Abstract
As a result of ocean warming, the species composition of the Arctic seas has begun to shift in a boreal direction. One ecosystem prone to fauna shifts is the Northeast Greenland shelf. The dispersal route taken by boreal fauna to this area is, however, not known. This knowledge is essential to predict to what extent boreal biota will colonise Arctic habitats. Using population genetics, we show that Atlantic cod (Gadus morhua), beaked redfish (Sebastes mentella), and deep-sea shrimp (Pandalus borealis) recently found on the Northeast Greenland shelf originate from the Barents Sea, and suggest that pelagic offspring were dispersed via advection across the Fram Strait. Our results indicate that boreal invasions of Arctic habitats can be driven by advection, and that the fauna of the Barents Sea can project into adjacent habitats with the potential to colonise putatively isolated Arctic ecosystems such as Northeast Greenland.
Similar content being viewed by others
Introduction
The Arctic is warming more rapidly than any other geographical region1. Increase in water temperature and loss of sea-ice2,3 are expected to induce a northward range expansion of boreal fauna4,5,6, a phenomenon that is already apparent in the Barents Sea7,8,9. Atlantic mackerel (Scomber scombrus) exemplifies this trend, having recently displayed an exceptional northward shift in distribution to Spitsbergen10. Such predatory newcomers and competitors pose a genuine threat to native Arctic fauna and thus to Arctic ecosystems as they may restructure trophic relationships if their occurrence becomes persistent8,11,12.
In 2015 and 2017, boreal species, i.e. juvenile Atlantic cod (Gadus morhua), juvenile beaked redfish (Sebastes mentella), and adult deep-sea shrimp (Pandalus borealis), were observed on the Northeast (NE) Greenland shelf (latitudes 74–77 °N, Fig. 1) for the first time since surveying began in 200213. This was well outside of their known distribution ranges4,14,15 (Fig. 2a,d,g). However, the route by which the three species had reached NE Greenland was unknown. The present study aims to determine their population of origin using genetic markers. This knowledge will allow us to infer the dispersal routes taken by these species – information that is critical to forecast (e.g.5) which boreal species may disperse into the Arctic and to what extent they may colonise Arctic habitats16.
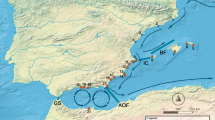
Stations (green full circles) of observation for Atlantic cod (Gadus morhua), beaked redfish (Sebastes mentella) and deep-sea shrimp (Pandalus borealis) (Methods, Table 1). Arrows indicate ocean currents (Source17,63). Atlantic surface currents (red arrows): IMC (Irminger Current), NAC (Norwegian Atlantic Current), WSC (West Spitsbergen Current), RAC (Return Atlantic Current). Atlantic sub-surface water (white arrows). Arctic surface currents (blue arrows): EGC (East Greenland Current), JMC (Jan Mayen Current). Arrow size indicates velocity. Map created using ESRI ArcMap (v. 10.6, www.arcgis.com).
Genetic evidence of Atlantic cod (Gadus morhua) (a–c), beaked redfish (Sebastes mentella) (d–f) and deep-sea shrimp (Pandalus borealis) (g–i) specimens found off Northeast Greenland originating from the Barents Sea. Maps (a,d,g) show species known distribution extent (shaded colours) in the Northeast Atlantic, catch sites of individuals in Northeast Greenland (NEG) waters (full circles), reference samples (hollow circles) and a proposed dispersal route (arrow). DAPC scatterplots (b,e,h) show how the NEG groups relate to the reference populations of the Northeast Atlantic Ocean. DAPC cluster ellipses were set to contain 95% of genotypes. DAPC scatterplots explain 94% (b), 92% (e) and 97% (h) of the total variation observed. STRUCTURE barplots (c,f,i) show membership probabilities (q) for NEG individuals based on the reference populations used. Maps were created using ESRI ArcMap (v. 10.6, www.arcgis.com).
We consider two main routes of dispersal to NE Greenland, either 1) via migration against the East Greenland Current17 from Iceland, or 2) via advection18 from the Barents Sea by the westbound Return Atlantic Current17,19,20 (Fig. 1) across the abyssal plains of the Fram Strait. The Norwegian Atlantic Current, along the Norwegian coast, and the West Spitsbergen Current17,20, along the Barents Sea shelf-break, are already known to advect cod, redfish, and shrimp offspring from the Norwegian coast and the Barents Sea proper to Spitsbergen, east of the Fram Strait21,22,23.
