Abstract
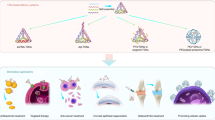
An active targeting strategy-enabled DNA tetrahedron delivery vehicle could facilitate stable drug encapsulation and stimuli-responsive on-demand release, building a universal platform for different drug delivery requirements. Owing to the excellent biocompatible nature, programmability and remarkable cell and tissue permeability, the tetrahedral DNA nanostructure (TDN) has proven its value in the delivery of various bioactive molecules. We previously described this as a static multifunctional complex in our earlier protocol. However, static structures and passive targeting behavior might introduce off-target effects under complicated biological conditions. Therefore, in this Protocol Extension, we present a major update of the TDN delivery vehicle enabling an active targeting strategy to be used for stimuli-sensitive conformation changes and on-site cargo release, which could avoid drawbacks, including complex and time-consuming fabrication processes and undetermined cell penetration ability of other DNA-based delivery vehicles. Upon exquisite design of TDN size based on cargo type, one-pot annealing is applied to fabricate the Tiamat-designed TDN exoskeleton. Then the design of the dynamic DNA apparatus can be based on the target and environmental stimuli, including DNA strand hybridization-based and pH-sensitive DNA apparatus, and careful titration of strand lengths and mismatches is achieved using polyacrylamide and agarose gel electrophoresis, or fluorophore modifications. Finally, cargo loading strategies are designed, including site and stand titration and cargo encapsulation verification. The dynamic structures show promising targetability and effectiveness in antitumor and anti-inflammatory treatment in vitro and in vivo. Assembly and characterization in the lab takes ~5 d, and the timing for the verification of biostability and biological applications depends on the uses.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Seeman, N. C. Nucleic acid nanostructures and topology. Angew. Chem. Int. Ed. Engl. 37, 3220–3238 (1998).
Zhu, J. et al. antiepilepticus effects of tetrahedral framework nucleic acid via inhibition of gliosis-induced downregulation of glutamine synthetase and increased AMPAR internalization in the postsynaptic membrane. Nano Lett. 22, 2381–2390 (2022).
Wang, Y. et al. Tetrahedral framework nucleic acids can alleviate taurocholate-induced severe acute pancreatitis and its subsequent multiorgan injury in mice. Nano Lett. 22, 1759–1768 (2022).
Yan, J. et al. Redox-responsive polyethyleneimine/tetrahedron DNA/doxorubicin nanocomplexes for deep cell/tissue penetration to overcome multidrug resistance. J. Control. Release 329, 36–49 (2021).
Zhang, T. et al. Design, fabrication and applications of tetrahedral DNA nanostructure-based multifunctional complexes in drug delivery and biomedical treatment. Nat. Protoc. 15, 2728–2757 (2020).
Zhang, Y. et al. Inhibiting methicillin-resistant Staphylococcus aureus by tetrahedral DNA nanostructure-enabled antisense peptide nucleic acid delivery. Nano Lett. 18, 5652–5659 (2018).
Ma, W. et al. An intelligent DNA nanorobot with in vitro enhanced protein lysosomal degradation of HER2. Nano Lett. 19, 4505–4517 (2019).
Li, S. et al. Bioswitchable delivery of microRNA by framework nucleic acids: application to bone regeneration. Small 17, e2104359 (2021).
Zhang, X. et al. Tetrahedral-framework nucleic acids carry small interfering RNA to downregulate toll-like receptor 2 gene expression for the treatment of sepsis. ACS Appl. Mater. Interfaces 14, 6442–6452 (2022).
Li, J. et al. Modulation of the crosstalk between Schwann cells and macrophages for nerve regeneration: a therapeutic strategy based on a multifunctional tetrahedral framework nucleic acids system. Adv. Mater. 28, 2202513 (2022).
Ma, W. et al. Biomimetic nanoerythrosome-coated aptamer–DNA tetrahedron/maytansine conjugates: pH-responsive and targeted cytotoxicity for HER2-positive breast cancer. Adv. Mater. 34, 2109609 (2022).
Fu, W. et al. Therapeutic siCCR2 loaded by tetrahedral framework DNA nanorobotics in therapy for intracranial hemorrhage. Adv. Funct. Mater. 31, 2101435 (2021).
