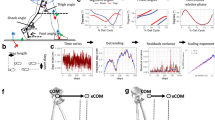
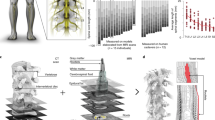
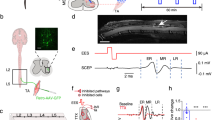
Abstract
Epidural electrical stimulation (EES) of the spinal cord and real-time processing of gait kinematics are powerful methods for the study of locomotion and the improvement of motor control after injury or in neurological disorders. Here, we describe equipment and surgical procedures that can be used to acquire chronic electromyographic (EMG) recordings from leg muscles and to implant targeted spinal cord stimulation systems that remain stable up to several months after implantation in rats and nonhuman primates. We also detail how to exploit these implants to configure electrical spinal cord stimulation policies that allow control over the degree of extension and flexion of each leg during locomotion. This protocol uses real-time processing of gait kinematics and locomotor performance, and can be configured within a few days. Once configured, stimulation bursts are delivered over specific spinal cord locations with precise timing that reproduces the natural spatiotemporal activation of motoneurons during locomotion. These protocols can also be easily adapted for the safe implantation of systems in the vicinity of the spinal cord and to conduct experiments involving real-time movement feedback and closed-loop controllers.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Bizzi, E., Giszter, S. F., Loeb, E., Mussa-Ivaldi, F. A. & Saltiel, P. Modular organization of motor behavior in the frog’s spinal cord. Trends Neurosci. 18, 442–446 (1995).
Levine, A. J. et al. Identification of a cellular node for motor control pathways. Nat. Neurosci. 17, 586–593 (2014).
van den Brand, R. et al. Restoring voluntary control of locomotion after paralyzing spinal cord injury. Science 336, 1182–1185 (2012).
Angeli, C. A., Edgerton, V. R., Gerasimenko, Y. P. & Harkema, S. J. Altering spinal cord excitability enables voluntary movements after chronic complete paralysis in humans. Brain 137, 1394–1409 (2014).
Harkema, S. et al. Effect of epidural stimulation of the lumbosacral spinal cord on voluntary movement, standing, and assisted stepping after motor complete paraplegia: a case study. Lancet 377, 1938–1947 (2011).
Danner, S. M. et al. Human spinal locomotor control is based on flexibly organized burst generators. Brain 138, 577–588 (2015).
Courtine, G. et al. Transformation of nonfunctional spinal circuits into functional states after the loss of brain input. Nat. Neurosci. 12, 1333–1342 (2009).
Capogrosso, M. et al. A brain–spine interface alleviating gait deficits after spinal cord injury in primates. Nature 539, 284–288 (2016).
Grahn, P. J. et al. Enabling task-specific volitional motor functions via spinal cord neuromodulation in a human with paraplegia. Mayo Clin. Proc. 92, 544–554 (2017).
Capogrosso, M. et al. A computational model for epidural electrical stimulation of spinal sensorimotor circuits. J. Neurosci. 33, 19326–19340 (2013).
Moraud, E. M. et al. Mechanisms underlying the neuromodulation of spinal circuits for correcting gait and balance deficits after spinal cord injury. Neuron 89, 814–828 (2016).
Rattay, F., Minassian, K. & Dimitrijevic, M. R. Epidural electrical stimulation of posterior structures of the human lumbosacral cord. 2. Quantitative analysis by computer modeling. Spinal Cord 38, 473–489 (2000).
Gerasimenko, Y. P. et al. Spinal cord reflexes induced by epidural spinal cord stimulation in normal awake rats. J. Neurosci. Methods 157, 253–263 (2006).
Minassian, K. et al. Human lumbar cord circuitries can be activated by extrinsic tonic input to generate locomotor-like activity. Hum. Mov. Sci. 26, 275–295 (2007).
Taccola, G., Sayenko, D., Gad, P., Gerasimenko, Y. & Edgerton, V. R. And yet it moves: recovery of volitional control after spinal cord injury. Prog. Neurobiol. 160, 64–81 (2018).
Stein, R. B. & Capaday, C. The modulation of human reflexes during functional motor tasks. Trends Neurosci. 11, 328–332 (1988).
