Abstract
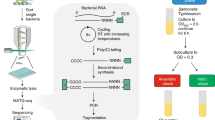
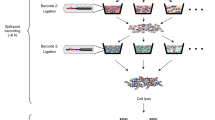
We recently developed a high-throughput functional genomics approach, named ‘SorTn-seq’, to identify factors affecting expression of any gene of interest in bacteria. Our approach facilitates high-throughput screening of complex mutant pools, a task previously hindered by a lack of suitable techniques. SorTn-seq combines high-density, Tn5-like transposon mutagenesis with fluorescence-activated cell sorting of a strain harboring a promoter-fluorescent reporter fusion, to isolate mutants with altered gene expression. The transposon mutant pool is sorted into different bins on the basis of fluorescence, and mutants are deep-sequenced to identify transposon insertions. DNA is prepared for sequencing by using commercial kits augmented with custom primers, enhancing ease of use and reproducibility. Putative regulators are identified by comparing the number of insertions per genomic feature in the different sort bins, by using existing bioinformatic pipelines and software packages. SorTn-seq can be completed in 1–2 weeks and requires general microbiology skills and basic flow cytometry experience.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
Data availability
Sequencing data originally published in ref. 13 are available in the Sequence Read Archive under BioProject number PRJNA601789. The annotated genome of Serratia sp. ATCC 39006-LacA is available through the National Center for Biotechnology Information (reference sequence NZ_CP025085.1). Source data are provided with this paper.
Code availability
R scripts and files required for data processing the Serratia csm dataset are available at Zenodo (https://doi.org/10.5281/zenodo.4554398) and on GitHub (https://github.com/JacksonLab/SorTn-seq)64.
References
Martínez-García, E. & de Lorenzo, V. Transposon-based and plasmid-based genetic tools for editing genomes of gram-negative bacteria. in Synthetic Gene Networks: Methods and Protocols (eds. Weber, W. & Fussenegger, M.) 267–283 (Springer, 2012).
Munoz-Lopez, M. & Garcia-Perez, J. L. DNA transposons: nature and applications in genomics. Curr. Genomics 11, 115–128 (2010).
Lampe, D. J., Grant, T. E. & Robertson, H. M. Factors affecting transposition of the Himar1 mariner transposon in vitro. Genetics 149, 179–187 (1998).
Rubin, E. J. et al. In vivo transposition of mariner-based elements in enteric bacteria and mycobacteria. Proc. Natl Acad. Sci. USA 96, 1645–1650 (1999).
Goryshin, I. Y., Miller, J. A., Kil, Y. V., Lanzov, V. A. & Reznikoff, W. S. Tn5/IS50 target recognition. Proc. Natl Acad. Sci. USA 95, 10716–10721 (1998).
van Opijnen, T. & Levin, H. L. Transposon insertion sequencing, a global measure of gene function. Annu. Rev. Genet 54, 337–365 (2020).
van Opijnen, T., Bodi, K. L. & Camilli, A. Tn-seq: high-throughput parallel sequencing for fitness and genetic interaction studies in microorganisms. Nat. Methods 6, 767–772 (2009).
Goodman, A. L. et al. Identifying genetic determinants needed to establish a human gut symbiont in its habitat. Cell Host Microbe 6, 279–289 (2009).
Langridge, G. C. et al. Simultaneous assay of every Salmonella Typhi gene using one million transposon mutants. Genome Res. 19, 2308–2316 (2009).
Barquist, L. et al. The TraDIS toolkit: sequencing and analysis for dense transposon mutant libraries. Bioinformatics 32, 1109–1111 (2016).
Davey, L. & Valdivia, R. H. Bacterial genetics and molecular pathogenesis in the age of high throughput DNA sequencing. Curr. Opin. Microbiol. 54, 59–66 (2020).
Cain, A. K. et al. A decade of advances in transposon-insertion sequencing. Nat. Rev. Genet 21, 526–540 (2020).
Smith, L. M. et al. The Rcs stress response inversely controls surface and CRISPR-Cas adaptive immunity to discriminate plasmids and phages. Nat. Microbiol. 6, 162–172 (2021).
Givan, A. L. Flow Cytometry: First Principles (John Wiley & Sons, 2013).
Robinson, M. D., McCarthy, D. J. & Smyth, G. K. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26, 139–140 (2010).
Pessi, G., Blumer, C. & Haas, D. lacZ fusions report gene expression, don’t they? Microbiology 147, 1993–1995 (2001).
