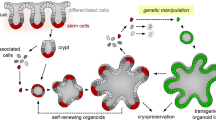
Abstract
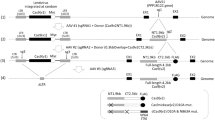
Human intestinal tissue–derived enteroids (HIEs; also called organoids) are a powerful ex vivo model for gastrointestinal research. Genetic modification of these nontransformed cultures allows new insights into gene function and biological processes involved in intestinal diseases as well as gastrointestinal and donor segment-specific function. Here we provide a detailed technical pipeline and protocol for using the CRISPR–Cas9 genome editing system to knock out a gene of interest specifically in HIEs by lentiviral transduction and single-cell cloning. This protocol differs from a previously published alternative using electroporation of human colonoids to deliver piggyback transposons or CRISPR–Cas9 constructs, as this protocol uses a modified, fused LentiCRISPRv2–small-guiding RNA to express Cas9 and small-guiding RNA in a lentivirus. The protocol also includes the steps of gene delivery and subsequent single-cell cloning of the knockout cells as well as verification of clones and sequence identification of the mutation sites to establish knockout clones. An overview flowchart, step-by-step guidelines and troubleshooting suggestions are provided to aid the researcher in obtaining the genetic knockout HIE line within 2–3 months. In this protocol, we further describe how to use HIEs as an ex vivo model to assess host restriction factors for viral replication (using human norovirus replication as an example) by knocking out host attachment factors or innate immunity genes. Other applications are discussed to broaden the utility of this system, for example, to generate knockin or conditional knockout HIE lines to investigate the function of essential genes in many biological processes including other types of organoids.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data are available from the authors upon request. Source data from Fig. 6 were generated using this protocol and were published in ref. 12. Source data are provided with this paper.
References
Sato, T. et al. Long-term expansion of epithelial organoids from human colon, adenoma, adenocarcinoma, and Barrett’s epithelium. Gastroenterology 141, 1762–1772 (2011).
Heo, I. et al. Modelling Cryptosporidium infection in human small intestinal and lung organoids. Nat. Microbiol. 3, 814–823 (2018).
Middendorp, S. et al. Adult stem cells in the small intestine are intrinsically programmed with their location-specific function. Stem Cells 32, 1083–1091 (2014).
Foulke-Abel, J. et al. Human enteroids as an ex-vivo model of host–pathogen interactions in the gastrointestinal tract. Exp. Biol. Med. 239, 1124–1134 (2014).
Rajan, R. et al. Novel segment- and host-specific patterns of enteroaggregative Escherichia coli adherence to human intestinal enteroids. mBio 9, e02419–17 (2018).
In, J. G. et al. Human mini-guts: new insights into intestinal physiology and host–pathogen interactions. Nat. Rev. Gastroenterol. Hepatol. 13, 633–642 (2016).
Zachos, N. C. et al. Human enteroids/colonoids and intestinal organoids functionally recapitulate normal intestinal physiology and pathophysiology. J. Biol. Chem. 291, 3759–3766 (2016).
Clevers, H. COVID-19: organoids go viral. Nat. Rev. Mol. Cell Biol. 21, 355–356 (2020).
Geurts, M. H., van der Vaart, J., Beumer, J. & Clevers, H. The organoid platform: promises and challenges as tools in the fight against COVID-19. Stem Cell Rep. 16, 412–418 (2021).
Hares, M. F. et al. Stem cell-derived enteroid cultures as a tool for dissecting host–parasite interactions in the small intestinal epithelium. Parasite Immunol. 43, e12765 (2021).
Ettayebi, K. et al. Replication of human noroviruses in stem cell-derived human enteroids. Science 353, 1387–1393 (2016).
Lin, S. C. et al. Human norovirus exhibits strain-specific sensitivity to host interferon pathways in human intestinal enteroids. Proc. Natl Acad. Sci. USA 117, 23782–23793 (2020).
Ettayebi, K. et al. New insights and enhanced human norovirus cultivation in human intestinal enteroids. mSphere 6, e01136–20 (2021).
Murakami, K. et al. Bile acids and ceramide overcome the entry restriction for GII.3 human norovirus replication in human intestinal enteroids. Proc. Natl Acad. Sci. USA 117, 1700–1710 (2020).
Haga, K. et al. Genetic manipulation of human intestinal enteroids demonstrates the necessity of a functional fucosyltransferase 2 gene for secretor-dependent human norovirus infection. mBio 11, e00251–20 (2020).
Ishino, Y., Shinagawa, H., Makino, K., Amemura, M. & Nakata, A. Nucleotide sequence of the iap gene, responsible for alkaline phosphatase isozyme conversion in Escherichia coli, and identification of the gene product. J. Bacteriol. 169, 5429–5433 (1987).
