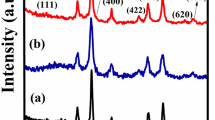
Abstract
Magnetic nanoparticles are increasingly used in medical applications, including cancer treatment by magnetic hyperthermia. This protocol describes a solvothermal-based process to prepare, at the gram scale, ferrite nanoparticles with well-defined shape, i.e., nanocubes, nanostars and other faceted nanoparticles, and with fine control of structural/magnetic properties to achieve point-of-reference magnetic hyperthermia performance. This straightforward method comprises simple steps: (i) making a homogeneous alcoholic solution of a surfactant and an alkyl amine; (ii) adding an organometallic metal precursor together with an aldehyde molecule, which acts as the key shape directing agent; and (iii) reacting the mixture in an autoclave for solvothermal crystallization. The shape of the ferrite nanoparticles can be controlled by the structure of the aldehyde ligand. Benzaldehyde and its aromatic derivatives favor the formation of cubic ferrite nanoparticles while aliphatic aldehydes result in spherical nanoparticles. The replacement of the primary amine, used in the nanocubes synthesis, with a secondary/tertiary amine results in nanoparticles with star-like shape. The well-defined control in terms of shape, narrow size distribution (below 5%), compositional tuning and crystallinity guarantees the preparation, at the gram scale, of nanocubes/star-like nanoparticles that possess, under magnetic field conditions of clinical use, specific adsorption rates comparable to or even superior to those obtained through thermal decomposition methods, which are typically prepared at the milligram scale. Here, gram-scale nanoparticle products with benchmark features for magnetic hyperthermia applications can be prepared in ~10 h with an average level of expertise in chemistry.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
Data availability
Supporting data for this protocol can be found at https://doi.org/10.6084/m9.figshare.20155535.v1.
References
Ferrari, M. Cancer nanotechnology: opportunities and challenges. Nat. Rev. Cancer 5, 161–171 (2005).
Simeonidis, K. et al. In-situ particles reorientation during magnetic hyperthermia application: shape matters twice. Sci. Rep. 6, 38382 (2016).
Ulbrich, K. et al. Targeted drug delivery with polymers and magnetic nanoparticles: covalent and noncovalent approaches, release control, and clinical studies. Chem. Rev. 116, 5338–5431 (2016).
Mai, B. T., Fernandes, S., Balakrishnan, P. B. & Pellegrino, T. Nanosystems based on magnetic nanoparticles and thermo-or pH-responsive polymers: an update and future perspectives. Acc. Chem. Res. 51, 999–1013 (2018).
Farzin, A., Etesami, S. A., Quint, J., Memic, A. & Tamayol, A. Magnetic nanoparticles in cancer therapy and diagnosis. Adv. Healthc. Mater. 9, 1901058 (2020).
Irvine, D. J. & Dane, E. L. Enhancing cancer immunotherapy with nanomedicine. Nat. Rev. Immunol. 20, 321–334 (2020).
Mahmoudi, K., Bouras, A., Bozec, D., Ivkov, R. & Hadjipanayis, C. Magnetic hyperthermia therapy for the treatment of glioblastoma: a review of the therapy’s history, efficacy and application in humans. Int. J. Hyperth. 34, 1316–1328 (2018).
Helena Gavilán, S. K. A. et al. Magnetic nanoparticles and clusters for magnetic hyperthermia: optimizing their heat performance and developing combinatorial therapies to tackle cancer. Chem. Soc. Rev. https://doi.org/10.1039/D1CS00427A (2021).
Tatarchuk, T., Bououdina, M., Judith Vijaya, J. & John Kennedy, L. Spinel ferrite nanoparticles: synthesis, crystal structure, properties, and perspective applications. In Int. Conf. on Nanotechnol. Nanomater. Vol. 195, 305–325 (Springer, 2016).