Results
We found that all cod (n = 10), and 95% of redfish (n = 61 out of 64) caught on either the NE Greenland shelf or in the Fram Strait, were genetically assigned to the Barents Sea North East Arctic Cod (NEAC) population (Fig. 2c), and the Norwegian Shallow (NSH) redfish population (Fig. 2f), respectively. All shrimp (n = 40) caught on the NE Greenland shelf, were genetically assigned to the Spitsbergen West (SPW) shrimp population (Fig. 2i). Assignment with STRUCTURE was supported by high membership probabilities (q > 0.8), which suggest that the three species on the NE Greenland shelf all originate from the Barents Sea. The only individuals to assign to an ‘Icelandic’ population were 5% of redfish (3 out of 64).
There was high consistency in the identified population of origin between the assignment methods (STRUCTURE and snapclust), where 90% of cod, 98% of redfish and 75% of shrimp tests formed a consensus (Supplementary S1, Table S1). All individuals of these three species were assigned with a greater probability to the Barents Sea populations than any other reference population.
Discriminant Analysis of Principle Components (DAPC) strongly support the assignment results by clustering the NE Greenland specimens with the corresponding Barents Sea populations (Fig. 2b,e,h). The 95% DAPC cluster ellipses between NE Greenland and the Barents Sea overlapped considerably, though overlap was also evident between the reference populations, most significantly for the cod and shrimp clusters. The redfish and shrimp neighbour-joining trees resulted in the same grouping as the assignment and DAPC analyses, and indicate a Nei’s Distance of <0.02 between the redfish caught in NE Greenland and the Norwegian Shallow population (Fig. 3a). Nei’s Distance was as comparatively low (0.02) between the Norwegian and Icelandic shrimp reference populations as between the shrimp specimens of NE Greenland and the Spitsbergen West population (Fig. 3b).
Discussion
Our results show that the NE Greenland shelf is readily reached by cod, redfish and shrimp from the Barents Sea, probably advected across the Fram Strait by the Return Atlantic Current, supporting recent simulation studies24,25,26. Advection plays an important role in the northward transport of plankton in the Barents Sea, via the West Spitsbergen Current18 and because up to 50% of this water is estimated to cross the Fram Strait27,28,29, the Return Atlantic Current provides a connection between the Barents Sea and the NE Greenland shelf ecosystems. The inflow of Atlantic water to the Barents Sea has doubled since 198030, resulting in a warmer West Spitsbergen Current31. Hence, there is reason to believe that conditions on both sides of the Fram Strait have become more favourable for boreal species in recent years. The copepod Calanus finmarchicus is the major prey for young cod32 and its abundance during the last warm period in the North Atlantic (1920–1965) has likely driven the range expansion of cod and other boreal species33. Low abundances of Calanus finmarchicus were observed on the NE Greenland shelf in autumn 200634, but in light of the West Spitsbergen Current warming, its abundance will likely increase in the Fram Strait and on the NE Greenland shelf35, thus providing ample food for boreal predators.
North East Arctic Cod (NEAC), the population of origin for the NE Greenland specimens, spawns along the Norwegian coast36 (latitudes 62–71 °N) during March and April where pelagic offspring drift by surface currents37 northwards and eastwards into the Barents Sea38. Depending on local wind-forcing, up to 1/3 of 0-group year-classes may advect off the Norwegian and Barents Sea shelf in some years and disperse over the Norwegian Sea37. We suggest that those 0-group cod advected off the shelf by wind-forcing26,39 either outside of their spawning grounds, or at any point until their northern-most report west of Spitsbergen21, are particularly susceptible to cross the Fram Strait by the Return Atlantic Current (Fig. 2a). By October, when cod are >80 mm in total length (TL), they gain motility, descend out of the pelagic layer, and become demersal40. Therefore, for our explanation to hold true, 0-group cod from the Norwegian coast/Barents Sea must advect to the NE Greenland shelf by October of their spawning year. The observations of 0-group cod in the Fram Strait with a genetic signature of the NEAC population, in September 2007 and 2017, demonstrate that this is happening.
