Abstract
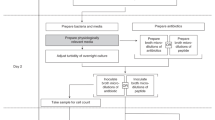
Bacterial biofilms demonstrate high broad-spectrum adaptive antibiotic resistance and cause two thirds of all infections, but there is a lack of approved antibiofilm agents. Unlike the standard minimal inhibitory concentration assay to assess antibacterial activity against planktonic cells, there is no standardized method to evaluate biofilm inhibition and/or eradication capacity of novel antibiofilm compounds. The protocol described here outlines simple and reproducible methods for assessing the biofilm inhibition and eradication capacities of novel antibiofilm agents against adherent bacterial biofilms grown in 96-well microtiter plates. It employs two inexpensive dyes: crystal violet to stain adhered biofilm biomass and 2,3,5-triphenyl tetrazolium chloride to quantify metabolism of the biofilm cells. The procedure is accessible to any laboratory with a plate reader, requires minimal technical expertise or training and takes 4 or 5 d to complete. Recommendations for how biofilm inhibition and eradication results should be interpreted and presented are also described.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The authors declare that all data generated or analyzed in this study are included in the article or in the accompanying supplementary information file. Source data are provided with this paper.
References
Flemming, H.-C. et al. Biofilms: an emergent form of bacterial life. Nat. Rev. Microbiol. 14, 563–575 (2016).
Fernández, L., Breidenstein, E. B. M. & Hancock, R. E. W. Creeping baselines and adaptive resistance to antibiotics. Drug Resist. Updat. 14, 1–21 (2011).
Bryers, J. D. Medical biofilms. Biotechnol. Bioeng. 100, 1–18 (2008).
Bjarnsholt, T. The role of bacterial biofilms in chronic infections. APMIS 121, 1–58 (2013).
Verderosa, A. D., Totsika, M. & Fairfull-Smith, K. E. Bacterial biofilm eradication agents: a current review. Front. Chem. 7, 824 (2019).
Coenye, T., Kjellerup, B., Stoodley, P. & Bjarnsholt, T. The future of biofilm research—report on the ‘2019 Biofilm Bash’. Biofilm 2, 100012 (2020).
Römling, U. & Balsalobre, C. Biofilm infections, their resilience to therapy and innovative treatment strategies. J. Intern. Med. 272, 541–561 (2012).
Taff, H. T., Nett, J. E. & Andes, D. R. Comparative analysis of Candida biofilm quantitation assays. Med. Mycol. 50, 214–218 (2012).
Haney, E., Trimble, M., Cheng, J., Vallé, Q. & Hancock, R. Critical assessment of methods to quantify biofilm growth and evaluate antibiofilm activity of host defence peptides. Biomolecules 8, 29 (2018).
Wiegand, I., Hilpert, K. & Hancock, R. E. W. Agar and broth dilution methods to determine the minimal inhibitory concentration (MIC) of antimicrobial substances. Nat. Protoc. 3, 163–175 (2008).
Beveridge, T. Use of the Gram stain in microbiology. Biotech. Histochem. 76, 111–118 (2001).
Berridge, M. V., Herst, P. M. & Tan, A. S. Tetrazolium dyes as tools in cell biology: New insights into their cellular reduction. Biotechnol. Annu. Rev. 11, 127–152 (2005).
Sabaeifard, P., Abdi-Ali, A., Soudi, M. R. & Dinarvand, R. Optimization of tetrazolium salt assay for Pseudomonas aeruginosa biofilm using microtiter plate method. J. Microbiol. Methods 105, 134–140 (2014).
Azeredo, J. et al. Critical review on biofilm methods. Crit. Rev. Microbiol. 43, 313–351 (2017).
Peeters, E., Nelis, H. J. & Coenye, T. Comparison of multiple methods for quantification of microbial biofilms grown in microtiter plates. J. Microbiol. Methods 72, 157–165 (2008).
Haney, E. F. et al. Computer-aided discovery of peptides that specifically attack bacterial biofilms. Sci. Rep. 8, 1871 (2018).
Haney, E. F., Mansour, S., Hilchie, A. L., de la Fuente-Núñez, C. & Hancock, R. E. W. High throughput screening methods for assessing antibiofilm and immunomodulatory activities of synthetic peptides. Peptides 71, 276–285 (2015).
de la Fuente-Núñez, C. et al. Inhibition of bacterial biofilm formation and swarming motility by a small synthetic cationic peptide. Antimicrob. Agents Chemother. 56, 2696–2704 (2012).
de la Fuente-Núñez, C., Reffuveille, F., Haney, E. F., Straus, S. K. & Hancock, R. E. W. Broad-spectrum anti-biofilm peptide that targets a cellular stress response. PLoS Pathog. 10, (2014).
