1.5, 2, 3 and 4. We associated all locations falling outside M and A to the state V. The three-state time series was then converted into three single-state presence/absence time series.
Preference for feeding sites
We calculated each individual’s preference for FS (\({h}_{FS}\)) as the relative use of FS over natural vegetation during the pre-closure phase (i.e., the proportion of GPS fixes classified as either M or A). Because preference is considered to be temporally dynamic37, we chose to evaluate \({h}_{FS}\) for each year separately in case individuals were manipulated in two separate winters. This reasoning allowed to account for the influence of individual condition and of the relative quality and quantity of vegetation resources on \({h}_{FS}\). We included \({h}_{FS}\) in all space-use, movement, and resource use analyses described below.
The variability of \({h}_{FS}\) across animal-years was maximal when FS attendance was defined as a GPS location within a distance equal to the population mean hourly step length \(l\) i.e., 61.2 m from the FS (interquartile range = 0.278, mean = 0.343; Supplementary Information S2: Table S1). Accordingly, the results described below are based on this definition (see Supplementary Information S6 for a sensitivity analysis). At this scale, \({h}_{FS}\) did not differ consistently between sex (mean for females = 0.346; mean for males = 0.336; t-test: p value = 0.901).
Analysis
We analysed how the experimental manipulation, and its interaction with both preference for FS and sex, affected roe deer space-use, movement behaviour, and resource use.
General modelling approach
We analysed the roe deer responses to the experiment using mixed effect models. The final fixed-effect structure was developed progressively, beginning with simple formulations and evaluating the consistency of our results to ascertain that our data could support more complex formulations. For example, regarding the analysis of home range size, we first fitted a simple function of the experimental phase (i.e., home range size ~ Phase), then evaluated a potential additive effect of preference for feeding sites (i.e., home range size ~ Phase + \({h}_{FS}\)), and then an interaction between the two covariates (i.e., home range size ~ Phase + \({h}_{FS}\)+ Phase:\({h}_{FS}\)). We repeated this procedure when evaluating the effects of sex, and eventually, assessed the full fixed effect structure. We did not find irregularities in the behaviour of the nested models (i.e., marked changes in absolute parameter values or sign). In the full model, fixed effect terms were dropped when statistically non-significant (p value > 0.05). We considered “animal-year” as the sampling unit to account for the fact that an individual may respond independently to manipulations in different years. The choice of an “animal-year” random effect (instead of an “animal” random effect) did not qualitatively affect our results (Supplementary Information S8).
Space-use
We assessed the changes of home range and core area sizes (P1.1), and space-use overlap (P1.2, P3.1) between experimental phases. We calculated utilization distributions (UD)49 for each animal-year and experimental phase using a Gaussian kernel density estimation. After visual inspection, we chose to compute the UDs at a spatial resolution of 10 m and with a fixed bandwidth set to half the average hourly movement distance (i.e., l/2 = 30.6 m).
For home range and core area sizes, we calculated the area (in hectares) corresponding to the 95% and 50% UD contours, respectively, during each experimental phase (Phase; three levels; reference level: Pre-closure). We then analysed the log-transformed areas using a linear mixed-effect model (LMM) with five fixed effects: Phase, \({h}_{FS}\), Sex (categorical predictor; reference level: Female), and two interaction terms (Phase:\({h}_{FS}\) and Phase:Sex). We included animal-year (ind) as random intercept.
We estimated the space-use overlaps for three pairs of UDs—pre- and post-closure, pre-closure and closure, and closure and post-closure (Contrast; three levels; reference level: Pre-/Closure)—using the volume of intersection statistic (VI)50. VI ranges from 0 (no overlap) to 1 (complete overlap). We analysed the logit-transformed overlaps using an LMM with Contrast, hFS, Sex, Contrast:hFS and Contrast:Sex as fixed effects, and ind as random intercept.