Jinek, M. et al. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science 337, 816–821 (2012).
Mali, P. et al. CAS9 transcriptional activators for target specificity screening and paired nickases for cooperative genome engineering. Nat. Biotechnol. 31, 833–838 (2013).
Cong, L. et al. Multiplex genome engineering using CRISPR/Cas systems. Science 339, 819–823 (2013).
Doudna, J. A. & Charpentier, E. Genome editing. The new frontier of genome engineering with CRISPR-Cas9. Science 346, 1258096 (2014).
Paques, F. & Haber, J. E. Multiple pathways of recombination induced by double-strand breaks in Saccharomyces cerevisiae. Microbiol. Mol. Biol. Rev. 63, 349–404 (1999).
Urnov, F. D., Rebar, E. J., Holmes, M. C., S., H. & Zhang, P. D. Gregory, Genome editing with engineered zinc finger nucleases. Nat. Rev. Genet. 11, 636–646 (2010).
Moore, J. K. & Haber, J. E. Cell cycle and genetic requirements of two pathways of nonhomologous end-joining repair of double-strand breaks in Saccharomyces cerevisiae. Mol. Cell Biol. 16, 2164–2173 (1996).
Durai, S. et al. Zinc finger nucleases: custom-designed molecular scissors for genome engineering of plant and mammalian cells. Nucleic Acids Res. 33, 5978–5990 (2005).
Shalem, O. et al. Genome-scale CRISPR–Cas9 knockout screening in human cells. Science 343, 84–87 (2014).
Chang-Graham, A. L. et al. Rotavirus induces intercellular calcium waves through ADP signaling. Science 370, eabc3621 (2020).
Giuliano, C. J., Lin, A., Girish, V. & Sheltzer, J. M. Generating single cell-derived knockout clones in mammalian cells with CRISPR/Cas9. Curr. Protoc. Mol. Biol. 128, e100 (2019).
Michels, B. E. et al. Human colon organoids reveal distinct physiologic and oncogenic Wnt responses. J. Exp. Med. 216, 704–720 (2019).
Ousterout, D. G. et al. Multiplex CRISPR/Cas9-based genome editing for correction of dystrophin mutations that cause Duchenne muscular dystrophy. Nat. Commun. 6, 6244 (2015).
Shimokawa, M. et al. Visualization and targeting of LGR5+ human colon cancer stem cells. Nature 545, 187–192 (2017).
Schwank, G. et al. Functional repair of CFTR by CRISPR/Cas9 in intestinal stem cell organoids of cystic fibrosis patients. Cell Stem Cell 13, 653–658 (2013).
Artegiani, B. et al. Fast and efficient generation of knock-in human organoids using homology-independent CRISPR–Cas9 precision genome editing. Nat. Cell Biol. 22, 321–331 (2020).
Hendriks, D., Clevers, H. & Artegiani, B. CRISPR-Cas tools and their application in genetic engineering of human stem cells and organoids. Cell Stem Cell 27, 705–731 (2020).
Fujii, M., Matano, M., Nanki, K. & Sato, T. Efficient genetic engineering of human intestinal organoids using electroporation. Nat. Protoc. 10, 1474–1485 (2015).
Matano, M. et al. Modeling colorectal cancer using CRISPR–Cas9-mediated engineering of human intestinal organoids. Nat. Med. 21, 256–262 (2015).
Teriyapirom, I., Batista-Rocha, A. S. & Koo, B. K. Genetic engineering in organoids. J. Mol. Med. 99, 555–568 (2021).
A. M. Gaebler et al. Universal and efficient electroporation protocol for genetic engineering of gastrointestinal organoids. J. Vis. Exp. https://doi.org/10.3791/60704 (2020).
H. Miyoshi, T. S. Stappenbeck, In vitro expansion and genetic modification of gastrointestinal stem cells in spheroid culture. Nat. Protoc. 8, 2471–2482 (2013).
Drost, J., Artegiani, B. & Clevers, H. The generation of organoids for studying Wnt signaling. Methods Mol. Biol. 1481, 141–159 (2016).
J. F. Van Lidth de Jeude, J. L. Vermeulen, P. S. Montenegro-Miranda, G. R. Van den Brink, J. Heijmans, A protocol for lentiviral transduction and downstream analysis of intestinal organoids. J. Vis. Exp. https://doi.org/10.3791/52531 (2015).
A. Merenda et al. A protocol for multiple gene knockout in mouse small intestinal organoids using a CRISPR-concatemer. J. Vis. Exp. https://doi.org/10.3791/55916 (2017).
Zhang, J., Chen, L., Zhang, J. & Wang, Y. Drug inducible CRISPR/Cas systems. Comput. Struct. Biotechnol. J. 17, 1171–1177 (2019).