Kefeni, K. K., Msagati, T. A., Nkambule, T. T. & Mamba, B. B. Spinel ferrite nanoparticles and nanocomposites for biomedical applications and their toxicity. Mater. Sci. Eng. C. 107, 110314 (2020).
Frey, N. A., Peng, S., Cheng, K. & Sun, S. Magnetic nanoparticles: synthesis, functionalization, and applications in bioimaging and magnetic energy storage. Chem. Soc. Rev. 38, 2532–2542 (2009).
Roca, A. et al. Progress in the preparation of magnetic nanoparticles for applications in biomedicine. J. Phys. Appl. Phys. 42, 224002 (2009).
Martinez-Boubeta, C. et al. Learning from nature to improve the heat generation of iron-oxide nanoparticles for magnetic hyperthermia applications. Sci. Rep. 3, 1652 (2013).
Gandia, D. et al. Unlocking the potential of magnetotactic bacteria as magnetic hyperthermia agents. Small 15, 1902626 (2019).
Alphandery, E., Faure, S., Seksek, O., Guyot, F. & Chebbi, I. Chains of magnetosomes extracted from AMB-1 magnetotactic bacteria for application in alternative magnetic field cancer therapy. ACS Nano 5, 6279–6296 (2011).
Kim, D. et al. Synthesis of uniform ferrimagnetic magnetite nanocubes. J. Am. Chem. Soc. 131, 454–455 (2009).
Cullity, B. D. & Graham, C. D. Introduction to Magnetic Materials (John Wiley & Sons, 2011).
Fock, J. et al. On the ‘centre of gravity’method for measuring the composition of magnetite/maghemite mixtures, or the stoichiometry of magnetite-maghemite solid solutions, via 57Fe Mössbauer spectroscopy. J. Phys. D. Appl. Phys. 50, 265005 (2017).
Salazar-Alvarez, G. et al. Cubic versus spherical magnetic nanoparticles: the role of surface anisotropy. J. Am. Chem. Soc. 130, 13234–13239 (2008).
Noh, S.-h et al. Nanoscale magnetism control via surface and exchange anisotropy for optimized ferrimagnetic hysteresis. Nano Lett. 12, 3716–3721 (2012).
Espinosa, A. et al. Duality of iron oxide nanoparticles in cancer therapy: amplification of heating efficiency by magnetic hyperthermia and photothermal bimodal treatment. ACS Nano 10, 2436–2446 (2016).
Guardia, P. et al. Water-soluble iron oxide nanocubes with high values of specific absorption rate for cancer cell hyperthermia treatment. ACS Nano 6, 3080–3091 (2012).
Guardia, P. et al. One pot synthesis of monodisperse water soluble iron oxide nanocrystals with high values of the specific absorption rate. J. Mater. Chem. B 2, 4426–4434 (2014).
Yang, L. et al. Composition tunable manganese ferrite nanoparticles for optimized T 2 contrast ability. Chem. Mater. 29, 3038–3047 (2017).
Lee, J.-H. et al. Exchange-coupled magnetic nanoparticles for efficient heat induction. Nat. Nanotechnol. 6, 418–422 (2011).
Sathya, A. et al. CoxFe3–xO4 nanocubes for theranostic applications: effect of cobalt content and particle size. Chem. Mater. 28, 1769–1780 (2016).
Silvestri, N. et al. Di-and tri-component spinel ferrite nanocubes: synthesis and their comparative characterization for theranostic applications. Nanoscale 13, 13665–13680 (2021).
Lartigue, L. et al. Biodegradation of iron oxide nanocubes: high-resolution in situ monitoring. Acs Nano 7, 3939–3952 (2013).
Kolosnjaj-Tabi, J. et al. Heat-generating iron oxide nanocubes: subtle “destructurators” of the tumoral microenvironment. ACS Nano 8, 4268–4283 (2014).
Mai, B. T. et al. Thermoresponsive iron oxide nanocubes for an effective clinical translation of magnetic hyperthermia and heat-mediated chemotherapy. ACS Appl. Mater. Interfaces 11, 5727–5739 (2019).