Movement behaviour
We investigated the movement responses of roe deer to the experiment (P1.3) by analysing the changes in hourly step length (Euclidean distance between two successive relocations) and turning angle \({\theta }_{t}\) (angle between two successive movement steps). We analysed the log-transformed step length, \({s}_{t}\) and, because turning angles range between \(-\pi\) and \(\pi\), and were symmetric around 0, the logit-transformed absolute turning angle, \({\varphi }_{t}=log\left(\frac{\left|{\theta }_{t}\right|}{1-\left|{\theta }_{t}\right|}\right)\). We used LMMs with Phase, \({h}_{FS}\), Sex, Phase:hFS and Phase:Sex as fixed effects, and ind as random intercept. Because step length was characterized by strong serial autocorrelation at short temporal lags and at circadian periodicities (a common pattern in animal movement trajectories51), we also included step length measured at lags 1, 2 and 24 h (i.e., \({s}_{t-1},{s}_{t-2}\),\({s}_{t-24}\)) as fixed effects to reduce the autocorrelation of the model residuals.
Resource use
To test whether the experiment led to a transitory change in resource use (P1.4a–b, P3.2), we fitted separate mixed-effect logistic regression models to the three single-state presence/absence time series (\({u}_{M,t}\), \({u}_{A,t}\) and \({u}_{V,t}\)) using Phase, \({h}_{FS}\), Sex, Phase:\({h}_{FS}\) and Phase:Sex as fixed effects, and ind as random intercept. The pre-closure level for Phase was dropped for \({u}_{V}\) to avoid circularity (\({h}_{FS}=1-{{\stackrel{-}{u}}_{V,t}}_{Pre-closure}\)). We also included the response variables measured at lags 1, 2 and 24 h (e.g., \({u}_{M,t-1},{u}_{M,t-2}\),\({u}_{M,t-24}\)) as fixed effects to reduce the autocorrelation of the model residuals. However, for the sake of conciseness and clarity, we omitted these response lags when visualizing resource use predictions. Because the model results were consistent regardless of the inclusion of the response lags (Supplementary Information S5: Tables S1, S2), this decision had no impact on the interpretation. Two animal-years were excluded from the analyses of resource use due to the absence of suitable A-state: F4-2017 did not seem to have visited any alternate FS (A) prior to the experiment; and F16-2016 had two distinct, highly-used FS during pre-closure, but only the second most visited FS could be manipulated (due to stakeholder acceptance). While the use of A was more variable when including these two outliers, the general patterns remained unchanged (Supplementary Information S5: Tables S1, S3).
Software
All analyses were conducted in the R environment52. We used the packages adehabitatLT and adehabitatHR53 for the spatial analyses, fitted all mixed-effect models via Maximum Likelihood with the package lme454. We obtained the p-values for the fixed effects using afex55 and coefficients of determination using MuMin56.
Ethical statement
All experimental protocols and data collection were approved by the Wildlife Committee of the Autonomous Province of Trento (Resolution of the Provincial Government n. 602, under approval of the Wildlife Committee of 20/09/2011, and successive integration approved on the 23/04/2015). All experiments and methods were performed in accordance with relevant guideline and regulations.
Data availability
Data are available from the Zenodo Digital Repository (www.zenodo.org): https://doi.org/10.5281/zenodo.3903805.
References
Van Moorter, B., Rolandsen, C. M., Basille, M. & Gaillard, J. M. Movement is the glue connecting home ranges and habitat selection. J. Anim. Ecol. 85, 21–31 (2016).
Stephens, D. W. & Krebs, J. R. Foraging Theory (Princeton University Press, Princeton, 1986).
Mueller, T. & Fagan, W. F. Search and navigation in dynamic environments behaviors to population distributions. Oikos 117, 654–664 (2008).
Mitchell, M. S. & Powell, R. A. A mechanistic home range model for optimal use of spatially distributed resources. Ecol. Model. 177, 209–232 (2004).
Wiens, J. A. Population responses to patchy environments. Annu. Rev. Ecol. Syst. 7, 81–120 (1976).