Dy, C. J. et al. Phase-dependent modulation of percutaneously elicited multisegmental muscle responses after spinal cord injury. J. Neurophysiol. 103, 2808–2820 (2010).
Sayenko, D. G., Angeli, C., Harkema, S. J., Edgerton, V. R. & Gerasimenko, Y. P. Neuromodulation of evoked muscle potentials induced by epidural spinal-cord stimulation in paralyzed individuals. J. Neurophysiol. 111, 1088–1099 (2014).
Wenger, N. et al. Closed-loop neuromodulation of spinal sensorimotor circuits controls refined locomotion after complete spinal cord injury. Sci. Transl. Med. 6, 255ra133 (2014).
Wenger, N. et al. Spatiotemporal neuromodulation therapies engaging muscle synergies improve motor control after spinal cord injury. Nat. Med. 22, 138–145 (2016).
Yakovenko, S., Mushahwar, V., VanderHorst, V., Holstege, G. & Prochazka, A. Spatiotemporal activation of lumbosacral motoneurons in the locomotor step cycle. J. Neurophysiol. 87, 1542–1553 (2002).
Ivanenko, Y. P. et al. Temporal components of the motor patterns expressed by the human spinal cord reflect foot kinematics. J. Neurophysiol. 90, 3555–3565 (2003).
Wenger, N. et al. Closed-loop neuromodulation of spinal sensorimotor circuits controls refined locomotion after complete spinal cord injury. Sci. Transl. Med. 6, 255ra133 (2014).
Courtine, G. et al. Transformation of nonfunctional spinal circuits into functional states after the loss of brain input. Nat. Neurosci. 12, 1333–1342 (2009).
Minev, I. R. et al. Electronic dura mater for long-term multimodal neural interfaces. Science 347, 159–163 (2015).
Capogrosso, M. et al. Advantages of soft subdural implants for the delivery of electrochemical neuromodulation therapies to the spinal cord. J. Neural Eng. 15, 026024 (2018).
Cheriyan, T. et al. Spinal cord injury models: a review. Spinal Cord 52, 588–595 (2014).
Lukovic, D. et al. Complete rat spinal cord transection as a faithful model of spinal cord injury for translational cell transplantation. Sci. Rep. 5, 9640 (2015).
Courtine, G. et al. Recovery of supraspinal control of stepping via indirect propriospinal relay connections after spinal cord injury. Nat. Med. 14, 69–74 (2008).
Courtine, G. et al. Can experiments in nonhuman primates expedite the translation of treatments for spinal cord injury in humans? Nat. Med. 13, 561–566 (2007).
Capogrosso, M. et al. A brain–spine interface alleviating gait deficits after spinal cord injury in primates. Nature 539, 284–288 (2016).
Freund, P. et al. Nogo-A-specific antibody treatment enhances sprouting and functional recovery after cervical lesion in adult primates. Nat. Med. 12, 790–792 (2006).
Slotkin, J. R. et al. Biodegradable scaffolds promote tissue remodeling and functional improvement in non-human primates with acute spinal cord injury. Biomaterials 123, 63–76 (2017).
Rosenzweig, E. S. et al. Extensive spontaneous plasticity of corticospinal projections after primate spinal cord injury. Nat. Neurosci. 13, 1505–1510 (2010).
Salegio, E. A. et al. A unilateral cervical spinal cord contusion injury model in non-human primates (Macaca mulatta). J. Neurotrauma 33, 439–459 (2016).
Reeve, C. & Reeve, D. The Spinal Cord (Elsevier, New York, 2008).
Coburn, B. A theoretical study of epidural electrical stimulation of the spinal cord—Part II: effects on long myelinated fibers. IEEE Trans. Biomed. Eng. 32, 978–986 (1985).
Holsheimer, J. Which neuronal elements are activated directly by spinal cord stimulation. Neuromodulation 5, 25–31 (2002).
Cuellar, C. A. et al. The role of functional neuroanatomy of the lumbar spinal cord in effect of epidural stimulation. Front. Neuroanat. 11, 82 (2017).
Gad, P. et al. Development of a multi-electrode array for spinal cord epidural stimulation to facilitate stepping and standing after a complete spinal cord injury in adult rats. J. Neuroeng. Rehabil. 10, 2 (2013).