Redfish larvae are extruded along the continental shelf break of the Norwegian and Barents Seas from latitudes 64–74 °N between March and June41. Redfish larvae have been observed in Atlantic water west of the continental shelf, and as far north as Spitsbergen23,42. We observed large numbers of 0-group redfish (TL ~40 mm) over the Fram Strait with a genetic signature of the Norwegian Shallow population. Juvenile redfish are pelagic until 40–50 mm TL at age 4–5 months when they gain motility and descend to deeper waters43. We propose that the 0-group redfish found in the Fram Strait in September 2017 were advected north to Spitsbergen along the shelf break by the West Spitsbergen Current, before crossing the Fram Strait by the Return Atlantic Current. The juvenile redfish had then reached the NE Greenland shelf along this route (Fig. 2d) by the time they were 4–5 months old.
Shrimp on the NE Greenland shelf also originated from the Barents Sea (see sampling of25). Shrimp spawn in autumn throughout the Barents Sea and the meroplanktonic larvae hatch in spring. Shrimp larvae are highly-mobile and distribute according to currents until 2–3 months of age when they settle as post-larvae14. We find it more likely that shrimp larvae from the north-west Barents Sea, i.e. Spitsbergen, would reach the NE Greenland shelf than larvae from the northern Norwegian Coast or central-eastern Barents Sea, due to Spitsbergen’s close proximity to the Return Atlantic Current (Fig. 2g).
The NE Greenland shelf ecosystem is severely understudied and biodiversity baselines are fragmentary with no timeline44. It is therefore difficult to establish whether our findings reflect a recent shift driven by ocean warming or constitute a common component of the NE Greenland Shelf fauna. Nonetheless, the Barents Sea is the most productive ecosystem in the NE Atlantic45 and presently supports the historically largest cod population40. In addition, Atlantic herring (Clupea harengus), Atlantic haddock (Melanogrammus aeglefinus) and Atlantic mackerel are nowadays abundant in Spitsbergen waters. Therefore, in the future we could expect to find more boreal species on the NE Greenland shelf, exemplified by a recent observation of capelin (Mallotus villosus) in this area13. The three species studied herein are clearly not exceptional in being capable of entering the NE Greenland shelf. A recent simulation study26 demonstrates that between 2.4% and 12% of 0-group NEAC may be transported northwest along the proposed route (Fig. 2a). Advection therefore has the potential to restructure Arctic ecosystems18 and the route identified here suggests that a boreal faunal invasion of NE Greenland shelf from the Barents Sea is plausible. Trophic relationships are likely to be strongly modified12 as boreal generalists such as cod are favoured by climate scenarios5. Cod feed on polar cod (Boreogadus saida), Arctic seabed fishes and zoobenthos11, and as a figurehead of boreal range expansions into the Arctic, gives a glimpse of what is to come for native Arctic fauna.
This is the first report to disclose the genetic origin of boreal species in Arctic waters and the connection between Atlantic and Arctic ecosystems. Our findings support the hypothesis that cod, redfish, and shrimp disperse from the Barents Sea across the Fram Strait to the NE Greenland shelf. Due to a lack of time series, we cannot conclude if this is a new phenomenon, or not. In any case, predators and food competitors from lower latitudes alter trophic relationships and impact native Arctic fauna and, with a warming ocean in mind, we suggest that the NE Greenland shelf is likely to become populated by a larger proportion of boreal species.
Methods
Specimens of juvenile cod (Gadus morhua, n = 7, TL: 216–443 mm, length-estimated age40: 2–4 years), juvenile redfish (Sebastes mentella, n = 32, TL: 80–172 mm, length-estimated age46: 1–4 years), and adult shrimp (Pandalus borealis, n = 40) were caught via bottom trawl (c.f.14) from 2007 to 2017 on the NE Greenland shelf (latitudes 74–79 °N), well outside of their known distributional range (Table 1). In addition, 0-group cod (n = 3) and 0-group redfish (n = 32) were caught via mid-water trawls (“Harstad” trawl, ~20 min, ~3 Knots) in the Fram Strait (Table 1) and are included in the analysis to support dispersal route hypotheses. Gill or muscle tissue samples from each specimen were preserved at sea in 96% ethanol and stored at −20 °C until further processing. Sampling was conducted using the R/V Helmer Hanssen as part of the TUNU-Programme47. A subset of (0-group) redfish and shrimp was used for genotyping, otherwise, genotyped individuals represent all specimens caught in the area. Access to sampling on the East Greenland shelf was permitted by the Government of Greenland under the remit of the TUNU-Programme at UiT, Norway.