Owen-Smith, N., Fryxell, J. M. & Merrill, E. H. Foraging theory upscaled: The behavioural ecology of herbivore movement. Philos. Trans. R. Soc. B Biol. Sci. 365, 2267–2278 (2010).
Mueller, T. et al. How landscape dynamics link individual- to population-level movement patterns: A multispecies comparison of ungulate relocation data. Glob. Ecol. Biogeogr. 20, 683–694 (2011).
Fagan, W. F. et al. Spatial memory and animal movement. Ecol. Lett. 16, 1316–1329 (2013).
Benhamou, S. Of scales and stationarity in animal movements. Ecol. Lett. 17, 261–272 (2014).
Couriot, O. et al. Truly sedentary? The multi-range tactic as a response to resource heterogeneity and unpredictability in a large herbivore. Oecologia 187, 47–60 (2018).
Krebs, J. R. Territory and breeding density in the great tit, Parus major L. Ecology 52, 2–22 (1971).
Brown, J. H. & Munger, J. C. Experimental manipulation of a desert rodent community: Food addition and species removal. Ecology 66, 1545–1563 (1985).
Cagnacci, F., Boitani, L., Powell, R. A. & Boyce, M. S. Animal ecology meets GPS-based radiotelemetry: A perfect storm of opportunities and challenges. Philos. Trans. R. Soc. B Biol. Sci. 365, 2157–2162 (2010).
Kilpatrick, H. J. & Stober, W. A. Effects of temporary bait sites on movements of suburban white-tailed deer. Wildl. Soc. Bull. 30, 760–766 (2002).
van Overveld, T. & Matthysen, E. Personality predicts spatial responses to food manipulations in free-ranging great tits (Parus major). Biol. Lett. 6, 187–190 (2010).
Andersen, R., Gaillard, J.-M., Linnell, J. D. C. & Duncan, P. Factors affecting maternal care in an income breeder, the European roe deer. J. Anim. Ecol. 69, 672–682 (2000).
Ossi, F. et al. Plastic response by a small cervid to supplemental feeding in winter across a wide environmental gradient. Ecosphere 8, e01629 (2017).
Morellet, N. et al. Seasonality, weather and climate affect home range size in roe deer across a wide latitudinal gradient within Europe. J. Anim. Ecol. 82, 1326–1339 (2013).
Peters, W. et al. Migration in geographic and ecological space by a large herbivore. Ecol. Monogr. 87, 297–320 (2017).
De Groeve, J. et al. Individual Movement-Sequence Analysis Method (IM-SAM): Characterizing spatio-temporal patterns of animal habitat use across landscapes. Int. J. Geogr. Inf. Sci. 34, 1530–1551 (2019).
De Groeve, J. et al. Extracting spatio-temporal patterns in animal trajectories: An ecological application of sequence analysis methods. Methods Ecol. Evol. 7, 369–379 (2016).
Ossi, F., Gaillard, J. M., Hebblewhite, M. & Cagnacci, F. Snow sinking depth and forest canopy drive winter resource selection more than supplemental feeding in an alpine population of roe deer. Eur. J. Wildl. Res. 61, 111–124 (2015).
Owen-Smith, N. Functional heterogeneity in resources within landscapes and herbivore population dynamics. Landsc. Ecol. 19, 761–771 (2004).
Rozen-Rechels, D. et al. Density-dependent, central-place foraging in a grazing herbivore: Competition and tradeoffs in time allocation near water. Oikos 124, 1142–1150 (2015).
Holbrook, K. M., Smith, T. B. & Hardesty, B. D. Implications of long-distance movements of frugivorous rain forest hornbills. Ecography (Cop.) 25, 745–749 (2002).
Normand, E., Ban, S. D. & Boesch, C. Forest chimpanzees (Pan troglodytes verus) remember the location of numerous fruit trees. Anim. Cogn. 12, 797–807 (2009).
Janson, C. H. Experimental evidence for spatial memory in foraging wild capuchin monkeys, Cebus apella. Anim. Behav. 55, 1229–1243 (1998).
