Abstract
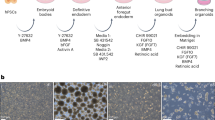
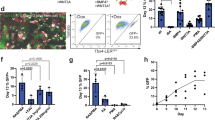
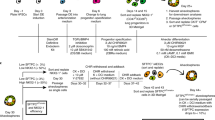
Lung and airway epithelial cells generated in vitro from human pluripotent stem cells (hPSCs) have applications in regenerative medicine, modeling of lung disease, drug screening and studies of human lung development. Here, we describe a strategy for directed differentiation of hPSCs into mature lung and airway epithelial cells obtained through maturation of NKX2.1+ hPSC-derived lung progenitors in a 3D matrix of collagen I in the absence of glycogen synthase kinase 3 inhibition. This protocol is an extension of our previously published protocol on the directed differentiation of lung and airway epithelium from hPSCs that modifies the technique and offers additional applications. This protocol is conducted in defined media conditions, has a duration of 50–80 d, does not require reporter lines and results in cultures containing mature alveolar type II and I cells as well as airway basal, ciliated, club and neuroendocrine cells. We also present a flow cytometry strategy to assess maturation in the cultures. Several of these populations, including mature NGFR+ basal cells, can be prospectively isolated by cell sorting and expanded for further investigation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
Data availability
The RNA sequencing and single-cell RNA sequencing data are deposited in GEO under accession number GSE101558. Example flow cytometry data sets have been uploaded to the flow cytometry repository (http://flowrepository.org) under the following repository IDs: FR-FCM-Z2VH, FR-FCM-Z2VM, FR-FCM-Z2VN, FR-FCM-Z3Z6 and FR-FCM-Z3Z7. Raw data supporting the findings presented in the figures are available in the accompanying source data files.
References
Green, M. D., Huang, S. X. L. & Snoeck, H. W. Stem cells of the respiratory system: from identification to differentiation into functional epithelium. BioEssays 35, 261–270 (2013).
Green, M. D. et al. Generation of anterior foregut endoderm from human embryonic and induced pluripotent stem cells. Nat. Biotechnol. 29, 267–272 (2011).
Mou, H. et al. Generation of multipotent lung and airway progenitors from mouse ESCs and patient-specific cystic fibrosis iPSCs. Cell Stem Cell 10, 385–397 (2012).
Wong, A. P. et al. Directed differentiation of human pluripotent stem cells into mature airway epithelia expressing functional CFTR protein. Nat. Biotechnol. 30, 876–882 (2012).
Huang, S. X. L. et al. Efficient generation of lung and airway epithelial cells from human pluripotent stem cells. Nat. Biotechnol. 32, 84–91 (2014).
Firth, A. L. et al. Generation of multiciliated cells in functional airway epithelia from human induced pluripotent stem cells. Proc. Natl Acad. Sci. USA 111, E1723–E1730 (2014).
Gotoh, S. et al. Generation of alveolar epithelial spheroids via isolated progenitor cells from human pluripotent stem cells. Stem Cell Rep. 3, 394–403 (2014).
Konishi, S. et al. Directed induction of functional multi-ciliated cells in proximal airway epithelial spheroids from human pluripotent stem cells. Stem Cell Rep. 6, 18–25 (2016).
Chen, Y.-W. et al. A three-dimensional model of human lung development and disease from pluripotent stem cells. Nat. Cell Biol. 19, 542–549 (2017).
McCauley, K. B. et al. Efficient derivation of functional human airway epithelium from pluripotent stem cells via temporal regulation of Wnt signaling. Cell Stem Cell 20, 844–857.e6 (2017).
Jacob, A. et al. Differentiation of human pluripotent stem cells into functional lung alveolar epithelial cells. Cell Stem Cell 21, 472–488.e10 (2017).
Yamamoto, Y. et al. Long-term expansion of alveolar stem cells derived from human iPS cells in organoids. Nat. Methods 14, 1097–1106 (2017).
de Carvalho, A. L. R. T. et al. Glycogen synthase kinase 3 induces multilineage maturation of human pluripotent stem cell-derived lung progenitors in 3D culture. Development 146, dev171652 (2019).
Dye, B. R. et al. In vitro generation of human pluripotent stem cell derived lung organoids. eLife 4, e05098 (2015).
Miller, A. J. et al. Generation of lung organoids from human pluripotent stem cells in vitro. Nat. Protoc. 14, 518–540 (2019).
Dye, B. R., Miller, A. J. & Spence, J. R. How to grow a lung: applying principles of developmental biology to generate lung lineages from human pluripotent stem cells. Curr. Pathobiol. Rep. 4, 47–57 (2016).