Genotyped reference populations (Table 2) for the Northeast Atlantic were obtained from several studies16,25,48. To ensure all relevant populations of each species in the Northeast Atlantic were well represented, the cod reference populations were supplemented by genotyping a representative cod population from Iceland, following the same procedure as listed below.
To our knowledge, the reference populations represent all the known spawning populations of these species within the NE Atlantic, which are relevant for this study. Cod and redfish are caught sporadically by commercial fishing in the waters around Jan Mayen Island. They are adult individuals fished during winter time (c.f.49), which are suspected to originate from Icelandic and Barents Sea populations (winter feeding migration)50. Therefore, our reference populations should be adequate for assigning the juvenile cod and redfish considered in the present manuscript back to their population of origin.
DNA was isolated from ethanol-fixed gill or muscle tissue using the DNeasy Blood and Tissue Kit (Qiagen, Hilden, Germany) or the E-Z 96 Tissue DNA Kit (Omega Bio-Tek Inc., Norcross, GA, USA) following the manufacturer’s instructions.
Microsatellite loci were arranged in multiplexes (Supplementary Table S2). A total of 10, 13 and 11 microsatellite loci were amplified using polymerase chain reaction (PCR) for cod, redfish and shrimp, respectively. PCR reactions (2.5 μL) contained ca. 1 x Qiagen Multiplex Master Mix, 0.1–1.0 μm primer, and 15–25 ng DNA. The 5′ end on the forward primers was labelled with a fluorescent dye by the manufacturer (Applied Biosystems, Foster City, CA, USA). Amplification was performed in a GeneAmp 2700 or 9700 thermal cycler (Applied Biosystems). PCR profiles were applied as per published protocols16,48,51 (Supplementary S2). PCR products were separated using an ABI 3130XL sequencer and GeneScan 500-LIZ (Applied Biosystems) was used as internal size standard. Alleles were automatically binned using GENEMAPPER software (v. 3.7, Applied Biosystems) and double-checked manually. Negative controls employed for extraction, amplification and fragmentation reported no contamination between samples. Replicates (33%) reported the repeatability and consistency of genotyping to be 100%.
Prior to analysis, reference genotypes that showed no amplification in >10% of loci were removed. This achieved amplification success >98% for each locus. All microsatellite loci were assessed for the presence of potential scoring errors, deviation from Hardy-Weinberg equilibrium (HWE), and non-neutrality (Supplementary S2). As the presence of scoring errors such as null alleles may introduce ambiguity around the true origin of the NE Greenland specimens, we ran analyses under two conditions, (1) removing loci showing potential scoring errors, and (2) inclusive of all loci (Supplementary S2, Table S2). This enabled us to retain loci subject to potential scoring errors where both conditions produced concurrent results, and to therefore minimise the loss of statistical power.
To increase the power of assignment (see Supplementary S3 for evaluation), only individuals with membership coefficients (q) lower/higher than 0.2/0.8 were used to establish reference population datasets (c.f.52, Supplementary S4, Table S4). As weak population differentiation was expected within all datasets, we adopted a conservative approach to infer q53 using a no-admixture model as implemented in the Bayesian clustering method, STRUCTURE (v.2.3.4)54. This approach has been shown not to bias the true structuring in datasets with weak genetic differentiation53. STRUCTURE was run assuming no admixture (NOADMIX = 1), correlated allele frequencies (FREQSCORR = 1) and utilising locality data (LOCPRIOR = 1). The program was run using K = number of reference populations, for 10 iterations, each with a burn-in period and MCMC replicates of 500,000. CLUMPAK55 was used to merge runs (merged barplots: Supplementary S4, Fig. S4), and reported similarity scores >0.95.
