Abstract
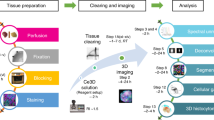
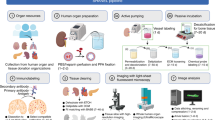
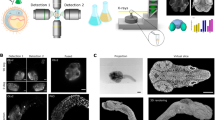
In vitro 3D organoid systems have revolutionized the modeling of organ development and diseases in a dish. Fluorescence microscopy has contributed to the characterization of the cellular composition of organoids and demonstrated organoids’ phenotypic resemblance to their original tissues. Here, we provide a detailed protocol for performing high-resolution 3D imaging of entire organoids harboring fluorescence reporters and upon immunolabeling. This method is applicable to a wide range of organoids of differing origins and of various sizes and shapes. We have successfully used it on human airway, colon, kidney, liver and breast tumor organoids, as well as on mouse mammary gland organoids. It includes a simple clearing method utilizing a homemade fructose–glycerol clearing agent that captures 3D organoids in full and enables marker quantification on a cell-by-cell basis. Sample preparation has been optimized for 3D imaging by confocal, super-resolution confocal, multiphoton and light-sheet microscopy. From organoid harvest to image analysis, the protocol takes 3 d.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article (and its supplementary information files).
References
Sato, T. & Clevers, H. SnapShot: growing organoids from stem cells. Cell 161, 1700–1700.e1 (2015).
Clevers, H. Modeling development and disease with organoids. Cell 165, 1586–1597 (2016).
Drost, J. & Clevers, H. Organoids in cancer research. Nat. Rev. Cancer 18, 407–418 (2018).
Bartfeld, S. & Clevers, H. Stem cell-derived organoids and their application for medical research and patient treatment. J. Mol. Med. 95, 729–738 (2017).
Broutier, L. et al. Human primary liver cancer-derived organoid cultures for disease modeling and drug screening. Nat. Med. 23, 1424–1435 (2017).
Dekkers, J. F. et al. Characterizing responses to CFTR-modulating drugs using rectal organoids derived from subjects with cystic fibrosis. Sci. Transl. Med. 8, 344ra384 (2016).
Dekkers, J. F. et al. A functional CFTR assay using primary cystic fibrosis intestinal organoids. Nat. Med. 19, 939–945 (2013).
Sato, T. et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 459, 262–265 (2009).
Karthaus, W. R. et al. Identification of multipotent luminal progenitor cells in human prostate organoid cultures. Cell 159, 163–175 (2014).
Lancaster, M. A. et al. Cerebral organoids model human brain development and microcephaly. Nature 501, 373–379 (2013).
Hu, H. et al. Long-term expansion of functional mouse and human hepatocytes as 3D organoids. Cell 175, 1591–1606.e19 (2018).
Huch, M. et al. Long-term culture of genome-stable bipotent stem cells from adult human liver. Cell 160, 299–312 (2015).
Nanki, K. et al. Divergent routes toward Wnt and R-spondin niche independency during human gastric carcinogenesis. Cell 174, 856–869.e17 (2018).
Jamieson, P. R. et al. Derivation of a robust mouse mammary organoid system for studying tissue dynamics. Development 144, 1065–1071 (2017).
Sachs, N. et al. A living biobank of breast cancer organoids captures disease heterogeneity. Cell 172, 373–386.e10 (2018).
Turco, M. Y. et al. Long-term, hormone-responsive organoid cultures of human endometrium in a chemically defined medium. Nat. Cell Biol. 19, 568–577 (2017).
Maimets, M. et al. Long-term in vitro expansion of salivary gland stem cells driven by Wnt signals. Stem Cell Rep. 6, 150–162 (2016).
Ren, W. et al. Single Lgr5- or Lgr6-expressing taste stem/progenitor cells generate taste bud cells ex vivo. Proc. Natl. Acad. Sci. USA 111, 16401–16406 (2014).
Richardson, D. S. & Lichtman, J. W. Clarifying tissue clearing. Cell 162, 246–257 (2015).
Richardson, D. S. & Lichtman, J. W. SnapShot: tissue clearing. Cell 171, 496–496.e1 (2017).
Rios, A. C. et al. Essential role for a novel population of binucleated mammary epithelial cells in lactation. Nat. Commun. 7, 11400 (2016).
Rios, A. C., Fu, N. Y., Lindeman, G. J. & Visvader, J. E. In situ identification of bipotent stem cells in the mammary gland. Nature 506, 322–327 (2014).
Rios, A. C. & Clevers, H. Imaging organoids: a bright future ahead. Nat. Methods 15, 24–26 (2018).
Sachs, N. et al. Long‐term expanding human airway organoids for disease modeling. EMBO J. 38, e100300 (2019).