Hawkins, F. & Kotton, D. N. Embryonic and induced pluripotent stem cells for lung regeneration. Ann. Am. Thorac. Soc. 12, S50–S53 (2015).
Swarr, D. T. & Morrisey, E. E. Lung endoderm morphogenesis: gasping for form and function. Annu. Rev. Cell Dev. Biol. 31, 553–573 (2015).
Huang, S. X. L. et al. The in vitro generation of lung and airway progenitor cells from human pluripotent stem cells. Nat. Protoc. 10, 413–425 (2015).
Rock, J. R. et al. Basal cells as stem cells of the mouse trachea and human airway epithelium. Proc. Natl Acad. Sci. USA 106, 12771–12775 (2009).
Hawkins, F. et al. Prospective isolation of NKX2-1–expressing human lung progenitors derived from pluripotent stem cells. J. Clin. Invest 127, 2277–2294 (2017).
Strikoudis, A. et al. Modeling of fibrotic lung disease using 3D organoids derived from human pluripotent stem cells. Cell Rep. 27, 3709–3723.e5 (2019).
Dye, B. R. et al. A bioengineered niche promotes in vivo engraftment and maturation of pluripotent stem cell derived human lung organoids. eLife 5, e19732 (2016).
Butler, C. R. et al. Rapid expansion of human epithelial stem cells suitable for airway tissue engineering. Am. J. Respir. Crit. Care Med 194, 156–168 (2016).
Ramani, S., Crawford, S. E., Blutt, S. E. & Estes, M. K. Human organoid cultures: transformative new tools for human virus studies. Curr. Opin. Virol. 29, 79–86 (2018).
Ciancanelli, M. J. et al. Infectious disease. Life-threatening influenza and impaired interferon amplification in human IRF7 deficiency. Science 348, 448–453 (2015).
Rafeeq, M. M. & Murad, H. A. S. Cystic fibrosis: current therapeutic targets and future approaches. J. Transl. Med. 15, 84 (2017).
Kaur, A., Mathai, S. K. & Schwartz, D. A. Genetics in idiopathic pulmonary fibrosis pathogenesis, prognosis, and treatment. Front. Med 4, 154 (2017).
Whitsett, J. A., Wert, S. E. & Weaver, T. E. Alveolar surfactant homeostasis and the pathogenesis of pulmonary disease. Annu. Rev. Med 61, 105–119 (2010).
Kim, C. F. B. et al. Identification of bronchioalveolar stem cells in normal lung and lung. cancer Cell 121, 823–835 (2005).
Xu, X. et al. Evidence for type II cells as cells of origin of K-Ras-induced distal lung adenocarcinoma. Proc. Natl Acad. Sci. USA 109, 4910–4915 (2012).
Chen, H. J. et al. Generation of pulmonary neuroendocrine cells and SCLC-like tumors from human embryonic stem cells. J. Exp. Med 216, 674–687 (2019).
Chen, Z. et al. Genetic engineering of human embryonic stem cells for precise cell fate tracing during human lineage development. Stem Cell Rep. 11, 1257–1271 (2018).
An, W. F. et al. Discovery of Potent and Highly Selective Inhibitors of GSK3b (National Center for Biotechnology Information, 2010).
Gonzales, L. W., Guttentag, S. H., Wade, K. C., Postle, A. D. & Ballard, P. L. Differentiation of human pulmonary type II cells in vitro by glucocorticoid plus cAMP. Am. J. Physiol. Lung Cell. Mol. Physiol. 283, L940–L951 (2002).
Hynds, R. E., Butler, C. R., Janes, S. M. & Giangreco, A. Expansion of human airway basal stem cells and their differentiation as 3D tracheospheres. Methods Mol. Biol. 1576, 43–53 (2019).
Gonzalez, R. F., Allen, L., Gonzales, L., Ballard, P. L. & Dobbs, L. G. HTII-280, a biomarker specific to the apical plasma membrane of human lung alveolar type II cells. J. Histochem. Cytochem. 58, 891–901 (2010).
Barkauskas, C. E. et al. Type 2 alveolar cells are stem cells in adult lung. J. Clin. Invest 123, 3025–3036 (2013).
Dobbs, L. G., Gonzalez, R. F., Allen, L. & Froh, D. K. HTI 56, an integral membrane protein specific to human alveolar type I cells. J. Histochem. Cytochem 47, 129–137 (1999).
Jarrard, J. A. et al. MUC1 is a novel marker for the type II pneumocyte lineage during lung carcinogenesis. Cancer Res 58, 5582–5589 (1998).