Liu, Y. et al. Tetrahedral framework nucleic acids deliver antimicrobial peptides with improved effects and less susceptibility to bacterial degradation. Nano Lett. 20, 3602–3610 (2020).
Zhang, M. et al. Anti-inflammatory activity of curcumin-loaded tetrahedral framework nucleic acids on acute gouty arthritis. Bioact. Mater. 8, 368–380 (2022).
Zhang, T., Tian, T. & Lin, Y. Functionalizing framework nucleic-acid-based nanostructures for biomedical application. Adv. Mater. 17, 2107820 (2022).
He, Z. et al. Gold nanorods/tetrahedral DNA composites for chemo-photothermal therapy. Regen. Biomater. 9, rbac032 (2022).
Tian, T. et al. A framework nucleic acid based robotic nanobee for active targeting therapy. Adv. Funct. Mater. 31, 2007342 (2020).
Gao, Y. et al. A lysosome-activated tetrahedral nanobox for encapsulated siRNA delivery. Adv. Mater. 5, e2201731 (2022).
Xie, X. et al. Overcoming drug-resistant lung cancer by paclitaxel loaded tetrahedral DNA nanostructures. Nanoscale 10, 5457–5465 (2018).
Shi, S. et al. Effects of tetrahedral framework nucleic acid/wogonin complexes on osteoarthritis. Bone Res. 8, 6 (2020).
Madhanagopal, B. R., Zhang, S., Demirel, E., Wady, H. & Chandrasekaran, A. R. DNA nanocarriers: programmed to deliver. Trends Biochem. Sci. 43, 997–1013 (2018).
Li, Y. et al. Tetrahedral framework nucleic acid-based delivery of resveratrol alleviates insulin resistance: from innate to adaptive immunity. Nanomicro Lett. 13, 86 (2021).
Qin, X. et al. Tetrahedral framework nucleic acids-based delivery of microRNA-155 inhibits choroidal neovascularization by regulating the polarization of macrophages. Bioact. Mater. 14, 134–144 (2022).
Li, J. et al. The neuroprotective effect of microRNA-22-3p modified tetrahedral framework nucleic acids on damaged retinal neurons via TrkB/BDNF signaling pathway. Adv. Funct. Mater. 31, 2104141 (2021).
Lee, H. et al. Molecularly self-assembled nucleic acid nanoparticles for targeted in vivo siRNA delivery. Nat. Nanotechnol. 7, 389–393 (2012).
Liang, L. et al. Single-particle tracking and modulation of cell entry pathways of a tetrahedral DNA nanostructure in live cells. Angew. Chem. Int. Ed. Engl. 53, 7745–7750 (2014).
Wiraja, C. et al. Framework nucleic acids as programmable carrier for transdermal drug delivery. Nat. Commun. 10, 1147 (2019).
Zhou, M. et al. A DNA nanostructure-based neuroprotectant against neuronal apoptosis via inhibiting Toll-like receptor 2 signaling pathway in acute ischemic stroke. ACS Nano 16, 1456–1470 (2022).
Li, Q. et al. Aptamer-modified tetrahedral DNA nanostructure for tumor-targeted drug delivery. ACS Appl. Mater. Interfaces 9, 36695–36701 (2017).
Cui, W. et al. Treating LRRK2-related Parkinson’s disease by inhibiting the mTOR signaling pathway to restore autophagy. Adv. Funct. Mater. 31, 2105152 (2021).
Sun, Y. et al. Erythromycin loaded by tetrahedral framework nucleic acids are more antimicrobial sensitive against Escherichia coli (E. coli). Bioact. Mater. 6, 2281–2290 (2021).
Gao, P. et al. Antitumor agents based on metal–organic frameworks. Angew. Chem. Int. Ed. Engl. 60, 16763–16776 (2021).
Wu, M. X. & Yang, Y. W. Metal–organic framework (MOF)-based drug/cargo delivery and cancer therapy. Adv. Mater. 29, 1606134 (2017).
Li, S. et al. A DNA nanorobot functions as a cancer therapeutic in response to a molecular trigger in vivo. Nat. Biotechnol. 36, 258–264 (2018).