Abuaita, B. H. & Withey, J. H. Genetic screening for bacterial mutants in liquid growth media by fluorescence-activated cell sorting. J. Microbiol. Methods 84, 109–113 (2011).
Binder, S. et al. A high-throughput approach to identify genomic variants of bacterial metabolite producers at the single-cell level. Genome Biol. 13, R40 (2012).
Hassan, K. A. et al. Fluorescence-based flow sorting in parallel with transposon insertion site sequencing identifies multidrug efflux systems in Acinetobacter baumannii. mBio 7, e01200-16 (2016).
Dorman, M. J., Feltwell, T., Goulding, D. A., Parkhill, J. & Short, F. L. The capsule regulatory network of Klebsiella pneumoniae defined by density-TraDISort. mBio 9, e01863-18 (2018).
Duncan, M. C. et al. High-throughput analysis of gene function in the bacterial predator Bdellovibrio bacteriovorus. mBio 10, e01040-19 (2019).
Rego, E. H., Audette, R. E. & Rubin, E. J. Deletion of a mycobacterial divisome factor collapses single-cell phenotypic heterogeneity. Nature 546, 153–157 (2017).
Fowler, C. C. & Galan, J. E. Decoding a Salmonella typhi regulatory network that controls typhoid toxin expression within human cells. Cell Host Microbe 23, 65–76.e6 (2018).
Thibault, D. et al. Droplet Tn-Seq combines microfluidics with Tn-Seq for identifying complex single-cell phenotypes. Nat. Commun. 10, 5729 (2019).
Goodman, A. L., Wu, M. & Gordon, J. I. Identifying microbial fitness determinants by insertion sequencing using genome-wide transposon mutant libraries. Nat. Protoc. 6, 1969–1980 (2011).
Barquist, L. et al. A comparison of dense transposon insertion libraries in the Salmonella serovars Typhi and Typhimurium. Nucleic Acids Res. 41, 4549–4564 (2013).
Christen, B. et al. The essential genome of a bacterium. Mol. Syst. Biol. 7, 528 (2011).
Aird, D. et al. Analyzing and minimizing PCR amplification bias in Illumina sequencing libraries. Genome Biol. 12, R18 (2011).
Mitra, A., Skrzypczak, M., Ginalski, K. & Rowicka, M. Strategies for achieving high sequencing accuracy for low diversity samples and avoiding sample bleeding using illumina platform. PLoS One 10, e0120520 (2015).
Schlechter, R. O. et al. Chromatic bacteria—a broad host-range plasmid and chromosomal insertion toolbox for fluorescent protein expression in bacteria. Front. Microbiol. 9, 3052 (2018).
Truong, L. & Ferre-D’Amare, A. R. From fluorescent proteins to fluorogenic RNAs: tools for imaging cellular macromolecules. Protein Sci. 28, 1374–1386 (2019).
Lambert, T. J. FPbase: a community-editable fluorescent protein database. Nat. Methods 16, 277–278 (2019).
Day, R. N. & Schaufele, F. Fluorescent protein tools for studying protein dynamics in living cells: a review. J. Biomed. Opt. 13, 031202 (2008).
Roederer, M. Spectral compensation for flow cytometry: visualization artifacts, limitations, and caveats. Cytometry 45, 194–205 (2001).
Williamson, N. R. et al. Biosynthesis of the red antibiotic, prodigiosin, in Serratia: identification of a novel 2-methyl-3-n-amyl-pyrrole (MAP) assembly pathway, definition of the terminal condensing enzyme, and implications for undecylprodigiosin biosynthesis in Streptomyces. Mol. Microbiol. 56, 971–989 (2005).
Andreyeva, I. N. & Ogorodnikova, T. I. Pigmentation of Serratia marcescens and spectral properties of prodigiosin. Microbiology 84, 28–33 (2015).
del Solar, G., Giraldo, R., Ruiz-Echevarria, M. J., Espinosa, M. & Diaz-Orejas, R. Replication and control of circular bacterial plasmids. Microbiol. Mol. Biol. Rev. 62, 434–464 (1998).
Melamed, S. et al. Global mapping of small RNA-target interactions in bacteria. Mol. Cell 63, 884–897 (2016).
Paige, J. S., Wu, K. Y. & Jaffrey, S. R. RNA mimics of green fluorescent protein. Science 333, 642–646 (2011).
Ouellet, J. RNA fluorescence with light-up aptamers. Front. Chem. 4, 29 (2016).
