Abstract
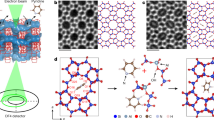
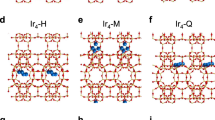
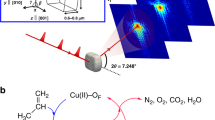
The encapsulation of subnanometric metal entities (isolated metal atoms and metal clusters with a few atoms) in porous materials such as zeolites can be an effective strategy for the stabilization of those metal species and therefore can be further used for a variety of catalytic reactions. However, owing to the complexity of zeolite structures and their low stability under the electron beam, it is challenging to obtain atomic-level structural information of the subnanometric metal species encapsulated in zeolite crystallites. In this protocol, we show the application of a scanning transmission electron microscopy (STEM) technique that records simultaneously the high-angle annular dark-field (HAADF) images and integrated differential phase-contrast (iDPC) images for structural characterization of subnanometric Pt and Sn species within MFI zeolite. The approach relies on the use of a computational model to simulate results obtained under different conditions where the metals are present in different positions within the zeolite. This imaging technique allows to obtain simultaneously the spatial information of heavy elements (Pt and Sn in this work) and the zeolite framework structure, enabling direct determination of the location of the subnanometric metal species. Moreover, we also present the combination of other spectroscopy techniques as complementary tools for the STEM–iDPC imaging technique to obtain global understanding and insights on the spatial distributions of subnanometric metal species in zeolite structure. These structural insights can provide guidelines for the rational design of uniform metal–zeolite materials for catalytic applications.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



























Similar content being viewed by others
Data availability
Data supporting this publication are available from the corresponding author upon reasonable request.
Code availability
The code and scripts used in this work are available from the corresponding author upon reasonable request.
References
Flytzani-Stephanopoulos, M. & Gates, B. C. Atomically dispersed supported metal catalysts. Annu. Rev. Chem. Biomol. Eng. 3, 545–574 (2012).
Liu, L. & Corma, A. Metal catalysts for heterogeneous catalysis: from single atoms to nanoclusters and nanoparticles. Chem. Rev. 118, 4981–5079 (2018).
Thomas, J. M., Raja, R. & Lewis, D. W. Single-site heterogeneous catalysts. Angew. Chem. Int. Ed. 44, 6456–6482 (2005).
Hansen, T. W. et al. Atomic-resolution in situ transmission electron microscopy of a promoter of a heterogeneous catalyst. Science 294, 1508–1510 (2001).
Hwang, S., Chen, X., Zhou, G. & Su, D. In situ transmission electron microscopy on energy-related catalysis. Adv. Energy Mater. https://doi.org/10.1002/aenm.201902105 (2019).
Li, Z. et al. Well-defined materials for heterogeneous catalysis: from nanoparticles to isolated single-atom sites. Chem. Rev. 120, 623–682 (2019).
Pelletier, J. D. & Basset, J. M. Catalysis by design: well-defined single-site heterogeneous catalysts. Acc. Chem. Res. 49, 664–677 (2016).
Kulkarni, A., Lobo-Lapidus, R. J. & Gates, B. C. Metal clusters on supports: synthesis, structure, reactivity, and catalytic properties. Chem. Commun. 46, 5997–6015 (2010).
Liu, L. et al. Evolution and stabilization of subnanometric metal species in confined space by in situ TEM. Nat. Commun. 9, 574 (2018).
Gates, B. C., Flytzani-Stephanopoulos, M., Dixon, D. A. & Katz, A. Atomically dispersed supported metal catalysts: perspectives and suggestions for future research. Catal. Sci. Technol. 7, 4259–4275 (2017).
Li, H., Wang, M., Luo, L. & Zeng, J. Static regulation and dynamic evolution of single-atom catalysts in thermal catalytic reactions. Adv. Sci 6, 1801471 (2019).
Liu, L. et al. Regioselective generation and reactivity control of subnanometric platinum clusters in zeolites for high-temperature catalysis. Nat. Mater. 18, 866–873 (2019).