Shutgens, F. et al. Tubuloids derived from human adult kidney and urine for personalized disease modelling. Nat. Biotechnol. 37, 303–313 (2019).
Eisenstein, M. Transparent tissues bring cells into focus for microscopy. Nature 564, 147–149 (2018).
Erturk, A. et al. Three-dimensional imaging of solvent-cleared organs using 3DISCO. Nat. Protoc. 7, 1983–1995 (2012).
Renier, N. et al. iDISCO: a simple, rapid method to immunolabel large tissue samples for volume imaging. Cell 159, 896–910 (2014).
Kubota, S. I. et al. Whole-body profiling of cancer metastasis with single-cell resolution. Cell Rep. 20, 236–250 (2017).
Murakami, T. C. et al. A three-dimensional single-cell-resolution whole-brain atlas using CUBIC-X expansion microscopy and tissue clearing. Nat. Neurosci. 21, 625–637 (2018).
Susaki, E. A. et al. Whole-brain imaging with single-cell resolution using chemical cocktails and computational analysis. Cell 157, 726–739 (2014).
Chung, K. et al. Structural and molecular interrogation of intact biological systems. Nature 497, 332–337 (2013).
Tomer, R., Ye, L., Hsueh, B. & Deisseroth, K. Advanced CLARITY for rapid and high-resolution imaging of intact tissues. Nat. Protoc. 9, 1682–1697 (2014).
Snippert, H. J. et al. Intestinal crypt homeostasis results from neutral competition between symmetrically dividing Lgr5 stem cells. Cell 143, 134–144 (2010).
Stelzer, E. H. Light-sheet fluorescence microscopy for quantitative biology. Nat. Methods 12, 23–26 (2015).
Huisken, J., Swoger, J., Del Bene, F., Wittbrodt, J. & Stelzer, E. H. Optical sectioning deep inside live embryos by selective plane illumination microscopy. Science 305, 1007–1009 (2004).
Acknowledgements
We are very grateful for the technical support from the Princess Máxima Center for Pediatric Oncology and to the Hubrecht Institute and Zeiss for imaging support and collaborations. All the imaging was performed at the Princess Máxima imaging center. This work was financially supported by the Princess Máxima Center for Pediatric Oncology. J.F.D. was supported by a Marie Curie Global Fellowship and a VENI grant from the Netherlands Organisation for Scientific Research (NWO). J.E.V. was supported by the Australian National Health and Medical Research Council (NHMRC).
Author information
Authors and Affiliations
Contributions
J.F.D. designed the study, performed experiments and interpreted data; J.F.D., M.A., L.M.W., H.C.R.A. and P.R.J. performed experiments and analyzed data; J.F.D., P.R.J., A.M.V., G.D.A., H.H. and J.M.B. performed the organoid culturing. K.C.O. and H.J.G.S. provided fluorescent constructs; A.C.R. helped design the study and carried out data interpretation; J.F.D. and A.C.R. cowrote the manuscript. J.E.V., H.C. and E.J.W. helped with data interpretation, manuscript writing and corrections.
Corresponding author
Ethics declarations
Competing interests
H.C. is named as inventor on several patents related to organoid technology. J.F.D. is named as inventor on one patent related to the organoid technology.
Additional information
Journal peer review information: Nature Protocols thanks Xavier Gidrol, Melissa Skala and other anonymous reviewer(s) for their contribution to the peer review of this work.
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
Hu, H. et al. Cell 175, 1591–1606.e19 (2018): https://doi.org/10.1016/j.cell.2018.11.013
Sachs, N. et al. EMBO J. 38, e100300 (2019): https://doi.org/10.15252/embj.2018100300
Supplementary information
Supplementary Video 1
This video highlights the intricate 3D features of delicate organoid structures that can be imaged at cellular or even subcellular resolution with this sample-preparation protocol.
Rights and permissions
About this article
Cite this article
Dekkers, J.F., Alieva, M., Wellens, L.M. et al. High-resolution 3D imaging of fixed and cleared organoids. Nat Protoc 14, 1756–1771 (2019). https://doi.org/10.1038/s41596-019-0160-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-019-0160-8
This article is cited by
-
Visualisation of gene expression within the context of tissues using an X-ray computed tomography-based multimodal approach
Scientific Reports (2024)
-
SOX2 and OCT4 mediate radiation and drug resistance in pancreatic tumor organoids
Cell Death Discovery (2024)
-
Vascularized organoid-on-a-chip: design, imaging, and analysis
Angiogenesis (2024)
-
BEHAV3D: a 3D live imaging platform for comprehensive analysis of engineered T cell behavior and tumor response
Nature Protocols (2024)
-
BECLIN1 is essential for intestinal homeostasis involving autophagy-independent mechanisms through its function in endocytic trafficking
Communications Biology (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.