Avugadda, S. K. et al. Esterase-cleavable 2D assemblies of magnetic iron oxide nanocubes: exploiting enzymatic polymer disassembling to improve magnetic hyperthermia heat losses. Chem. Mater. 31, 5450–5463 (2019).
Zyuzin, M. V. et al. Confining iron oxide nanocubes inside submicrometric cavities as a key strategy to preserve magnetic heat losses in an intracellular environment. ACS Appl. Mater. Interfaces 11, 41957–41971 (2019).
Roca, A. G. et al. Design strategies for shape-controlled magnetic iron oxide nanoparticles. Adv. Drug Deliv. Rev. 138, 68–104 (2019).
Kovalenko, M. V. et al. Fatty acid salts as stabilizers in size-and shape-controlled nanocrystal synthesis: the case of inverse spinel iron oxide. J. Am. Chem. Soc. 129, 6352–6353 (2007).
Guardia, P., Pérez, N., Labarta, A. & Batlle, X. Controlled synthesis of iron oxide nanoparticles over a wide size range. Langmuir 26, 5843–5847 (2010).
Gao, G. et al. Shape-controlled synthesis and magnetic properties of monodisperse Fe3O4 nanocubes. Cryst. Growth Des. 10, 2888–2894 (2010).
Xu, Z., Shen, C., Tian, Y., Shi, X. & Gao, H.-J. Organic phase synthesis of monodisperse iron oxide nanocrystals using iron chloride as precursor. Nanoscale 2, 1027–1032 (2010).
Wang, Z. L. Transmission electron microscopy of shape-controlled nanocrystals and their assemblies. J. Phys. Chem. B 104, 1153–1175 (2000).
Khurshid, H. et al. Synthesis and magnetic properties of core/shell FeO/Fe3O4 nano-octopods. J. Appl. Phys. 113, 17B508 (2013).
Niculaes, D. et al. Asymmetric assembling of iron oxide nanocubes for improving magnetic hyperthermia performance. ACS Nano 11, 12121–12133 (2017).
Muro-Cruces, J. et al. Precise size control of the growth of Fe3O4 nanocubes over a wide size range using a rationally designed one-pot synthesis. ACS Nano 13, 7716–7728 (2019).
Gilbert, K. & Gajewski, J. Coal liquefaction model studies: free radical chain decomposition of diphenylpropane, dibenzyl ether, and phenethyl phenyl ether via. beta.-scission reactions. J. Org. Chem. 47, 4899–4902 (1982).
Qiao, L. et al. Standardizing size-and shape-controlled synthesis of monodisperse magnetite (Fe3O4) nanocrystals by identifying and exploiting effects of organic impurities. ACS Nano 11, 6370–6381 (2017).
Johannsen, M. et al. Clinical hyperthermia of prostate cancer using magnetic nanoparticles: presentation of a new interstitial technique. Int. J. Hyperth. 21, 637–647 (2005).
Johannsen, M. et al. Thermotherapy of prostate cancer using magnetic nanoparticles: feasibility, imaging, and three-dimensional temperature distribution. Eur. Urol. 52, 1653–1662 (2007).
Jordan, A. et al. Presentation of a new magnetic field therapy system for the treatment of human solid tumors with magnetic fluid hyperthermia. J. Magn. Magn. Mater. 225, 118–126 (2001).
Maier-Hauff, K. et al. Efficacy and safety of intratumoral thermotherapy using magnetic iron-oxide nanoparticles combined with external beam radiotherapy on patients with recurrent glioblastoma multiforme. J. Neurooncol. 103, 317–324 (2011).
Thiesen, B. & Jordan, A. Clinical applications of magnetic nanoparticles for hyperthermia. Int. J. Hyperth. 24, 467–474 (2008).
Hühn, J. et al. Selected standard protocols for the synthesis, phase transfer, and characterization of inorganic colloidal nanoparticles. Chem. Mater. 29, 399–461 (2017).