Juneau, M. et al. Characterization of metal–zeolite composite catalysts: determining the environment of the active phase. ChemCatChem https://doi.org/10.1002/cctc.201902039 (2019).
Shamzhy, M., Opanasenko, M., Concepcion, P. & Martinez, A. New trends in tailoring active sites in zeolite-based catalysts. Chem. Soc. Rev. 48, 1095–1149 (2019).
Weckhuysen, B. M. Stable platinum in a zeolite channel. Nat. Mater. 18, 778–779 (2019).
Gai, P. L. & Calvino, J. J. Electron microscopy in the catalysis of alkane oxidation, environmental control, and alternative energy sources. Annu. Rev. Mater. Res. 35, 465–504 (2005).
Gates, B. C. Atomically dispersed supported metal catalysts: seeing is believing. Trends Chem. 1, 99–110 (2019).
Yang, S. et al. Bridging dealumination and desilication for the synthesis of hierarchical MFI zeolites. Angew. Chem. Int. Ed. 56, 12553–12556 (2017).
Lupulescu, A. I. & Rimer, J. D. In situ imaging of silicalite-1 surface growth reveals the mechanism of crystallization. Science 344, 729–732 (2014).
Anderson, M. W. et al. Modern microscopy methods for the structural study of porous materials. Chem. Commun. (8), 907–916 (2004).
Mayoral, A., Carey, T., Anderson, P. A., Lubk, A. & Diaz, I. Atomic resolution analysis of silver ion-exchanged zeolite A. Angew. Chem. Int. Ed. 50, 11230–11233 (2011).
Lu, J., Aydin, C., Browning, N. D. & Gates, B. C. Imaging isolated gold atom catalytic sites in zeolite NaY. Angew. Chem. Int. Ed. 51, 5842–5846 (2012).
Ortalan, V., Uzun, A., Gates, B. C. & Browning, N. D. Direct imaging of single metal atoms and clusters in the pores of dealuminated HY zeolite. Nat. Nanotechnol. 5, 506–510 (2010).
Yang, M. et al. Catalytically active Au-O(OH)x-species stabilized by alkali ions on zeolites and mesoporous oxides. Science 346, 1498–1501 (2014).
Liu, L. et al. Generation of subnanometric platinum with high stability during transformation of a 2D zeolite into 3D. Nat. Mater. 16, 132–138 (2017).
Slater, B., Ohsuna, T., Liu, Z. & Terasaki, O. Insights into the crystal growth mechanisms of zeolites from combined experimental imaging and theoretical studies. Faraday Discussions 136, 125–141 (2007).
Diaz, I. & Mayoral, A. TEM studies of zeolites and ordered mesoporous materials. Micron 42, 512–527 (2011).
Wan, W., Su, J., Zou, X. D. & Willhammar, T. Transmission electron microscopy as an important tool for characterization of zeolite structures. Inorg. Chem. Frontiers 5, 2836–2855 (2018).
Mishra, R., Ishikawa, R., Lupini, A. R. & Pennycook, S. J. Single-atom dynamics in scanning transmission electron microscopy. MRS Bulletin 42, 644–652 (2017).
DeLaRiva, A. T., Hansen, T. W., Challa, S. R. & Datye, A. K. In situ transmission electron microscopy of catalyst sintering. J. Catal. 308, 291–305 (2013).
Lazic, I., Bosch, E. G. T. & Lazar, S. Phase contrast STEM for thin samples: integrated differential phase contrast. Ultramicroscopy 160, 265–280 (2016).
Shen, B. et al. Atomic spatial and temporal imaging of local structures and light elements inside zeolite frameworks. Adv. Mater. e1906103 (2019).
Egerton, R. F. & Watanabe, M. Characterization of single-atom catalysts by EELS and EDX spectroscopy. Ultramicroscopy 193, 111–117 (2018).
Bonilla, G. et al. Zeolite (MFI) crystal morphology control using organic structure-directing agents. Chem. Mater. 16, 5697–5705 (2004).
Karwacki, L. et al. Morphology-dependent zeolite intergrowth structures leading to distinct internal and outer-surface molecular diffusion barriers. Nat. Mater. 8, 959–965 (2009).