Barnard, A., Wolfe, A. & Busby, S. Regulation at complex bacterial promoters: how bacteria use different promoter organizations to produce different regulatory outcomes. Curr. Opin. Microbiol. 7, 102–108 (2004).
Thoma, S. & Schobert, M. An improved Escherichia coli donor strain for diparental mating. FEMS Microbiol. Lett. 294, 127–132 (2009).
Jackson, S. A., Fellows, B. J. & Fineran, P. C. Complete genome sequences of the Escherichia coli donor strains ST18 and MFDpir. Microbiol. Resour. Announc. 9, e01014-20 (2020).
Patterson, A. G., Chang, J. T., Taylor, C. & Fineran, P. C. Regulation of the Type I-F CRISPR-Cas system by CRP-cAMP and GalM controls spacer acquisition and interference. Nucleic Acids Res. 43, 6038–6048 (2015).
Mesarich, C. H. et al. Transposon insertion libraries for the characterization of mutants from the kiwifruit pathogen Pseudomonas syringae pv. actinidiae. PLoS One 12, e0172790 (2017).
Monson, R. et al. A plasmid-transposon hybrid mutagenesis system effective in a broad range of Enterobacteria. Front. Microbiol. 6, 1442 (2015).
Karsi, A. & Lawrence, M. L. Broad host range fluorescence and bioluminescence expression vectors for Gram-negative bacteria. Plasmid 57, 286–295 (2007).
Stuurman, N. et al. Use of green fluorescent protein color variants expressed on stable broad-host-range vectors to visualize rhizobia interacting with plants. Mol. Plant Microbe Interact. 13, 1163–1169 (2000).
Garcia-Cayuela, T. et al. Fluorescent protein vectors for promoter analysis in lactic acid bacteria and Escherichia coli. Appl. Microbiol. Biotechnol. 96, 171–181 (2012).
Charpentier, E. et al. Novel cassette-based shuttle vector system for gram-positive bacteria. Appl. Environ. Microbiol. 70, 6076–6085 (2004).
Aymanns, S., Mauerer, S., van Zandbergen, G., Wolz, C. & Spellerberg, B. High-level fluorescence labeling of gram-positive pathogens. PLoS One 6, e19822 (2011).
Barbier, M. & Damron, F. H. Rainbow vectors for broad-range bacterial fluorescence labeling. PLoS One 11, e0146827 (2016).
Birkholz, N., Fagerlund, R. D., Smith, L. M., Jackson, S. A. & Fineran, P. C. The autoregulator Aca2 mediates anti-CRISPR repression. Nucleic Acids Res. 47, 9658–9665 (2019).
Genove, G., Glick, B. S. & Barth, A. L. Brighter reporter genes from multimerized fluorescent proteins. Biotechniques 39, 814 (2005). 816, 818 passim.
Yasir, M. et al. TraDIS-Xpress: a high-resolution whole-genome assay identifies novel mechanisms of triclosan action and resistance. Genome Res. 30, 239–249 (2020).
Baba, T. et al. Construction of Escherichia coli K-12 in-frame, single-gene knockout mutants: the Keio collection. Mol. Syst. Biol. 2, 2006.0008 (2006).
Kitagawa, M. et al. Complete set of ORF clones of Escherichia coli ASKA library (a complete set of E. coli K-12 ORF archive): unique resources for biological research. DNA Res. 12, 291–299 (2005).
Peters, J. M. et al. Enabling genetic analysis of diverse bacteria with Mobile-CRISPRi. Nat. Microbiol. 4, 244–250 (2019).
Wickham, H. et al. Welcome to the Tidyverse. J. Open Source Softw. 4, 1686 (2019).
Andrews, S. FastQC: A Quality Control Tool for High Throughput Sequence Data (Babraham Bioinformatics, Babraham Institute, 2010).
Bolger, A. M., Lohse, M. & Usadel, B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30, 2114–2120 (2014).
Quinlan, A. R. BEDTools: the Swiss-Army tool for genome feature analysis. Curr. Protoc. Bioinform. 47, 11.12.11–11.12.34 (2014).
The R Foundation. R: A Language and Environment for Statistical Computing. https://www.r-project.org/ (2020).
Jackson, S. A. JacksonLab/SorTn-seq: 1.0.0 (Version v). Zenodo https://doi.org/10.5281/zenodo.4554398 (2021).
RStudio. RStudio: Integrated Development for R. http://www.rstudio.com/ (2020).