Fortin, J.-P. et al. Size-sorted anionic iron oxide nanomagnets as colloidal mediators for magnetic hyperthermia. J. Am. Chem. Soc. 129, 2628–2635 (2007).
Shin, Y. J. et al. Centrifugal shape sorting and optical response of polyhedral gold nanoparticles. Adv. Mater. 25, 4023–4027 (2013).
Park, J. et al. Ultra-large-scale syntheses of monodisperse nanocrystals. Nat. Mater. 3, 891–895 (2004).
Gonzales-Weimuller, M., Zeisberger, M. & Krishnan, K. M. Size-dependant heating rates of iron oxide nanoparticles for magnetic fluid hyperthermia. J. Magn. Magn. Mater. 321, 1947–1950 (2009).
Castellanos-Rubio, I. et al. Outstanding heat loss via nano-octahedra above 20 nm in size: from wustite-rich nanoparticles to magnetite single-crystals. Nanoscale 11, 16635–16649 (2019).
Lak, A. et al. Fe2+ deficiencies, FeO subdomains, and structural defects favor magnetic hyperthermia performance of iron oxide nanocubes into intracellular environment. Nano Lett. 18, 6856–6866 (2018).
Krishnan, S., Bhardwaj, S., Liu, S., Xing, R. & Chavali, M. Handbook of Greener Synthesis of Nanomaterials and Compounds (Elsevier Amsterdam, The Netherlands:, 2021).
Tunison, B. R. Industrial alcohol. J. Franklin Inst. 190, 373–420 (1920).
Richardson, J. J. & Caruso, F. Nanomedicine Toward 2040 (ACS Publications, 2020).
Das, R. et al. Tunable high aspect ratio iron oxide nanorods for enhanced hyperthermia. J. Phys. Chem. C. 120, 10086–10093 (2016).
Das, R. et al. Boosted hyperthermia therapy by combined AC magnetic and photothermal exposures in Ag/Fe3O4 nanoflowers. ACS Appl. Mater. Interfaces 8, 25162–25169 (2016).
Sun, H. et al. Solvothermal synthesis of tunable electroactive magnetite nanorods by controlling the side reaction. J. Phys. Chem. C. 116, 5476–5481 (2012).
Nemati, Z. et al. Enhanced magnetic hyperthermia in iron oxide nano-octopods: size and anisotropy effects. J. Phys. Chem. C. 120, 8370–8379 (2016).
Byrappa, K. & Yoshimura, M. Handbook of Hydrothermal Technology (William Andrew, 2012).
Quarta, A., Curcio, A., Kakwere, H. & Pellegrino, T. J. N. Polymer coated inorganic nanoparticles: tailoring the nanocrystal surface for designing nanoprobes with biological implications. Nanoscale 4, 3319–3334 (2012).
Connord, V., Mehdaoui, B., Tan, R., Carrey, J. & Respaud, M. An air-cooled Litz wire coil for measuring the high frequency hysteresis loops of magnetic samples—a useful setup for magnetic hyperthermia applications. Rev. Sci. Instrum. 85, 093904 (2014).
Gneveckow, U. et al. Description and characterization of the novel hyperthermia‐and thermoablation‐system for clinical magnetic fluid hyperthermia. J. Med. Phys. 31, 1444–1451 (2004).
Del Sol-Fernández, S. et al. Flower-like Mn-doped magnetic nanoparticles functionalized with α v β3-integrin-ligand to efficiently induce intracellular heat after alternating magnetic field exposition, triggering glioma cell death. ACS Appl. Mater. Interfaces 11, 26648–26663 (2019).
Hergt, R. & Dutz, S. Magnetic particle hyperthermia—biophysical limitations of a visionary tumour therapy. J. Magn. Magn. Mater. 311, 187–192 (2007).
Gutiérrez, L. et al. Aggregation effects on the magnetic properties of iron oxide colloids. Nanotechnology 30, 112001 (2019).