Grand, J. et al. One-pot synthesis of silanol-free nanosized MFI zeolite. Nat. Mater. 16, 1010–1015 (2017).
Varela, M. et al. Materials characterization in the aberration-corrected scanning transmission electron microscope. Annu. Rev. Mater. Res. 35, 539–569 (2005).
Mitchell, DavidR. G. & Mitchell, J. B. Nancarrow probe current determination in analytical TEM/STEM and its application to the characterization of large area EDS detectors. Microscopy Res. Technique 78, 886–893 (2015).
Erni, R. Aberration-Corrected Imaging in Transmission Electron Microscopy (Imperial College Press, 2010).
Díaz, I., Kokkoli, E., Terasaki, O. & Tsapatsis, M. Surface structure of zeolite (MFI) crystals. Chem. Mater. 16, 5226–5232 (2004).
Bernal, S. et al. The interpretation of HREM images of supported metal catalysts using image simulation: profile view images. Ultramicroscopy 72, 135–164 (1998).
Kirkland, E. J. Advanced Computing in Electron Microscopy (Springer, 2010).
Corma, A., Fornes, V., Pergher, S. B., Maesen, T. L. M. & Buglass, J. G. Delaminated zeolite precursors as selective acidic catalysts. Nature 396, 353–356 (1998).
Pei, Y. et al. Catalytic properties of intermetallic platinum–tin nanoparticles with non-stoichiometric compositions. J. Catal. 374, 136–142 (2019).
Moliner, M. et al. Reversible transformation of Pt nanoparticles into single atoms inside high-silica chabazite zeolite. J. Am. Chem. Soc. 138, 15743–15750 (2016).
de Graaf, J., van Dillen, A. J., de Jong, K. P. & Koningsberger, D. C. Preparation of highly dispersed Pt particles in zeolite Y with a narrow particle size distribution: characterization by hydrogen chemisorption, TEM, EXAFS spectroscopy, and particle modeling. J. Catal. 203, 307–321 (2001).
Bare, S. R. et al. Uniform catalytic site in Sn-beta-zeolite determined using X-ray absorption fine structure. J. Am. Chem. Soc. 127, 12924–12932 (2005).
Hammond, C. et al. Identification of active and spectator Sn sites in Sn-beta following solid-state stannation, and consequences for Lewis acid catalysis. ChemCatChem 7, 3322–3331 (2015).
Liu, L. et al. Determination of the evolution of heterogeneous single metal atoms and nanoclusters under reaction conditions: which are the working catalytic sites? ACS Catal. 9, 10626–10639 (2019).
Derevyannikova, E. A. et al. Structural insight into strong Pt–CeO2 interaction: from single Pt atoms to PtOx clusters. J. Phys. Chem. C 123, 1320–1334 (2018).
Sun, Q. et al. Subnanometer bimetallic Pt–Zn clusters in zeolites for propane dehydrogenation. Angew. Chem. Int. Ed. https://doi.org/10.1002/anie.202003349 (2020).
Concepcion, P. et al. The promotional effect of Sn-beta zeolites on platinum for the selective hydrogenation of alpha,beta-unsaturated aldehydes. Phys. Chem. Chem. Phys. 15, 12048–12055 (2013).
Heiz, U., Sanchez, A., Abbet, S. & Schneider, W. D. Catalytic oxidation of carbon monoxide on monodispersed platinum clusters: each atom counts. J. Am. Chem. Soc. 121, 3214–3217 (1999).
Serykh, A. I. et al. Stable subnanometre Pt clusters in zeolite NaX via stoichiometric carbonyl complexes: probing of negative charge by DRIFT spectroscopy of adsorbed CO and H2. Phys. Chem. Chem. Phys. 2, 5647–5652 (2000).
Drozdová, L. et al. Subnanometer platinum clusters in zeolite NaEMT via stoichiometric carbonyl clusters. Microporous Mesoporous Mater. 35–36, 511–519 (2000).
Mishra, D. K., Dabbawala, A. A. & Hwang, J.-S. Ruthenium nanoparticles supported on zeolite Y as an efficient catalyst for selective hydrogenation of xylose to xylitol. J. Mol. Catal. A: Chem. 376, 63–70 (2013).