Schmieder, R. & Edwards, R. Quality control and preprocessing of metagenomic datasets. Bioinformatics 27, 863–864 (2011).
Rutherford, K. et al. Artemis: sequence visualization and annotation. Bioinformatics 16, 944–945 (2000).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Ponstingl, H. & Ning, Z. SMALT-a new mapper for DNA sequencing reads. F1000Posters 1, 313 (poster) (2010).
Robinson, M. D. & Smyth, G. K. Moderated statistical tests for assessing differences in tag abundance. Bioinformatics 23, 2881–2887 (2007).
Robinson, M. D. & Smyth, G. K. Small-sample estimation of negative binomial dispersion, with applications to SAGE data. Biostatistics 9, 321–332 (2008).
Acknowledgements
This work was supported by the Marsden Fund from the Royal Society of New Zealand and the School of Biomedical Sciences Bequest Fund from the University of Otago. L.M.S. was supported by a University of Otago Doctoral Scholarship and Postgraduate Publishing Bursary. We thank M. Wilson for help with cell sorting and A. Jeffs of the Otago Genomics Facility for assistance with sequencing. We thank J. Ussher and R. Hannaway for help with flow cytometry. We thank H. Hampton, D. Mayo-Muñoz, N. Birkholz and members of the Fineran laboratory for helpful discussions.
Author information
Authors and Affiliations
Contributions
L.M.S. designed all experiments with input from S.A.J. and P.C.F. L.M.S. performed all experiments (except where a specialized facility was used), analyzed all data and prepared all figures. S.A.J. wrote data processing scripts, and P.P.G. provided input into bioinformatic analyses. P.C.F. conceived the project. S.A.J., P.P.G. and P.C.F. supervised the project. L.M.S. and P.C.F. wrote the manuscript. All authors edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Protocols thanks the anonymous reviewers for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key reference using this protocol
Smith, L. et al. Nat. Microbiol. 6, 162–172 (2021): https://doi.org/10.1038/s41564-020-00822-7
Extended data
Extended Data Fig. 1 Gating on a secondary reporter eliminates noise from the primary reporter fluorescence distribution.
The primary reporter (eYFP) fluorescence distribution is shown for without (a) or with (b) gating on a secondary reporter (mCherry). In a and b, events are first gated on SSC and FSC parameters area (A) and height (H) to isolate individual bacteria (‘singlets’ and ‘cells’). a, A characteristic secondary peak is observed centered around zero. Many of the events comprising this secondary peak exhibit negative fluorescence levels and therefore cannot be sorted. This non-fluorescent population is probably comprised of dead/dormant cells, cellular debris, bubbles or electronic noise generated by the instrument. b, The addition of a gate around mCherry-positive (mCherry+) events results in the removal of the secondary peak from the eYFP fluorescence distribution. This improved distribution allows for more accurate placement of gates during cell sorting.
Extended Data Fig. 2 Promoter region and reporter plasmid used during SorTn-seq.
Type III-A CRISPR-Cas expression was measured from an eYFP fusion to 250 nt upstream of the csm operon. Key features in the promoter are indicated (−35 and −10) as are the transcription start site (+1), native RBS and the start codon (ATG, green). An IPTG-inducible 2nd fluorophore (mCherry) is under the control of the T5-lac promoter (PT5-lac). The pBR322 origin of replication facilitates ~15–20 copies of the reporter per cell.
Extended Data Fig. 3 Transposon delivery vector and transposon organization.
a, Organization of the pKRCPN2 transposon delivery vector. b, Schematic of the transposon Tn-DS1028uidAKm, which contains a transcriptional uidA reporter, origin of replication (R6Kγ) and kanamycin resistance gene (KmR).
Supplementary information
Supplementary Information
Supplementary Figs. 1–3, Supplementary Tables 1–8 and Supplementary Protocol
Supplementary Data 1
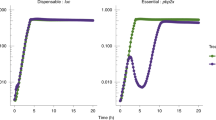
Results of the SorTn-seq differential enrichment analysis
Source data
Source Data Fig. 6
Number of cells sorted into each bin during replicate library sorting
Rights and permissions
About this article
Cite this article
Smith, L.M., Jackson, S.A., Gardner, P.P. et al. SorTn-seq: a high-throughput functional genomics approach to discovering regulators of bacterial gene expression. Nat Protoc 16, 4382–4418 (2021). https://doi.org/10.1038/s41596-021-00582-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-021-00582-6
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.