STRUCTURE was employed as the principle tool to assign the NE Greenland individuals to previously identified populations. For this, STRUCTURE was run under the assignment mode (POPFLAG = 1), and assumed no admixture (NOADMIX = 1), correlated allele frequencies (FREQSCORR = 1) and utilised locality data (LOCPRIOR = 1). The program was run using K = number of reference populations, for 10 iterations, each with a burn-in period and MCMC replicates of 500,000. CLUMPAK reported run similarity scores > 0.95. STRUCTURE barplots were visualised in R (v. 3.2.3)56 using the pophelper package (v. 2.2.5)57.
The maximum-likelihood clustering tool snapclust58, within the R package adegenet (v. 2.1.1)59, was used to corroborate the membership probabilities output by STRUCTURE. The function snapclust was run without optimization, and priors for the NE Greenland individuals were set to the reference population identified by STRUCTURE as the most probable origin. Runs used zero iterations (max.iter = 0) and membership coefficients were interpreted as output.
As an exploratory tool, Discriminant Analysis of Principle Components (DAPC)60, within the R package adegenet, was used to explore how the NE Greenland individuals relate to the reference populations. DAPC is a geometric clustering method free of HWE and linkage disequilibrium (LD) assumptions, that attempts to maximise the inter-variation between clusters while minimising the intra-variation observed within clusters.
DAPC clusters were set a priori to the number of reference populations plus one, including NE Greenland individuals as part of the DAPC model. The x.val function indicated the number of principle components (PC’s) to retain, but when this method resulted in the selection of too many PC’s, which would lead to overfitting, the optim.a.score function was preferred, based on an initial selection of all PC’s before refinement. All discriminant functions were retained due to the few clusters present (c.f.59).
To identify the genetic distance between the NE Greenland individuals and reference populations, neighbour-joining trees were produced using the aboot function in the R package poppr (v. 2.3.0)61. This method utilised Nei’s Distance62 and 1000 bootstrap replicates. Due to the small sample size of NE Greenland cod, neighbour-joining trees were only produced for redfish (n = 64) and shrimp (n = 40) data.
Pre-analysis testing where loci subject to potential scoring errors were removed from analyses resulted in the same outcome as analysis retaining all loci (Supplementary S5). We therefore suggest that potential scoring errors had little impact on assignment and thus present our final analyses utilising all loci available; 10, 13, and 11 for cod, redfish and shrimp, respectively.
References
IPCC Intergovernmental Panel on Climate Change. Climate Change 2013: The Physical Science Basis. Working Group II Contribution to the IPCC 5th Assessment Report (2013).
Polyakov, I. V. et al. Greater role for Atlantic inflows on sea-ice loss in the Eurasian Basin of the Arctic. Ocean. Science 356(6335), 285–291 (2017).
Smedsrud, L. H., Halvorsen, M. H., Stroeve, J. C., Zhang, R. & Kloster, K. Fram Strait sea ice export variability and September Arctic sea ice extent over the last 80 years. The Cryosphere 11(1), 65–79 (2017).
Drinkwater, K. F. The response of Atlantic cod (Gadus morhua) to future climate change. ICES Journal of Marine Science 62(7), 1327–1337 (2005).
Cheung, W. W. et al. Large‐scale redistribution of maximum fisheries catch potential in the global ocean under climate change. Global Change Biology 16(1), 24–35 (2010).
Hollowed, A. B., Planque, B. & Loeng, H. Potential movement of fish and shellfish stocks from the sub-Arctic to the Arctic Ocean. Fisheries Oceanography 22, 355–370 (2013).
Fossheim, M. et al. Recent warming leads to rapid borealization of fish communities in the Arctic. Nature Climate Change 5, 673–678 (2015).
Kortsch, S., Primicerio, R., Fossheim, M., Dolgov, A. V. & Aschan, M. Climate change alters the structure of arctic marine food webs due to poleward shifts of boreal generalists. Proceedings of the Royal Society B 282(1814), 20151546 (2015).
Haug, T. et al. Future harvest of living resources in the Arctic Ocean north of the Nordic and Barents Seas: a review of possibilities and constraints. Fisheries Research 188, 38–57 (2017).
Mecklenburg, C. W. et al. Marine Fishes of the Arctic Region Volume 1. Conservation of Arctic Flora and Fauna (2018).
Christiansen, J. S. No future for Euro-Arctic ocean fishes? Marine Ecology Progress Series 575, 217–227 (2017).
