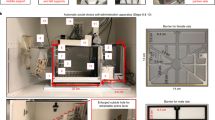
Abstract
Little is known about how social factors contribute to neurobiology or neuropsychiatric disorders. The use of mice allows one to probe the neurobiological bases of social interaction, offering the genetic diversity and versatility to identify cell types and neural circuits of social behavior. However, mice typically show lower social motivation compared with rats, leading to the question of whether mice should be used to model complex social behaviors displayed by humans. Studies on mouse social behavior often rely on measures such as time spent in contact with a social partner or preference for a social-paired context, but fail to assess volitional (subject-controlled) rewarding social interaction. Here, we describe a volitional social self-administration and choice model that is an extension of our previous work on rats. Using mice, we systematically compared female adolescent and adult C57BL/6 mice and outbred CD1 mice, showing that operant social self-administration, social seeking during periods of isolation and choice of social interaction over palatable food is significantly stronger in female CD1 mice than in female C57BL/6J mice, independently of age. We describe the requirements for building the social self-administration and choice apparatus and we provide guidance for studying the role of operant social reward in mice. We also discuss its use to study brain mechanisms of operant social reward, potentially extending its application to mouse models of neuropsychiatric disorders. The training commonly requires ~4 weeks for stable social self-administration and 3–4 additional weeks for tests, including social seeking and choice.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The main data discussed in this protocol are available in the supporting primary research paper (https://doi.org/10.1016/j.biopsych.2021.10.023)
Code availability
The Med-Associates programs are available upon request of the corresponding authors (L.A.R. and M.V.). Files necessary for 3D printing the partner chamber are provided in the Supplementary Software.
References
Heilig, M., Epstein, D. H., Nader, M. A. & Shaham, Y. Time to connect: bringing social context into addiction neuroscience. Nat. Rev. Neurosci. 17, 592–599 (2016).
Vanderschuren, L. J., Achterberg, E. J. & Trezza, V. The neurobiology of social play and its rewarding value in rats. Neurosci. Biobehav. Rev. 70, 86–105 (2016).
Yizhar, O. & Levy, D. R. The social dilemma: prefrontal control of mammalian sociability. Curr. Opin. Neurobiol. 68, 67–75 (2021).
Venniro, M., Banks, M. L., Heilig, M., Epstein, D. H. & Shaham, Y. Improving translation of animal models of addiction and relapse by reverse translation. Nat. Rev. Neurosci. 21, 625–643 (2020).
Venniro, M., Russell, T. I., Zhang, M. & Shaham, Y. Operant social reward decreases incubation of heroin craving in male and female rats. Biol. Psychiatry 86, 848–856 (2019).
Venniro, M. & Shaham, Y. An operant social self-administration and choice model in rats. Nat. Protoc. 15, 1542–1559 (2020).
Venniro, M. et al. Volitional social interaction prevents drug addiction in rat models. Nat. Neurosci. 21, 1520–1529 (2018).
Venniro, M., Panlilio, L. V., Epstein, D. H. & Shaham, Y. The protective effect of operant social reward on cocaine self-administration, choice, and relapse is dependent on delay and effort for the social reward. Neuropsychopharmacology https://doi.org/10.1038/s41386-021-01148-6 (2021).
Venniro, M. et al. Abstinence-dependent dissociable central amygdala microcircuits control drug craving. Proc. Natl Acad. Sci. USA 117, 8126–8134 (2020).
Ellenbroek, B. & Youn, J. Rodent models in neuroscience research: is it a rat race? Dis. Model Mech. 9, 1079–1087 (2016).
Dolen, G., Darvishzadeh, A., Huang, K. W. & Malenka, R. C. Social reward requires coordinated activity of nucleus accumbens oxytocin and serotonin. Nature 501, 179–184 (2013).
Felix-Ortiz, A. C. & Tye, K. M. Amygdala inputs to the ventral hippocampus bidirectionally modulate social behavior. J. Neurosci. 34, 586–595 (2014).
Gunaydin, L. A. et al. Natural neural projection dynamics underlying social behavior. Cell 157, 1535–1551 (2014).
Levy, D. R. et al. Dynamics of social representation in the mouse prefrontal cortex. Nat. Neurosci. 22, 2013–2022 (2019).
Nardou, R. et al. Oxytocin-dependent reopening of a social reward learning critical period with MDMA. Nature 569, 116–120 (2019).
Wei, D. et al. Endocannabinoid signaling mediates oxytocin-driven social reward. Proc. Natl Acad. Sci. USA 112, 14084–14089 (2015).
Yizhar, O. et al. Neocortical excitation/inhibition balance in information processing and social dysfunction. Nature 477, 171–178 (2011).
Netser, S. et al. Distinct dynamics of social motivation drive differential social behavior in laboratory rat and mouse strains. Nat. Commun. 11, 5908 (2020).
Poole, T. B. & Fish, J. An investigation of playful behaviour in Rattus norvegicus and Mus musculus (Mammalia). J. Zool. Soc. Lond. 175, 61–71 (1975).
Trezza, V., Campolongo, P. & Vanderschuren, L. J. Evaluating the rewarding nature of social interactions in laboratory animals. Dev. Cogn. Neurosci. 1, 444–458 (2011).
Hu, R. K. et al. An amygdala-to-hypothalamus circuit for social reward. Nat. Neurosci. https://doi.org/10.1038/s41593-021-00828-2 (2021).
Martin, L. & Iceberg, E. Quantifying social motivation in mice using operant conditioning. J. Vis. Exp. https://doi.org/10.3791/53009 (2015).