Ramirez, M. I. et al. T1α, a lung type I cell differentiation gene, is required for normal lung cell proliferation and alveolus formation at birth. Dev. Biol. 256, 62–73 (2003).
Rock, J. R. et al. Notch-dependent differentiation of adult airway basal stem cells. Cell Stem Cell 8, 639–648 (2011).
Kim, K. C. & Lillehoj, E. P. MUC1 mucin: a peacemaker in the lung. Am. J. Respir. Cell Mol. Biol. 39, 644–647 (2008).
McCauley, K. B. et al. Single-cell transcriptomic profiling of pluripotent stem cell-derived SCGB3A2+ airway epithelium. Stem Cell Rep. 10, 1579–1595 (2018).
Roost, M. S. et al. KeyGenes, a tool to probe tissue differentiation using a human fetal transcriptional atlas. Stem Cell Rep. 4, 1112–1124 (2015).
Lindahl, M., Ståhlbom, B. & Tagesson, C. Newly identified proteins in human nasal and bronchoalveolar lavage fluids: potential biomedical and clinical applications. Electrophoresis 20, 3670–3676 (1999).
Porotto, M. et al. Authentic modeling of human respiratory virus infection in human pluripotent stem cell-derived lung organoids. mBio 10, e00723-19 (2019).
Branche, A. & Falsey, A. Parainfluenza virus infection. Semin. Respir. Crit. Care Med 37, 538–554 (2016).
Henrickson, K. J. Parainfluenza viruses. Clin. Microbiol. Rev. 16, 242–264 (2003).
Schomacker, H., Schaap-Nutt, A., Collins, P. L. & Schmidt, A. C. Pathogenesis of acute respiratory illness caused by human parainfluenza viruses. Curr. Opin. Virol. 2, 294–299 (2012).
Zhang, L. et al. Infection of ciliated cells by human parainfluenza virus type 3 in an in vitro model of human airway epithelium. J. Virol. 79, 1113–1124 (2005).
Acknowledgements
This work was supported by the US National Institutes of Health grants HL120046 and 1U01HL134760 (to H.-W.S.) and AI31971 (to A.M.), the Thomas R. Kully IPF Research Fund (to H.-W.S.) and the Fundação para a Ciência e a Tecnologia (fellowship PD/BD/52320/2013 to A.L.R.T.d.C.). Flow cytometry was performed in the CCTI Flow Cytometry Core, supported in part by the Office of the Director, National Institutes of Health under awards S10RR027050 and S10OD020056.
Author information
Authors and Affiliations
Contributions
A.L.R.T.d.C. developed the lung maturation protocol, contributed to the concept and co-wrote the manuscript. Y.-W.C. and H.-Y.L. contributed to the development of the protocol. A.M. and M.P. generated and provided virology reagents, contributed to the design and provided instructions for HPIV infection experiments. H.-W.S. developed the concept, contributed to protocol development and co-wrote the manuscript with A.L.R.T.d.C.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Protocols thanks the anonymous reviewers for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
de Carvalho, A. L. R. T. et al. Development 146, dev171652 (2019): https://doi.org/10.1242/DEV.171652
Huang, S. X. L. et al. Nat. Biotechnol. 32, 84–91 (2014): https://doi.org/10.1038/nbt.2754
This protocol is an extension to: Nat. Protoc. 10, 413–425 (2015): https://doi.org/10.1038/nprot.2015.023
Supplementary information
Supplementary Information
Supplementary Figs. 1–6, Supplementary Tables 1–3 and Supplementary Methods.
Supplementary Data 1
Source data for Supplementary Table 2 (flow cytometry) and Supplementary Table 3 (flow cytometry).
Source data
Source Data Fig. 8
RT-aPCR data for Fig. 8d.
Rights and permissions
About this article
Cite this article
Rodrigues Toste de Carvalho, A.L., Liu, HY., Chen, YW. et al. The in vitro multilineage differentiation and maturation of lung and airway cells from human pluripotent stem cell–derived lung progenitors in 3D. Nat Protoc 16, 1802–1829 (2021). https://doi.org/10.1038/s41596-020-00476-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-020-00476-z
This article is cited by
-
Endogenous retroviral ERVH48-1 promotes human urine cell reprogramming
Cell Regeneration (2024)
-
A distal lung organoid model to study interstitial lung disease, viral infection and human lung development
Nature Protocols (2023)
-
Reconstructing the pulmonary niche with stem cells: a lung story
Stem Cell Research & Therapy (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.