Frainer, A. et al. Climate-driven changes in functional biogeography of Arctic marine fish communities. Proceedings of the National Academy of Sciences 114(46), 12202–12207 (2017).
Christiansen, J. S. et al. Novel biodiversity baselines outpace models of fish distribution in Arctic waters. The Science of Nature 103, 8 (2016).
Bergström, B. I. The biology of Pandalus. Advances in Marine Biology 38, 55–245 (2000).
Saha, A. et al. Geographic extent of introgression in Sebastes mentella and its effect on genetic population structure. Evolutionary Applications 10(1), 77–90 (2017).
Murphy, E. J. et al. Understanding the structure and functioning of polar pelagic ecosystems to predict the impacts of change. Proceedings of the Royal Society B 283, 20161646 (2016).
Håvik, L. et al. Evolution of the East Greenland current from Fram Strait to Denmark strait: synoptic measurements from summer 2012. Journal of Geophysical Research: Oceans 122(3), 1974–1994 (2017).
Hunt, G. L. Jr. et al. Advection in polar and sub-polar environments: Impacts on high latitude marine ecosystems. Progress in Oceanography 149, 40–81 (2016).
Bourke, R. H., Weigel, A. M. & Paquette, R. G. The westward turning branch of the West Spitsbergen Current. Journal of Geophysical Research: Oceans 93, 14065–14077 (1988).
Eldevik, T. et al. Observed sources and variability of Nordic seas overflow. Nature Geoscience 2(6), 406 (2009).
Foyn, L., von Quillfeldt, C. H. & Olsen, E. Miljø- og ressursbeskrivelse av området Lofoten - Barentshavet [A description of the marine environment and resources in the area Lofoten - Barents Sea]. Fisken og Havet 6, 83 (2002).
Pedersen, O. P., Aschan, M., Rasmussen, T., Tande, K. S. & Slagstad, D. Larval dispersal and mother populations of Pandalus borealis investigated by a Lagrangian particle-tracking model. Fisheries Research 65(1–3), 173–190 (2003).
Drevetnyak, K. & Nedreaas, K. H. Historical movement pattern of juvenile beaked redfish (Sebastes mentella Travin) in the Barents Sea as inferred from long-term research survey series. Marine Biology Research 5(1), 86–100 (2009).
Koszalka, I., LaCasce, J. H., Andersson, M., Orvik, K. A. & Mauritzen, C. Surface circulation in the Nordic Seas from clustered drifters. Deep Sea Research Part I: Oceanographic Research Papers 58(4), 468–485 (2011).
Jorde, P. E. et al. Genetically distinct populations of northern shrimp, Pandalus borealis, in the North Atlantic: adaptation to different temperatures as an isolation factor. Molecular Ecology 24(8), 1742–1757 (2015).
Strand, K. O., Sundby, S., Albretsen, J. & Vikebø, F. B. The Northeast Greenland shelf as a potential habitat for the Northeast Arctic cod. Frontiers in Marine Science 4, 304 (2017).
Rudels, B. On the mass balance of the Polar Ocean, with special emphasis on the Fram Strait. Norsk Polarinstitutt skrifter 188, 1–53 (1987).
Manley, T. O. Branching of Atlantic Water within the Greenland-Spitsbergen Passage: An estimate of recirculation. Journal of Geophysical Research: Oceans 100, 20627–20634 (1995).
de Steur, L., Hansen, E., Mauritzen, C., Beszczynska-Möller, A. & Fahrbach, E. Impact of recirculation on the East Greenland Current in Fram Strait: Results from moored current meter measurements between 1997 and 2009. Deep Sea Research Part I: Oceanographic Research Papers 92, 26–40 (2014).
Oziel, L., Sirven, J. & Gascard, J. C. The Barents Sea frontal zones and water masses variability (1980–2011). Ocean Science 12(1), 169 (2016).
Beszczynska-Möller, A., Fahrbach, E., Schauer, U. & Hansen, E. Variability in Atlantic water temperature and transport at the entrance to the Arctic Ocean, 1997–2010. ICES Journal of Marine Science 69(5), 852–63 (2012).
Sundby, S. Recruitment of Atlantic cod stocks in relation to temperature and advection of copepod populations. Sarsia 85(4), 277–298 (2000).