Bhatia, D., Surana, S., Chakraborty, S., Koushika, S. P. & Krishnan, Y. A synthetic icosahedral DNA-based host–cargo complex for functional in vivo imaging. Nat. Commun. 2, 339 (2011).
Ma, W. et al. Biomimetic nanoerythrosome-coated aptamer-DNA tetrahedron/maytansine conjugates: pH-responsive and targeted cytotoxicity for HER2-positive breast cancer. Adv. Mater. 22, e2109609 (2022).
Banerjee, A. et al. Controlled release of encapsulated cargo from a DNA icosahedron using a chemical trigger. Angew. Chem. Int. Ed. Engl. 52, 6854–6857 (2013).
Andersen, E. S. et al. Self-assembly of a nanoscale DNA box with a controllable lid. Nature 459, 73–U75 (2009).
Ranjbar, R. & Hafezi-Moghadam, M. S. Design and construction of a DNA origami drug delivery system based on MPT64 antibody aptamer for tuberculosis treatment. Electron. Physician 8, 1857–1864 (2016).
Tan, X. et al. Blurring the role of oligonucleotides: spherical nucleic acids as a drug delivery vehicle. J. Am. Chem. Soc. 138, 10834–10837 (2016).
Rajwar, A. et al. Geometry of a DNA nanostructure influences its endocytosis: Cellular study on 2D, 3D, and in vivo systems. ACS Nano 16, 10496–10508 (2022).
Zhang, B. et al. Facilitating in-situ tumor imaging with a tetrahedral DNA framework-enhanced hybridization chain reaction probe. Adv. Funct. Mater. 32, 2109728 (2022).
Zhang, T., Tian, T. & Lin, Y. Functionalizing framework nucleic-acid-based nanostructures for biomedical application. Adv. Mater. 34, 2107820 (2022).
Shi, S. et al. Biological effect of differently sized tetrahedral framework nucleic acids: Endocytosis, proliferation, migration, and biodistribution. ACS Appl. Mater. Interfaces 13, 57067–57074 (2021).
Tian, T., Li, Y. & Lin, Y. Prospects and challenges of dynamic DNA nanostructures in biomedical applications. Bone Res. 10, 40 (2022).
Sefah, K., Shangguan, D., Xiong, X., O’Donoghue, M. B. & Tan, W. Development of DNA aptamers using Cell-SELEX. Nat. Protoc. 5, 1169–1185 (2010).
Xing, H., Wong, N. Y., Xiang, Y. & Lu, Y. DNA aptamer functionalized nanomaterials for intracellular analysis, cancer cell imaging and drug delivery. Curr. Opin. Chem. Biol. 16, 429–435 (2012).
Hirao, I., Kimoto, M. & Lee, K. H. DNA aptamer generation by ExSELEX using genetic alphabet expansion with a mini-hairpin DNA stabilization method. Biochimie 145, 15–21 (2018).
Cansiz, S. et al. DNA aptamer based nanodrugs: molecular engineering for efficiency. Chem. Asian J. 10, 2084–2094 (2015).
Lin, M. et al. Electrochemical detection of nucleic acids, proteins, small molecules and cells using a DNA-nanostructure-based universal biosensing platform. Nat. Protoc. 11, 1244–1263 (2016).
Skolakova, P. et al. Systematic investigation of sequence requirements for DNA i-motif formation. Nucleic Acids Res. 47, 2177–2189 (2019).
Abou Assi, H., Garavis, M., Gonzalez, C. & Damha, M. J. i-Motif DNA: structural features and significance to cell biology. Nucleic Acids Res. 46, 8038–8056 (2018).
Guo, K. et al. Formation of pseudosymmetrical G-quadruplex and i-motif structures in the proximal promoter region of the RET oncogene. J. Am. Chem. Soc. 129, 10220–10228 (2007).
Chu, B., Zhang, D. & Paukstelis, P. J. A DNA G-quadruplex/i-motif hybrid. Nucleic Acids Res. 47, 11921–11930 (2019).