Merrill, D. R., Bikson, M. & Jefferys, J. G. Electrical stimulation of excitable tissue: design of efficacious and safe protocols. J. Neurosci. Methods 141, 171–198 (2005).
Cogan, S. F. Neural stimulation and recording electrodes. Annu. Rev. Biomed. Eng. 10, 275–309 (2008).
de Jongste, M. J. et al. Stimulation characteristics, complications, and efficacy of spinal cord stimulation systems in patients with refractory angina: a prospective feasibility study. Pacing Clin. Electrophysiol. 17, 1751–1760 (1994).
Alo, K. et al. Factors affecting impedance of percutaneous leads in spinal cord stimulation. Neuromodulation 9, 128–135 (2006).
Hofstoetter, U. S. et al. Periodic modulation of repetitively elicited monosynaptic reflexes of the human lumbosacral spinal cord. J. Neurophysiol. 114, 400–410 (2015).
Segev, I., Fleshman, J. W. Jr. & Burke, R. E. Computer simulation of group Ia EPSPs using morphologically realistic models of cat α-motoneurons. J. Neurophysiol. 64, 648–660 (1990).
Gerasimenko, Y. P. et al. Spinal cord reflexes induced by epidural spinal cord stimulation in normalawakerats. J. Neurosci. Methods. 157, 253–263 (2006).
Edgerton, V. R. et al. Training locomotor networks. Brain Res. Rev. 57, 241–254 (2008).
Holinski, B. J. et al. Intraspinal microstimulation produces over-ground walking in anesthetized cats. J. Neural Eng. 13, 056016 (2016).
Hehenberger, L., Seeber, M. & Scherer, R. Estimation of gait parameters from EEG source oscillations. IEEE International Conference on Systems, Man, and Cybernetics (SMC) 004182-004187, https://doi.org/10.1109/SMC.2016.7844888 (2016).
Presacco, A., Goodman, R., Forrester, L. & Contreras-Vidal, J. L. Neural decoding of treadmill walking from noninvasive electroencephalographic signals. J. Neurophysiol. 106, 1875–1887 (2011).
Presacco, A., Forrester, L. W. & Contreras-Vidal, J. L. Decoding intra-limb and inter-limb kinematics during treadmill walking from scalp electroencephalographic (EEG) signals. IEEE Trans. Neural Syst. Rehabil. Eng. 20, 212–219 (2012).
Artoni, F. et al. Unidirectional brain to muscle connectivity reveals motor cortex control of leg muscles during stereotyped walking. Neuroimage 159, 403–416 (2017).
Rouse, A. G., Williams, J. J., Wheeler, J. J. & Moran, D. W. Cortical adaptation to a chronic micro-electrocorticographic brain computer interface. J. Neurosci. 33, 1326–1330 (2013).
McCrimmon, C. M. et al. Electrocorticographic encoding of human gait in the leg primary motor cortex. Cereb. Cortex 28, 2752–2762 (2018).
Fitzsimmons, N. A., Lebedev, M. A., Peikon, I. D. & Nicolelis, M. A. Extracting kinematic parameters for monkey bipedal walking from cortical neuronal ensemble activity. Front. Integr. Neurosci. 3, 3 (2009).
Ma, X. et al. Decoding lower limb muscle activity and kinematics from cortical neural spike trains during monkey performing stand and squat movements. Front. Neurosci. 11, 44 (2017).
Waldert, S. et al. A review on directional information in neural signals for brain–machine interfaces. J. Physiol. Paris 103, 244–254 (2009).
Yin, M. et al. Wireless neurosensor for full-spectrum electrophysiology recordings during free behavior. Neuron 84, 1170–1182 (2014).
Borton, D. A., Yin, M., Aceros, J. & Nurmikko, A. An implantable wireless neural interface for recording cortical circuit dynamics in moving primates. J. Neural Eng. 10, 026010 (2013).
Foster, J. D. et al. A freely-moving monkey treadmill model. J. Neural Eng. 11, 046020 (2014).
Schwarz, D. A. et al. Chronic, wireless recordings of large-scale brain activity in freely moving rhesus monkeys. Nat. Methods 11, 670–676 (2014).