Haney, E. F., Barbosa, S. C., Baquir, B. & Hancock, R. E. W. Influence of non-natural cationic amino acids on the biological activity profile of innate defense regulator peptides. J. Med. Chem. 62, 10294–10304 (2019).
Ommen, P., Zobek, N. & Meyer, R. L. Quantification of biofilm biomass by staining: Non-toxic safranin can replace the popular crystal violet. J. Microbiol. Methods 141, 87–89 (2017).
Toté, K., Berghe, D. V., Maes, L. & Cos, P. A new colorimetric microtitre model for the detection of Staphylococcus aureus biofilms. Lett. Appl. Microbiol. 46, 249–254 (2008).
Rajamani, S. et al. Robust biofilm assay for quantification and high throughput screening applications. J. Microbiol. Methods 159, 179–185 (2019).
Navarro, G. et al. Image-based 384-well high-throughput screening method for the discovery of skyllamycins A to C as biofilm inhibitors and inducers of biofilm detachment in Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 58, 1092–1099 (2014).
Tremblay, Y. D. N., Vogeleer, P., Jacques, M. & Harel, J. High-throughput microfluidic method to study biofilm formation and host-pathogen interactions in pathogenic Escherichia coli. Appl. Environ. Microbiol. 81, 2827–2840 (2015).
Bridier, A., Dubois-Brissonnet, F., Boubetra, A., Thomas, V. & Briandet, R. The biofilm architecture of sixty opportunistic pathogens deciphered using a high throughput CLSM method. J. Microbiol. Methods 82, 64–70 (2010).
Bernardi, T. et al. High-throughput screening of metal-N-heterocyclic carbene complexes against biofilm formation by pathogenic bacteria. ChemMedChem 9, 1140–1144 (2014).
Olivares, E. et al. The BioFilm Ring Test: a rapid method for routine analysis of Pseudomonas aeruginosa biofilm formation kinetics. J. Clin. Microbiol. 54, 657–661 (2016).
Ceri, H. et al. The Calgary Biofilm Device: new technology for rapid determination of antibiotic susceptibilities of bacterial biofilms. J. Clin. Microbiol. 37, 1771–1776 (1999).
Harrison, J. J. et al. Microtiter susceptibility testing of microbes growing on peg lids: a miniaturized biofilm model for high-throughput screening. Nat. Protoc. 5, 1236–1254 (2010).
Kragh, K. N., Alhede, M., Kvich, L. & Bjarnsholt, T. Into the well—a close look at the complex structures of a microtiter biofilm and the crystal violet assay. Biofilm 1, 100006 (2019).
Trunk, T., Khalil, H. S. & Leo, J. C. Bacterial autoaggregation. AIMS Microbiol 4, 140–164 (2018).
Kragh, K. N. et al. Role of multicellular aggregates in biofilm formation. mBio 7, (2016).
Harrison, J. J. et al. The use of microscopy and three-dimensional visualization to evaluate the structure of microbial biofilms cultivated in the Calgary Biofilm Device. Biol. Proced. Online 8, 194–215 (2006).
Molina-Manso, D. et al. In vitro susceptibility to antibiotics of staphylococci in biofilms isolated from orthopaedic infections. Int. J. Antimicrob. Agents 41, 521–523 (2013).
Ali, L., Khambaty, F. & Diachenko, G. Investigating the suitability of the Calgary Biofilm Device for assessing the antimicrobial efficacy of new agents. Bioresour. Technol. 97, 1887–1893 (2006).
Wong, H. S., Townsend, K. M., Fenwick, S. G., Trengove, R. D. & O’Handley, R. M. Comparative susceptibility of planktonic and 3-day-old Salmonella Typhimurium biofilms to disinfectants. J. Appl. Microbiol. 108, 2222–2228 (2010).
Rivardo, F., Martinotti, M. G., Turner, R. J. & Ceri, H. The activity of silver against Escherichia coli biofilm is increased by a lipopeptide biosurfactant. Can. J. Microbiol. 56, 272–278 (2010).
Karygianni, L., Ren, Z., Koo, H. & Thurnheer, T. Biofilm matrixome: extracellular components in structured microbial communities. Trends Microbiol 28, 668–681 (2020).
Karunakaran, E., Mukherjee, J., Ramalingam, B. & Biggs, C. A. “Biofilmology”: a multidisciplinary review of the study of microbial biofilms. Appl. Microbiol. Biotechnol. 90, 1869–1881 (2011).
Benov, L. Effect of growth media on the MTT colorimetric assay in bacteria. PLoS ONE 14, e0219713 (2019).
Colquhoun, J. M., Wozniak, R. A. F. & Dunman, P. M. Clinically relevant growth conditions alter Acinetobacter baumannii antibiotic susceptibility and promote identification of novel antibacterial agents. PLoS ONE 10, e0143033 (2015).