Shi, S. et al. Targeted and effective glioblastoma therapy via aptamer-modified tetrahedral framework nucleic acid-paclitaxel nanoconjugates that can pass the blood brain barrier. Nanomed 21, 102061 (2019).
Tucker, W. O., Shum, K. T. & Tanner, J. A. G-quadruplex DNA aptamers and their ligands: structure, function and application. Curr. Pharm. Des. 18, 2014–2026 (2012).
Mondragón-Sánchez, J. A., Santamaria, R. & Garduño-Juárez, R. Docking on the DNA G-quadruplex: a molecular electrostatic potential study. Biopolymers 95, 641–650 (2011).
Williams, B. A. et al. Creating protein affinity reagents by combining peptide ligands on synthetic DNA scaffolds. J. Am. Chem. Soc. 131, 17233–17241 (2009).
Liu, H. et al. Kinetics of RNA and RNA:DNA hybrid strand displacement. ACS Synth. Biol. 10, 3066–3073 (2021).
Erben, C. M., Goodman, R. P. & Turberfield, A. J. Single-molecule protein encapsulation in a rigid DNA cage. Angew. Chem. Int. Ed. Engl. 45, 7414–7417 (2006).
Ke, Y., Bellot, G., Voigt, N. V., Fradkov, E. & Shih, W. M. Two design strategies for enhancement of multilayer-DNA-origami folding: underwinding for specific intercalator rescue and staple-break positioning. Chem. Sci. 3, 2587–2597 (2012).
Li, S. et al. A tetrahedral framework DNA-based bioswitchable miRNA inhibitor delivery system: application to skin anti-aging. Adv. Mater. 28, e2204287 (2022).
Xing, S. et al. Constructing higher-order DNA nanoarchitectures with highly purified DNA nanocages. ACS Appl. Mater. Interfaces 7, 13174–13179 (2015).
Acknowledgements
This study was supported by the National Key R&D Program of China (2019YFA0110600), National Natural Science Foundation of China (81970916, 81671031, 81800947 and 82101077), Sichuan Science and Technology Program (2022JDTD0021 and 2022JDRC0143) and Postdoctoral Science Foundation of China (2020T130443, 2021M702331, 2021TQ0224 and 2021M692271), and Research Funding from West China School/Hospital of Stomatology Sichuan University (RCDWJS2021-20 and RCDWJS2022-12).
Author information
Authors and Affiliations
Contributions
Y.L. supervised and conceived of the study. T.T., Y.G., S.S. and T.Z. designed the TDN-based delivery system and completed the corresponding experiments. T.Z., T.T., S.S., X.C. and Y. G. prepared the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Protocols thanks Dhiraj Bhatia, Chunhai Fan and Bin He for their contribution to the peer review of this work.
Additional information
Related links
Key references using this protocol
Tian, T. T. et al. Adv. Funct. Mater. 31, 2007342 (2021): https://doi.org/10.1002/adfm.202007342
Gao, Y. et al. Adv. Mater. 34, 2201731 (2022): https://doi.org/10.1002/adma.202201731
This protocol is an extension to: Nat. Protoc. 15, 2728–2757 (2020): https://doi.org/10.1038/s41596-020-0355-z
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tian, T., Zhang, T., Shi, S. et al. A dynamic DNA tetrahedron framework for active targeting. Nat Protoc 18, 1028–1055 (2023). https://doi.org/10.1038/s41596-022-00791-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-022-00791-7
This article is cited by
-
Tetrahedral framework nucleic acids for improving wound healing
Journal of Nanobiotechnology (2024)
-
DNA framework signal amplification platform-based high-throughput systemic immune monitoring
Signal Transduction and Targeted Therapy (2024)
-
Tetrahedral framework nucleic acids/hyaluronic acid-methacrylic anhydride hybrid hydrogel with antimicrobial and anti-inflammatory properties for infected wound healing
International Journal of Oral Science (2024)
-
A DNA tetrahedron-based ferroptosis-suppressing nanoparticle: superior delivery of curcumin and alleviation of diabetic osteoporosis
Bone Research (2024)
-
Cell unit-inspired natural nano-based biomaterials as versatile building blocks for bone/cartilage regeneration
Journal of Nanobiotechnology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.