Suner, S., Fellows, M. R., Vargas-Irwin, C., Nakata, G. K. & Donoghue, J. P. Reliability of signals from a chronically implanted, silicon-based electrode array in non-human primate primary motor cortex. IEEE Trans. Neural Syst. Rehabil. Eng. 13, 524–541 (2005).
Barrese, J. C. et al. Failure mode analysis of silicon-based intracortical microelectrode arrays in non-human primates. J. Neural Eng. 10, 066014 (2013).
Simeral, J. D., Kim, S. P., Black, M. J., Donoghue, J. P. & Hochberg, L. R. Neural control of cursor trajectory and click by a human with tetraplegia 1000 days after implant of an intracortical microelectrode array. J. Neural Eng. 8, 025027 (2011).
Chestek, C. A. et al. Long-term stability of neural prosthetic control signals from silicon cortical arrays in rhesus macaque motor cortex. J. Neural Eng. 8, 045005 (2011).
Milekovic, T., Ball, T., Schulze-Bonhage, A., Aertsen, A. & Mehring, C. Detection of error related neuronal responses recorded by electrocorticography in humans during continuous movements. PLoS ONE 8, e55235 (2013).
Flouty, O. E. et al. Intracranial somatosensory responses with direct spinal cord stimulation in anesthetized sheep. PLoS ONE 8, e56266 (2013).
Musienko, P., van den Brand, R., Maerzendorfer, O., Larmagnac, A. & Courtine, G. Combinatory electrical and pharmacological neuroprosthetic interfaces to regain motor function after spinal cord injury. IEEE Trans. Biomed. Eng. 56, 2707–2711 (2009).
Moritz, C. T., Lucas, T. H., Perlmutter, S. I. & Fetz, E. E. Forelimb movements and muscle responses evoked by microstimulation of cervical spinal cord in sedated monkeys. J. Neurophysiol. 97, 110–120 (2007).
Sunshine, M. D. et al. Cervical intraspinal microstimulation evokes robust forelimb movements before and after injury. J. Neural Eng. 10, 036001 (2013).
Zimmermann, J. B., Seki, K. & Jackson, A. Reanimating the arm and hand with intraspinal microstimulation. J. Neural Eng. 8, 054001 (2011).
Calixto, R. & Mushahwar, V. Understanding the mechanisms and sites of action of intraspinal microstimulation. in P roceedings of the 12th Annual Conference of the International Functional Electrical Stimulation Society.
Rattay, F. Analysis of models for external stimulation of axons. IEEE Trans. Biomed. Eng. 33, 974–977 (1986).
McIntyre, C. C. & Grill, W. M. Extracellular stimulation of central neurons: influence of stimulus waveform and frequency on neuronal output. J. Neurophysiol. 88, 1592–1604 (2002).
Gaunt, R. A., Prochazka, A., Mushahwar, V. K., Guevremont, L. & Ellaway, P. H. Intraspinal microstimulation excites multisegmental sensory afferents at lower stimulus levels than local α- motoneuron responses. J. Neurophysiol. 96, 2995–3005 (2006).
Iles, J. F. Central terminations of muscle afferents on motoneurones in the cat spinal cord. J. Physiol. 262, 91–117 (1976).
Brown, A. G. & Fyffe, R. E. The morphology of group Ia afferent fibre collaterals in the spinal cord of the cat. J. Physiol. 274, 111–127 (1978).
Burke, R. E. & Glenn, L. L. Horseradish peroxidase study of the spatial and electrotonic distribution of group Ia synapses on type-identified ankle extensor motoneurons in the cat. J. Comp. Neurol. 372, 465–485 (1996).
Minassian, K., Hofstoetter, U., Tansey, K. & Mayr, W. Neuromodulation of lower limb motor control in restorative neurology. Clin. Neurol. Neurosurg. 114, 489–497 (2012).
Ethier, C., Oby, E. R., Bauman, M. J. & Miller, L. E. Restoration of grasp following paralysis through brain-controlled stimulation of muscles. Nature 485, 368–371 (2012).
Bouton, C. E. et al. Restoring cortical control of functional movement in a human with quadripleaturgia. Nature 533, 247–250 (2016).