Linnell, J. D. C. & Andersen, R. Territorial fidelity and tenure in roe deer bucks. Acta Theriol. (Warsz) 43, 67–75 (1998).
Riotte-Lambert, L., Benhamou, S. & Chamaillé-Jammes, S. How memory-based movement leads to nonterritorial spatial segregation. Am. Nat. 185, E103-116 (2015).
Van Moorter, B. et al. Memory keeps you at home: A mechanistic model for home range emergence. Oikos 118, 641–652 (2009).
Fryxell, J. M. et al. Multiple movement modes by large herbivores at multiple spatiotemporal scales. PNAS 105, 19114–19119 (2008).
Eifler, D. A. Experimental manipulation of spacing patterns in the widely foraging lizard Cnemidophorus uniparens. Herpetologica 52, 477–486 (1996).
Ims, R. A. Responses in spatial organization and behaviour to manipulations of the food resource in the vole Clethrionomys rufocanus. J. Anim. Ecol. 56, 585–596 (1987).
Mysterud, A. et al. Partial migration in expanding red deer populations at northern latitudes—A role for density dependence?. Oikos 120, 1817–1825 (2011).
Liberg, O., Johansson, A., Andersen, R. & Linnell, J. D. C. Mating system, mating tactics and the function of male territory in roe deer. In The European Roe Deer: The Biology of Success (eds Andersen, R. et al.) 221–256 (Scandinavian University Press, Oslo, 1998).
Walhström, L. K. & Kjellander, P. Ideal free distribution and natal dispersal in female roe deer. Oecologia 103, 302–308 (1995).
Beyer, H. L. et al. The interpretation of habitat preference metrics under use-availability designs. Philos. Trans. R. Soc. B Biol. Sci. 365, 2245–2254 (2010).
Bonnot, N. et al. Interindividual variability in habitat use: Evidence for a risk management syndrome in roe deer?. Behav. Ecol. 26, 105–114 (2015).
Spiegel, O., Leu, S. T., Bull, C. M. & Sih, A. What’s your move? Movement as a link between personality and spatial dynamics in animal populations. Ecol. Lett. 20, 3–18 (2017).
Cote, J., Clobert, J., Brodin, T., Fogarty, S. & Sih, A. Personality-dependent dispersal: Characterization, ontogeny and consequences for spatially structured populations. Philos. Trans. R. Soc. B Biol. Sci. 365, 4065–4076 (2010).
Piper, W. H. Making habitat selection more “familiar”: A review. Behav. Ecol. Sociobiol. 65, 1329–1351 (2011).
Dalziel, B. D., Morales, J. M. & Fryxell, J. M. Fitting probability distributions to animal movement trajectories: Using artificial neural networks to link distance, resources, and memory. Am. Nat. 172, 248–258 (2008).
Merkle, J. A., Fortin, D. & Morales, J. M. A memory-based foraging tactic reveals an adaptive mechanism for restricted space use. Ecol. Lett. 17, 924–931 (2014).
Van Moorter, B., Visscher, D., Herfindal, I., Basille, M. & Mysterud, A. Inferring behavioural mechanisms in habitat selection studies getting the null-hypothesis right for functional and familiarity responses. Ecography (Cop.) 36, 323–330 (2013).
Tufto, J., Andersen, R. & Linnell, J. Habitat use and ecological correlates of home range size in a small cervid: The roe deer. J. Anim. Ecol. 65, 715–724 (1996).
Bongi, P. et al. Anti-predator behaviour, space use and habitat selection in female roe deer during the fawning season in a wolf area. J. Zool. 276, 242–251 (2008).
Gehr, B. et al. Stay home, stay safe—Site familiarity reduces predation risk in a large herbivore in two contrasting study sites. J. Anim. Ecol. 89, 1329–1339 (2020).