Chen, Y. et al. Engineering human stem cell lines with inducible gene knockout using CRISPR/Cas9. Cell Stem Cell 17, 233–244 (2015).
Doench, J. G. et al. Optimized sgRNA design to maximize activity and minimize off-target effects of CRISPR–Cas9. Nat. Biotechnol. 34, 184–191 (2016).
Altschul, S. F., Gish, W., Miller, W., Myers, E. W. & Lipman, D. J. Basic local alignment search tool. J. Mol. Biol. 215, 403–410 (1990).
Hay, E. A. et al. An analysis of possible off target effects following CAS9/CRISPR targeted deletions of neuropeptide gene enhancers from the mouse genome. Neuropeptides 64, 101–107 (2017).
Saxena, K. et al. Human intestinal enteroids: a new model to study human rotavirus infection, host restriction, and pathophysiology. J. Virol. 90, 43–56 (2016).
Wang, T., Wei, J. J., Sabatini, D. M. & Lander, E. S. Genetic screens in human cells using the CRISPR–Cas9 system. Science 343, 80–84 (2014).
Jiang, X., Wang, M., Graham, D. Y. & Estes, M. K. Expression self-assembly and antigenicity of the Norwalk virus capsid protein. J. Virol. 66, 6527–6532 (1992).
VanDussen, K. L., Sonnek, N. M. & Stappenbeck, T. S. L-WRN conditioned medium for gastrointestinal epithelial stem cell culture shows replicable batch-to-batch activity levels across multiple research teams. Stem Cell Res. 37, 101430 (2019).
Sastry, L., Johnson, T., Hobson, M. J., Smucker, B. & Cornetta, K. Titering lentiviral vectors: comparison of DNA, RNA and marker expression methods. Gene Ther. 9, 1155–1162 (2002).
Barczak, W., Suchorska, W., Rubiś, B. & Kulcenty, K. Universal real-time PCR-based assay for lentiviral titration. Mol. Biotechnol. 57, 195–200 (2015).
Joung, J. et al. Genome-scale CRISPR–Cas9 knockout and transcriptional activation screening. Nat. Protoc. 12, 828–863 (2017).
Loisy, F. et al. Real-time RT-PCR for norovirus screening in shellfish. J. Virol. Methods 123, 1–7 (2005).
Acknowledgements
We thank B. Lee and the Cell-Based Assay Screening Service (C-BASS) Core in BCM for lentivirus packaging plasmids. This work was supported in part by Public Health Service grants PO1AI057788, U19AI116497, U19AI144297, P30DK56338 and contract HHSN2722017000381 from the National Institutes of Health, by The Cancer Prevention Institute of Texas (CPRIT) RP160283—Baylor College of Medicine Comprehensive Cancer Training Program and RP17005, by NIH P30 shared resource grant CA125123, NIEHS grants P30ES030285 and P42ES0327725, and by the John S. Dunn Research Foundation.
Author information
Authors and Affiliations
Contributions
S.-C.L. and K.H. designed the experiments. S.-C.L. developed the protocol with X.-L.Z. S.-C.L. and X.-L.Z. performed the experiments. S.-C.L., K.H. and M.K.E. wrote the protocol.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Protocols thanks Devanjali Dutta and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
Lin, S. C. et al. Proc. Natl Acad. Sci. USA 117, 23782–23793 (2020): https://doi.org/10.1073/pnas.2010834117
Haga, K. et al. mBio 11, e00251-20 (2020): https://doi.org/10.1128/mBio.00251-20
Supplementary information
Supplementary Information
Supplementary Methods.
Source data
Source Data Fig. 6
Unprocessed western blots for Fig. 6a and original images for Fig. 6b.
Source Data Fig. 6
Statistical source data for Fig. 6c.
Rights and permissions
About this article
Cite this article
Lin, SC., Haga, K., Zeng, XL. et al. Generation of CRISPR–Cas9-mediated genetic knockout human intestinal tissue–derived enteroid lines by lentivirus transduction and single-cell cloning. Nat Protoc 17, 1004–1027 (2022). https://doi.org/10.1038/s41596-021-00669-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-021-00669-0
This article is cited by
-
Tumor-derived exosomes induce initial activation by exosomal CD19 antigen but impair the function of CD19-specific CAR T-cells via TGF-β signaling
Frontiers of Medicine (2024)
-
Recent advances in therapeutic CRISPR-Cas9 genome editing: mechanisms and applications
Molecular Biomedicine (2023)
-
CRISPR/Cas9 therapeutics: progress and prospects
Signal Transduction and Targeted Therapy (2023)
-
Blockade of interferon signaling decreases gut barrier integrity and promotes severe West Nile virus disease
Nature Communications (2023)
-
Applications of human organoids in the personalized treatment for digestive diseases
Signal Transduction and Targeted Therapy (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.