Seneviratne, C. J., Yip, J. W. Y., Chang, J. W. W., Zhang, C. F. & Samaranayake, L. P. Effect of culture media and nutrients on biofilm growth kinetics of laboratory and clinical strains of Enterococcus faecalis. Arch. Oral Biol. 58, 1327–1334 (2013).
Palmer, K. L., Aye, L. M. & Whiteley, M. Nutritional cues control Pseudomonas aeruginosa multicellular behavior in cystic fibrosis sputum. J. Bacteriol. 189, 8079–8087 (2007).
Pompilio, A. et al. Potential novel therapeutic strategies in cystic fibrosis: antimicrobial and anti-biofilm activity of natural and designed α-helical peptides against Staphylococcus aureus, Pseudomonas aeruginosa, and Stenotrophomonas maltophilia. BMC Microbiol 12, 145 (2012).
Haney, E. F. et al. Aggregation and its influence on the immunomodulatory activity of synthetic innate defense regulator peptides. Cell Chem. Biol. 24, 969–980.e4 (2017).
O’Toole, G. A. Microtiter dish biofilm formation assay. J. Vis. Exp. https://doi.org/10.3791/2437 (2011).
Coffey, B. M. & Anderson, G. G. in Pseudomonas Methods and Protocols (eds Filloux, A. & Ramos, J.-L.) 631–641 (Springer, 2014).
de la Fuente-Núñez, C. et al. D-enantiomeric peptides that eradicate wild-type and multidrug-resistant biofilms and protect against lethal Pseudomonas aeruginosa infections. Chem. Biol. 22, 196–205 (2015).
McBain, A. J. in Advances in Applied Microbiology Vol. 69, Ch. 4, 99–132 (Academic Press, 2009).
Lebeaux, D., Chauhan, A., Rendueles, O. & Beloin, C. From in vitro to in vivo models of bacterial biofilm-related infections. Pathogens 2, 288–356 (2013).
Acknowledgements
Our own biofilm work was supported by the Canadian Institutes of Health Research (CIHR), funding reference number FDN-154287. REWH holds a Canada Research Chair in Health and Genomics and a UBC Killam Professorship.
Author information
Authors and Affiliations
Contributions
E.F.H. and M.J.T. developed the protocols, collected and analyzed data, and wrote and edited the first draft of the manuscript. R.E.W.H. was involved in development of the protocol and extensively edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
E.F.H. and R.E.W.H. have invented and filed for patent protection on related antibiofilm peptide sequences. This patent has been assigned to their employer, the University of British Columbia, and licensed to ABT Innovations, in which R.E.W.H. has an ownership position. M.J.T. declares no competing interests.
Additional information
Peer review information Nature Protocols thanks Tom Coenye and Henny C. van der Mei for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
Haney, E. F., Trimble, M. J., Cheng, J. T., Vallé, Q. & Hancock, R. E. Biomolecules 8, 29 (2018): https://doi.org/10.3390/biom8020029
Haney, E. F. et al. Sci. Rep. 8, 1871 (2018): https://doi.org/10.1038/s41598-018-19669-4
Haney, E. F., Barbosa, S. C., Baquir, B. & Hancock, R. E. W. J. Med. Chem. 62, 10294–10304 (2019): https://doi.org/10.1021/acs.jmedchem.9b01344
Supplementary information
Supplementary Information
Supplementary Figs. 1–6.
Supplementary Data 1
Data for Supplementary Figs. 2, 4 and 5
Source data
Rights and permissions
About this article
Cite this article
Haney, E.F., Trimble, M.J. & Hancock, R.E.W. Microtiter plate assays to assess antibiofilm activity against bacteria. Nat Protoc 16, 2615–2632 (2021). https://doi.org/10.1038/s41596-021-00515-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-021-00515-3
This article is cited by
-
Quenching of quorum sensing in multi-drug resistant Pseudomonas aeruginosa: insights on halo-bacterial metabolites and gamma irradiation as channels inhibitors
Annals of Clinical Microbiology and Antimicrobials (2024)
-
Antibiofilm activity of marine microbial natural products: potential peptide- and polyketide-derived molecules from marine microbes toward targeting biofilm-forming pathogens
Journal of Natural Medicines (2024)
-
Extraction, characterization, and therapeutic potential of Omega-3 fatty acids from Belone belone skin
3 Biotech (2024)
-
Comprehensive analysis of Seriphidium kurramense: GC/MS profiling, antibacterial and antibiofilm activities, molecular docking study and in-silico ADME profiling
Discover Applied Sciences (2024)
-
Artificial intelligence-based optimization for chitosan nanoparticles biosynthesis, characterization and in‑vitro assessment of its anti-biofilm potentiality
Scientific Reports (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.