Mancinelli, S., Peters, W., Boitani, L., Hebblewhite, M. & Cagnacci, F. Roe deer summer habitat selection at multiple spatio-temporal scales in an alpine environment. Hystrix 26, 132–140 (2015).
Worton, B. J. Kernel methods for estimating the utilization cistribution in home-range studies. Ecology 70, 164–168 (1989).
Fieberg, J. & Kochany, C. O. Quantifying home-range overlap: The importance of the utilization distribution. J. Wildl. Manag. 69, 1346–1359 (2005).
Fieberg, J., Matthiopoulos, J., Hebblewhite, M., Boyce, M. S. & Frair, J. L. Correlation and studies of habitat selection: Problem, red herring or opportunity?. Philos. Trans. R. Soc. B Biol. Sci. 365, 2233–2244 (2010).
R Development Core Team. R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, Vienna, 2016); www.r-project.org.
Calenge, C. The package ‘adehabitat’ for the R software: A tool for the analysis of space and habitat use by animals. Ecol. Model. 197, 516–519 (2006).
Bates, D., Mächler, M., Bolker, B. & Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 67, 1–48 (2015).
Singmann, H., Bolker, B., Westfall, J. & Aust, F. afex: Analysis of Factorial Experiments. R Package Version 0.22-1. https://CRAN.R-project.org/package=afex. (2018).
Bartón, K. MuMIn: Multi-Model Inference. R Package Version 1.42.1. https://CRANR-project.org/package=MuMIn. (2018).
Acknowledgements
N. Ranc was supported by Harvard University Graduate Fellowship and Fondazione Edmund Mach International Doctoral Programme Fellowship. F. Cagnacci was supported by the Sarah and Daniel Hrdy Fellowship 2015–2016 at Harvard University OEB during part of the development of this manuscript. We are grateful to all the people involved in the roe deer captures and data collection, and especially M.B. Almeida, P. Bonanni, A. Corradini, J. De Groeve, G. Ferrari, G. Lilli, S. Mumme, S. Nicoloso, D. Righetti, M. Rocca, M. Salvatori, M. Sanchez and P. Semenzato. We thank J.W. Cain, B. Ölveczky, N. Pierce, all the members of the Cagnacci lab and of the Moorcroft lab, and the graduate students of the Harvard Statistics Consulting Service for their valuable suggestions. We thank the editor (D. Wang) and three anonymous reviewers for the valuable suggestions and feedback on earlier version of this article. We also thank the Wildlife and Forestry Service of the Autonomous Province of Trento and the Trentino Hunting Association (ACT) for their support. Finally, we express our gratitude to the hunters of Albiano, Civezzano, Fornace and Trento Nord without whose help this study could not be conducted.
Author information
Authors and Affiliations
Contributions
N.R., F.C. and P.R.M. conceived the ideas and designed the methodology; F.C. supervised the field study; N.R. and F.O. conducted the field measurements with support from W.H. and T.S.; A.B. and E.F. provided support for the fieldwork and local expert knowledge; N.R. analysed the data with support from W.H. and T.S.; N.R. led the writing of the manuscript, together with F.C. and P.R.M. P.R.M. and F.C. are co-senior authors. All authors contributed critically to the drafts and gave final approval for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ranc, N., Moorcroft, P.R., Hansen, K.W. et al. Preference and familiarity mediate spatial responses of a large herbivore to experimental manipulation of resource availability. Sci Rep 10, 11946 (2020). https://doi.org/10.1038/s41598-020-68046-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-68046-7
This article is cited by
-
The role of memory-based movements in the formation of animal home ranges
Journal of Mathematical Biology (2024)
-
Memory pays off
Nature Ecology & Evolution (2023)
-
Back and forth: day–night alternation between cover types reveals complementary use of habitats in a large herbivore
Landscape Ecology (2023)
-
Is it the road or the fence? Influence of linear anthropogenic features on the movement and distribution of a partially migratory ungulate
Movement Ecology (2022)
-
Roads constrain movement across behavioural processes in a partially migratory ungulate
Movement Ecology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.