Ajiboye, A. B. et al. Restoration of reaching and grasping movements through brain-controlled muscle stimulation in a person with tetraplegia: a proof-of-concept demonstration. Lancet 389, 1821–1830 (2017).
Giat, Y., Mizrahi, J. & Levy, M. A musculotendon model of the fatigue profiles of paralyzed quadriceps muscle under FES. IEEE Trans. Biomed. Eng. 40, 664–674 (1993).
Popovic, M. R., Popovic, D. B. & Keller, T. Neuroprostheses for grasping. Neurol. Res. 24, 443–452 (2002).
House, P. A., MacDonald, J. D., Tresco, P. A. & Normann, R. A. Acute microelectrode array implantation into human neocortex: preliminary technique and histological considerations. Neurosurg. Focus 20, 1–4 (2006).
Gerasimenko, Y., Roy, R. R. & Edgerton, V. R. Epidural stimulation: comparison of the spinal circuits that generate and control locomotion in rats, cats and humans. Exp. Neurol. 209, 417–425 (2008).
Acknowledgements
The illustration in Fig. 1 was created by C. Beach. N.W. is a participant in the Charité Clinical Scientist Program funded by the Charité–Universitätsmedizin Berlin and the Berlin Institute of Health. This work was supported by Medtronic, the European Community’s Seventh Framework Programme (CP-IP 258654, NeuWALK), the International Paraplegic foundation (IRP), a Consolidator Grant from the European Research Council (ERC-2015-CoG HOW2WALKAGAIN 682999), the Wyss Center in Geneva, the Russian Science Foundation (RSF grant 14-15-00788, P.M.), a Wings for Life Fellowship to G.C., Marie Curie COFUND EPFL fellowships to F.B.W. and T.M., and a Morton Cure Paralysis Fund fellowship to T.M., as well as by the Swiss National Science Foundation, including a Bonus of Excellence (310030B_166674), the National Center of Competence in Research (NCCR) Robotics, the Sino-Swiss Science and Technology Cooperation (IZLCZ3_156331), the NanoTera.ch program (SpineRepair) and the Sinergia program (CRSII3_160696).
Author information
Authors and Affiliations
Contributions
M.C., F.B.W., J.G., E.M.M., N.W., T.M. and G.C. developed the methods to control the stimulation and optimize the electrode placement. P.S., N.P., P.M., J.B. and G.C. developed the surgical procedures. M.C., F.B.W., J.G., E.M.M. and T.M. performed the experiments in monkeys. N.W., E.M.M., J.G. and M.C. performed the experiments in rats. E.B. and G.C. supervised the experiments and animal procedures. M.C., F.B.W., J.G., E.M.M., N.W. and T.M. analyzed the data. M.C., F.B.W. and J.G. created the figures. M.C., F.B.W. and G.C. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
G.C., M.C., E.M.M., N.W., T.M., F.B.W., J.G. and J.B. hold various patents related to the present work. E.B. reports receipt of personal fees from Motac Neuroscience Ltd. UK and is a shareholder of Motac Holding, UK, and Plenitudes SARL, France. G.C. and J.B. are founders and shareholders of GTX Medical BV. The other authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
1. Wenger, N. et al. Nat. Med. 22, 138–145 (2016) https://doi.org/10.1038/nm.4025
2. Capogrosso, M. et al. Nature 539, 284–288 (2016) https://doi.org/10.1038/nature20118
Rights and permissions
About this article
Cite this article
Capogrosso, M., Wagner, F.B., Gandar, J. et al. Configuration of electrical spinal cord stimulation through real-time processing of gait kinematics . Nat Protoc 13, 2031–2061 (2018). https://doi.org/10.1038/s41596-018-0030-9
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-018-0030-9
This article is cited by
-
Fully implanted battery-free high power platform for chronic spinal and muscular functional electrical stimulation
Nature Communications (2023)
-
Epidural stimulation of the cervical spinal cord for post-stroke upper-limb paresis
Nature Medicine (2023)
-
Neurotechnologies to restore hand functions
Nature Reviews Bioengineering (2023)
-
Longitudinal interrogation of sympathetic neural circuits and hemodynamics in preclinical models
Nature Protocols (2023)
-
Neuroprosthetics: from sensorimotor to cognitive disorders
Communications Biology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.