Drinkwater, K. F. The regime shift of the 1920s and 1930s in the North Atlantic. Progress in Oceanography 68(2–4), 134–151 (2006).
Svensen, C., Seuthe, L., Vasilyeva, Y., Pasternak, A. & Hansen, E. Zooplankton distribution across Fram Strait in autumn: Are small copepods and protozooplankton important? Progress in Oceanography 91(4), 534–544 (2011).
Weydmann, A., Walczowski, W., Carstensen, J. & Kwaśniewski, S. Warming of Subarctic waters accelerates development of a key marine zooplankton Calanus finmarchicus. Global Change Biology 24(1), 172–183 (2018).
Sundby, S. & Nakken, O. Spatial shifts in spawning habitats of Arcto- Norwegian cod related to multidecadal climate oscillations and climate change. ICES Journal of Marine Science 65(6), 953–962 (2008).
Sundby, S., Bjørke, H., Soldal, A. V. & Olsen, S. Mortality rates during the early life stages and year class strength of the Arcto-Norwegian cod (Gadus morhua L.). Rapports et procès- verbaux des Réunions. Conseil permanent international pour l’Exploration de la Mer 191, 351–358 (1989).
Vikebø, F., Sundby, S., Ådlandsvik, B. & Fiksen, Ø. The combined effect of transport and temperature on distribution and growth of larvae and pelagic juveniles of Arcto-Norwegian cod. ICES Journal of Marine Science 62(7), 1375–1386 (2005).
Vikebø, F., Jørgensen, C., Kristiansen, T. & Fiksen, Ø. Drift, growth, and survival of larval Northeast Arctic cod with simple rules of behaviour. Marine Ecology Progress Series 347, 207–220 (2007).
Yaragina, N. A., Aglen, A., & Sokolov, K. M. Cod. In: Jakobsen, T. & Ozhigin, V. K. (ed.) The Barents Sea ecosystem, resources, management, Half a Century of Russian–Norwegian Cooperation. Tapir Academic Press, Trondheim, Norway 225–270 (2011).
Cadrin, S. X. et al. Population structure of beaked redfish, Sebastes mentella: evidence of divergence associated with different habitats. ICES Journal of Marine Science 67, 1617–1630 (2010).
Hylen, A., Korsbrekke, K., Nakken, O. & Ona, E. Comparison of the capture efficiency of 0-group fish in the pelagic trawls. In: Hylen, A. (ed.) Precision & relevance of pre-recruit studies for fishery management related to fish stocks in the Barents Sea and adjacent waters, proceedings of the sixth IMR-PINRO symposium, Institute of Marine Research, Bergen, Norway, 145–156 (1995).
Kelly, G. F. & Barker, A. M. Vertical distribution of young redfish in the Gulf of Maine. ICNAF Special Publications. 3, 220–233 (1961).
Christiansen, J. S., Mecklenburg, C. W. & Karamushko, O. V. Arctic marine fishes and their fisheries in light of global change. Global Change Biology 20(2), 352–359 (2014).
Wassmann, P. Arctic marine ecosystems in an era of rapid climate change. Progress in Oceanography 90(1–4), 1–17 (2011).
Saborido-Rey, F., Garabana, D. & Cervino, S. Age and growth of redfish (Sebastes marinus, S. mentella, and S. fasciatus) on the Flemish Cap (Northwest Atlantic). ICES Journal of Marine Science 61(2), 231–242 (2004).
Christiansen, J. S. The TUNU-Programme: Euro-Arctic marine fishes—diversity and adaptation. In: di Prisco, G. & Verde, C (ed.) Adaptation and evolution in marine environments, vol. 1, From pole to pole. Springer, Berlin Heidelberg, Germany 35–50 (2012).
Westgaard, J. I. & Fevolden, S. E. Atlantic cod (Gadus morhua L.) in inner and outer coastal zones of northern Norway display divergent genetic signature at non neutral loci. Fisheries Research 85(3), 306–315 (2007).
Wienerroither, R. M. et al. The marine fishes of Jan Mayen Island, NE. Atlantic–past and present. Marine Biodiversity 41(3), 395–411 (2011).