Brodkin, E. S., Hagemann, A., Nemetski, S. M. & Silver, L. M. Social approach-avoidance behavior of inbred mouse strains towards DBA/2 mice. Brain Res. 1002, 151–157 (2004).
Panksepp, J. B. et al. Affiliative behavior, ultrasonic communication and social reward are influenced by genetic variation in adolescent mice. PLoS ONE 2, e351 (2007).
Panksepp, J. B. & Lahvis, G. P. Social reward among juvenile mice. Genes Brain Behav. 6, 661–671 (2007).
Golden, S. A. et al. Compulsive addiction-like aggressive behavior in mice. Biol. Psychiatry 82, 239–248 (2017).
Golden, S. A. et al. Nucleus accumbens Drd1-expressing neurons control aggression self-administration and aggression seeking in mice. J. Neurosci. 39, 2482–2496 (2019).
Aleyasin, H. et al. Cell-type-specific role of DeltaFosB in nucleus accumbens in modulating intermale aggression. J. Neurosci. 38, 5913–5924 (2018).
Ramsey, L. A., Holloman, F. M., Hope, B. T., Shaham, Y. & Venniro, M. Waving through the window: A model of volitional social interaction in female mice. Biol. Psychiatry https://doi.org/10.1016/j.biopsych.2021.10.023 (2022).
Miczek, K. A., Maxson, S. C., Fish, E. W. & Faccidomo, S. Aggressive behavioral phenotypes in mice. Behav. Brain Res. 125, 167–181 (2001).
Cann, C., Venniro, M., Hope, B. T. & Ramsey, L. A. Parametric investigation of social place preference in adolescent mice. Behav. Neurosci. 134, 435–443 (2020).
Kummer, K. K. et al. Differences in social interaction- vs. cocaine reward in mouse vs. rat. Front. Behav. Neurosci. 8, 363 (2014).
Pierce, R. C., Crawford, C. A., Nonneman, A. J., Mattingly, B. A. & Bardo, M. T. Effect of forebrain dopamine depletion on novelty-induced place preference behavior in rats. Pharmacol. Biochem. Behav. 36, 321–325 (1990).
Joel, D. & McCarthy, M. M. Incorporating sex as a biological variable in neuropsychiatric research: where are we now and where should we be? Neuropsychopharmacology 42, 379–385 (2017).
Beery, A. K. & Zucker, I. Sex bias in neuroscience and biomedical research. Neurosci. Biobehav. Rev. 35, 565–572 (2011).
Hughes, R. N. Sex does matter: comments on the prevalence of male-only investigations of drug effects on rodent behaviour. Behav. Pharmacol. 18, 583–589 (2007).
Golden, S. A. & Shaham, Y. Aggression addiction and relapse: a new frontier in psychiatry. Neuropsychopharmacology 43, 224–225 (2018).
Richardson, N. R. & Roberts, D. C. Progressive ratio schedules in drug self-administration studies in rats: a method to evaluate reinforcing efficacy. J. Neurosci. Methods 66, 1–11 (1996).
Venniro, M., Caprioli, D. & Shaham, Y. Animal models of drug relapse and craving: from drug priming-induced reinstatement to incubation of craving after voluntary abstinence. Prog. Brain Res. 224, 25–52 (2016).
Shalev, U., Grimm, J. W. & Shaham, Y. Neurobiology of relapse to heroin and cocaine seeking: a review. Pharmacol. Rev. 54, 1–42 (2002).
Venniro, M. et al. Factors modulating the incubation of drug and non-drug craving and their clinical implications. Neurosci. Biobehav. Rev. 131, 847–864 (2021).
Acknowledgements
We thank E. Hart for his help with 3D printing. We thank F. Vautier and the NIDA Transgenic Breeding Facility for assistance in breeding the FosGFP × CD1 hybrid mice. We thank Drs. Y. Shaham and B. Hope for input and support in developing the model. The research was supported by the NIDA Intramural Research Program (L.A.R.), a grant from NIDA (DA047976) (M.V.) and a BBRF Young Investigator Grant Award (28897; M.V.).
Author information
Authors and Affiliations
Contributions
L.A.R., F.M.H., S.S.L. and M.V. contributed to various aspects of the study, including the design and performance of the research and the writing of the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Protocols thanks Loren Martin and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
Ramsey, L. A. et al. Biol. Psychiatry 91, 988–997 (2022): https://doi.org/10.1016/j.biopsych.2021.10.023
Venniro, M. & Shaham, Y. Nat. Prot. 15, 1542–1559 (2020): https://doi.org/10.1038/s41596-020-0296-6
This protocol is an extension to: Nat. Protoc. 15, 1542–1559 (2020): https://doi.org/10.1038/s41596-020-0296-6
Supplementary information
Supplementary Software 1
Design file template for partner chamber (Partner chamber.stl). 2. Design file template for partner door (Partner door.stl). 3. Design file template for bedding box (bedding box partner chamber.stl). 4. Design file template for bedding box [modifiable] (bedding box partner chamber.gcode). 5. Design file template for partner chamber door [modifiable] (Door partner chamber 1 box.form). 6. Design file template for Partner chamber door [modifiable] (Door partner chamber 3 box.form).
Rights and permissions
About this article
Cite this article
Ramsey, L.A., Holloman, F.M., Lee, S.S. et al. An operant social self-administration and choice model in mice. Nat Protoc 18, 1669–1686 (2023). https://doi.org/10.1038/s41596-023-00813-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-023-00813-y
This article is cited by
-
Social odor choice buffers drug craving
Neuropsychopharmacology (2024)
-
Operant social self-administration in male CD1 mice
Psychopharmacology (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.