Périgo, E. A. et al. Fundamentals and advances in magnetic hyperthermia. Appl. Phys. Rev. 2, 041302 (2015).
Pellegrino, T. et al. Hydrophobic nanocrystals coated with an amphiphilic polymer shell: a general route to water soluble nanocrystals. Nano Lett. 4, 703–707 (2004).
Leal, M. P. et al. Long-circulating PEGylated manganese ferrite nanoparticles for MRI-based molecular imaging. Nanoscale 7, 2050–2059 (2015).
Salas, G. et al. Modulation of magnetic heating via dipolar magnetic interactions in monodisperse and crystalline iron oxide nanoparticles. J. Phys. Chem. C. 118, 19985–19994 (2014).
Salas, G. et al. Controlled synthesis of uniform magnetite nanocrystals with high-quality properties for biomedical applications. J. Mater. Chem. 22, 21065–21075 (2012).
Persano, S. et al. Elucidating the innate immunological effects of mild magnetic hyperthermia on U87 human glioblastoma cells: an in vitro study. Pharmaceutics 13, 1668 (2021).
Bordet, A. et al. Magnetically induced continuous CO2 hydrogenation using composite iron carbide nanoparticles of exceptionally high heating power. Angew. Chem. Int. Ed. 55, 15894–15898 (2016).
Martínez-Prieto, L. M. et al. Ultrastable magnetic nanoparticles encapsulated in carbon for magnetically induced catalysis. ACS Appl. Nano Mater. 3, 7076–7087 (2020).
Tucek, J., Kemp, K. C., Kim, K. S. & Zboril, R. Iron-oxide-supported nanocarbon in lithium-ion batteries, medical, catalytic, and environmental applications. ACS Nano 8, 7571–7612 (2014).
Talapin, D. V., Lee, J.-S., Kovalenko, M. V. & Shevchenko, E. V. Prospects of colloidal nanocrystals for electronic and optoelectronic applications. Chem. Rev. 110, 389–458 (2010).
Pellegrino, T. et al. Method for the gram-scale preparation of cubic ferrite nanocrystals for biomedical applications. International patent WO 2020/222133 (2020): https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2020222133
Pellegrino, T. et al. Method for the gram-scale preparation of ferrite nanoparticles for magnetic hyperthermia applications. International patent WO 2022/090316 (2022): https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2022090316
Acknowledgements
This work was funded by EC | EC Seventh Framework Programm | FP7 Ideas: European Research Council (FP7-IDEAS-ERC–Specific Programme: “Ideas” Implementing the Seventh Framework Programme of the European Community for Research, Technological Development and Demonstration Activities (2007 to 2013))—ICARO no. 678109 (T.P.) and the AIRC Associazione per la Ricerca sul Cancro Foundation (AIRC IG-14527) (T.P.).
Author information
Authors and Affiliations
Contributions
H.G., G.M.R.R., N.S., B.T.M. and T.P. contributed intellectually and practically to the development of the technique.
Corresponding authors
Peer review
Peer review information
Nature Protocols thanks Ali Dadfar, Twan Lammers and Pablo Taboada for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
Persano, S. et al. Pharmaceutics 13, 1668 (2021): https://doi.org/10.3390/pharmaceutics13101668
Pellegrino, T. et al. International patent WO 2020/222133 (2020): https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2020222133
Pellegrino, T. et al. International patent WO 2022/090316 (2022): https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2022090316
Supplementary information
Supplementary Information
Supplementary experimental section, water transfer protocols (Boxes 1–4), characterizations, Figs. 1–23, Tables 1–3 and References.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gavilán, H., Rizzo, G.M.R., Silvestri, N. et al. Scale-up approach for the preparation of magnetic ferrite nanocubes and other shapes with benchmark performance for magnetic hyperthermia applications. Nat Protoc 18, 783–809 (2023). https://doi.org/10.1038/s41596-022-00779-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-022-00779-3
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.