Iversen, T. Sydøstgrønland, Jan Mayen: fiskeriundersøkelser. Fiskeridirektoratets Skrifter Serie Havundersøkelser 5(1), 179 (1936).
Pereyra, R. T. et al. Isolation and characterization of nuclear microsatellite loci in the northern shrimp, Pandalus borealis. Conservation Genetics Resources 4(1), 109–112 (2012).
Vähä, J. P. & Primmer, C. R. Efficiency of model-based Bayesian methods for detecting hybrid individuals under different hybridization scenarios and with different numbers of loci. Molecular Ecology 15(1), 63–72 (2006).
Hubisz, M. J., Falush, D., Stephens, M. & Pritchard, J. K. Inferring weak population structure with the assistance of sample group information. Molecular Ecololgy Resources 9, 1322–1332 (2009).
Pritchard, J. K., Stephens, M. & Donnelly, P. Inference of population structure using multilocus genotype data. Genetics 155(2), 945–959 (2000).
Kopelman, N. M., Mayzel, J., Jakobsson, M., Rosenberg, N. A. & Mayrose, I. Clumpak: a program for identifying clustering modes and packaging population structure inferences across K. Molecular Ecology Resources 15(5), 1179–1191 (2015).
R Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. Available at, www.r-project.org (2018).
Francis, R. M. Pophelper: an R package and web app to analyse & visualize population structure. Molecular Ecology Resources 17(1), 27–32 (2017).
Beugin, M. P., Gayet, T., Pontier, D., Devillard, S. & Jombart, T. A fast likelihood solution to the genetic clustering problem. Methods in Ecology and Evolution 9(4), 1006–1016 (2018).
Jombart, T. adegenet: a R package for the multivariate analysis of genetic markers. Bioinformatics 24(11), 1403–1405 (2008).
Jombart, T., Devillard, S. & Balloux, F. Discriminant analysis of principal components: a new method for the analysis of genetically structured populations. BMC Genetics 11(1), 94 (2010).
Kamvar, Z. N., Brooks, J. C. & Grünwald, N. J. Novel R tools for analysis of genome-wide population genetic data with emphasis on clonality. Frontiers in Genetics 6, 208 (2015).
Nei, M. Molecular Evolutionary Genetics. Columbia University Press, New York, USA (1987).
Koltermann, K. P. & Luethje, H. Hydrographic atlas of the Greenland and northern Norwegian Seas (1979–1987) Deutsches Hydrographisches Institut, Hamburg, Germany (1989).
Acknowledgements
Thanks are extended to the Government of Greenland and the crew on board R/V Helmer Hanssen for access to the Greenland Sea. We thank Tanja L. Hanebrekke for laboratory assistance and Oleg V. Karamushko for fish data. This study is part of the TUNU-Programme, UiT The Arctic University of Norway, and was part-funded by a Small Research Grant from The Fisheries Society of the British Isles. The publication charges for this article have been funded by a grant from the publication fund of UiT The Arctic University of Norway.
Author information
Authors and Affiliations
Contributions
K.P. conceived the study. A.J.A., J.S.C., S.B., A.L., C.P. and K.P. collected tissues for analysis. A.J.A., J.S.C. and K.P. designed the analysis. J.S.C., A.L., K.P. and J.I.W. contributed data. A.J.A., S.B. and K.P. performed the analysis. A.J.A., J.S.C. and K.P. wrote the manuscript. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Andrews, A.J., Christiansen, J.S., Bhat, S. et al. Boreal marine fauna from the Barents Sea disperse to Arctic Northeast Greenland. Sci Rep 9, 5799 (2019). https://doi.org/10.1038/s41598-019-42097-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-42097-x
This article is cited by
-
North Atlantic deep-sea benthic biodiversity unveiled through sponge natural sampler DNA
Communications Biology (2024)
-
Occurrence and patterns of fin whale songs reveal alternative migration strategies in Svalbard Islands, Norway
Scientific Reports (2023)
-
Jan Mayen—a new spawning and fishing area for Atlantic cod Gadus morhua
Polar Biology (2023)
-
Vertical redistribution of principle water masses on the Northeast Greenland Shelf
Nature Communications (2022)
-
Spatial occurrence and abundance of marine zooplankton in Northeast Greenland
Marine Biodiversity (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.