Visser, T. et al. Promotion effects in the oxidation of CO over zeolite-supported Pt nanoparticles. J. Phys. Chem. B 109, 3822–3831 (2005).
Rivallan, M. et al. Platinum sintering on H-ZSM-5 followed by chemometrics of CO adsorption and 2D pressure-jump IR spectroscopy of adsorbed species. Angew. Chem. Int. Ed. 49, 785–789 (2010).
Balakrishnan, K. A chemisorption and XPS study of bimetallic Pt-Sn/Al2O3 catalysts. J. Catal. 127, 287–306 (1991).
Panja, C. & Koel, B. E. Probing the influence of alloyed Sn on Pt(100) surface chemistry by CO chemisorption. Israel J. Chem 38, 365–374 (1998).
Chen, Y. et al. Single-atom catalysts: synthetic strategies and electrochemical applications. Joule 2, 1242–1264 (2018).
Wang, A., Li, J. & Zhang, T. Heterogeneous single-atom catalysis. Nat. Rev. Chem. 2, 65–81 (2018).
Zhang, H., Liu, G., Shi, L. & Ye, J. Single-atom catalysts: emerging multifunctional materials in heterogeneous catalysis. Adv. Energy Mater. 8, 1701343 (2018).
Wiktor, C., Meledina, M., Turner, S., Lebedev, O. I. & Fischer, R. A. Transmission electron microscopy on metal–organic frameworks – a review. J. Mater. Chem. A 5, 14969–14989 (2017).
Liu, L. et al. Direct imaging of atomically dispersed molybdenum that enables location of aluminum in the framework of zeolite ZSM-5. Angew. Chem. Int. Ed. 59, 819–825 (2020).
Zuo, Q. et al. Ultrathin metal–organic framework nanosheets with ultrahigh loading of single Pt atoms for efficient visible-light-driven photocatalytic H2 evolution. Angew. Chem. Int. Ed. 58, 10198–10203 (2019).
Liu, L. et al. Imaging defects and their evolution in a metal-organic framework at sub-unit-cell resolution. Nat. Chem. 11, 622–628 (2019).
Zhou, Y. et al. Local structure evolvement in MOF single crystals unveiled by scanning transmission electron microscopy. Chem. Mater. 32, 4966–4972 (2020).
Borisevich, A. Y., Lupini, A. R. & Pennycook, S. J. Depth sectioning with the aberration-corrected scanning transmission electron microscope. Proc. Natl Acad. Sci. USA 103, 3044–3048 (2006).
Zecevic, J., van der Eerden, A. M., Friedrich, H., de Jongh, P. E. & de Jong, K. P. Heterogeneities of the nanostructure of platinum/zeolite y catalysts revealed by electron tomography. ACS Nano 7, 3698–3705 (2013).
Schmidt, J. E., Oord, R., Guo, W., Poplawsky, J. D. & Weckhuysen, B. M. Nanoscale tomography reveals the deactivation of automotive copper-exchanged zeolite catalysts. Nat. Commun. 8, 1666 (2017).
Chen, Z. et al. Direct synthesis of core-shell MFI zeolites with spatially tapered trimodal mesopores via controlled orthogonal self-assembly. Nanoscale 11, 16667–16676 (2019).
Kliewer, C. E. in Zeolite Characterization and Catalysis (eds. Chester, A. W. & Derouane, E. G.) (Springer, 2009).
Deng, D. et al. Catalysis with two-dimensional materials and their heterostructures. Nat. Nanotechnol. 11, 218–230 (2016).
Li, Z. et al. Reactive metal–support interactions at moderate temperature in two-dimensional niobium-carbide-supported platinum catalysts. Nat. Catal. 1, 349–355 (2018).
Goldsmith, B. R., Peters, B., Johnson, J. K., Gates, B. C. & Scott, S. L. Beyond ordered materials: understanding catalytic sites on amorphous solids. ACS Catal. 7, 7543–7557 (2017).
van Deelen, T. W., Hernández Mejía, C. & de Jong, K. P. Control of metal–support interactions in heterogeneous catalysts to enhance activity and selectivity. Nat. Catal. 2, 955–970 (2019).
Hodnik, N., Dehm, G. & Mayrhofer, K. J. Importance and challenges of electrochemical in situ liquid cell electron microscopy for energy conversion research. Acc. Chem. Res. 49, 2015–2022 (2016).
Boyes, E. D., LaGrow, A. P., Ward, M. R., Mitchell, R. W. & Gai, P. L. Single atom dynamics in chemical reactions. Acc. Chem. Res. 53, 390–399 (2020).
Nakamura, E. Atomic-resolution transmission electron microscopic movies for study of organic molecules, assemblies, and reactions: the first 10 years of development. Acc. Chem. Res. 50, 1281–1292 (2017).
Li, T. et al. Cryo-TEM and electron tomography reveal leaching-induced pore formation in ZSM-5 zeolite. J. Mater. Chem. A 7, 1442–1446 (2019).
Li, Y. et al. Cryo-EM structures of atomic surfaces and host–guest chemistry in metal-organic frameworks. Matter 1, 428–438 (2019).
Liu, L. & Corma, A. Evolution of isolated atoms and clusters in catalysis. Trends Chem. 2, 383–400 (2020).
Barthel, J. Dr. Probe: a software for high-resolution STEM image simulation. Ultramicroscopy 193, 1–11 (2018).
De Wael, A., De Backer, A., Jones, L., Nellist, P. D. & Van Aert, S. Hybrid statistics-simulations based method for atom-counting from ADF STEM images. Ultramicroscopy 177, 69–77 (2017).
Hwang, J., Zhang, J. Y., D’Alfonso, A. J., Allen, L. J. & Stemmer, S. Three-dimensional imaging of individual dopant atoms in SrTiO3. Phys. Rev. Lett. 111, 266101 (2013).
Acknowledgements
This work was supported by the European Union through the European Research Council (grant ERC-AdG-2014-671093, SynCatMatch) and the Spanish government through the ‘Severo Ochoa Program’ (SEV-2016-0683). The authors also thank the Microscopy Service of UPV for the TEM and STEM measurements. The XAS measurements were carried out in CLAESS beamline of ALBA synchrotron. HR STEM measurements were performed at the DME-UCA node of the ELECMI Singular Infrastructure at Cadiz University, with financial support from FEDER/MINECO (MAT2017-87579-R and MAT2016-81118-P). The authors thank C. W. Lopes and P. Concepcion for their help with the analysis of spectroscopic results. The financial support from ExxonMobil on this project is also gratefully acknowledged.
Author information
Authors and Affiliations
Contributions
A.C. conceived the project and directed the study. L.L. carried out the synthesis and characterizations of the Pt–zeolite materials. M.L.-H. and J.J.C. carried out the HR STEM measurements, image analysis and simulations, with assistance from L.L. All authors discussed the results and contributed to the formation of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Protocols thanks M. A. Goula, Laurent Piccolo and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key reference using this protocol
Liu, L. et al. Nat. Mater. 18, 866−873 (2019): https://www.nature.com/articles/s41563-019-0412-6
Key data used in this protocol
Liu, L. et al. Nat. Mater. 18, 866−873 (2019): https://www.nature.com/articles/s41563-019-0412-6
Supplementary information
Rights and permissions
About this article
Cite this article
Liu, L., Lopez-Haro, M., Calvino, J.J. et al. Tutorial: structural characterization of isolated metal atoms and subnanometric metal clusters in zeolites. Nat Protoc 16, 1871–1906 (2021). https://doi.org/10.1038/s41596-020-0366-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-020-0366-9
This article is cited by
-
Quantitative Evaluation of Supported Catalysts Key Properties from Electron Tomography Studies: Assessing Accuracy Using Material-Realistic 3D-Models
Topics in Catalysis (2022)
-
Solid catalysts for the dehydrogenation of long-chain alkanes: lessons from the dehydrogenation of light alkanes and homogeneous molecular catalysis
Science China Chemistry (2022)
-
Identification of the active sites in supported subnanometric metal catalysts
Nature Catalysis (2021)
-
Regioirregular and catalytic Mizoroki–Heck reactions
Nature Catalysis (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.