Abstract
In Alzheimer’s disease, amyloid deposits along the brain vasculature lead to a condition known as cerebral amyloid angiopathy (CAA), which impairs blood–brain barrier (BBB) function and accelerates cognitive degeneration. Apolipoprotein (APOE4) is the strongest risk factor for CAA, yet the mechanisms underlying this genetic susceptibility are unknown. Here we developed an induced pluripotent stem cell-based three-dimensional model that recapitulates anatomical and physiological properties of the human BBB in vitro. Similarly to CAA, our in vitro BBB displayed significantly more amyloid accumulation in APOE4 compared to APOE3. Combinatorial experiments revealed that dysregulation of calcineurin–nuclear factor of activated T cells (NFAT) signaling and APOE in pericyte-like mural cells induces APOE4-associated CAA pathology. In the human brain, APOE and NFAT are selectively dysregulated in pericytes of APOE4 carriers, and inhibition of calcineurin–NFAT signaling reduces APOE4-associated CAA pathology in vitro and in vivo. Our study reveals the role of pericytes in APOE4-mediated CAA and highlights calcineurin–NFAT signaling as a therapeutic target in CAA and Alzheimer’s disease.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All requests for raw and analyzed data and materials are promptly reviewed by the Massachusetts Institute of Technology, Technology Licensing Office to verify whether the request is subject to any intellectual property or confidentiality obligations. Patient-related data not included in the paper were generated as part of clinical trials and may be subject to patient confidentiality. Any data and materials that can be shared will be released via a Material Transfer Agreement. All raw and analyzed sequencing data can be found at the NCBI Sequence Read Archive (accession number: GSE125869).
Change history
29 January 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41591-021-01250-8
References
Andreone, B. J., Lacoste, B. & Gu, C. Neuronal and vascular interactions. Annu. Rev. Neurosci. 38, 25–46 (2015).
Zlokovic, B. V. Neurovascular mechanisms of Alzheimer’s neurodegeneration. Trends Neurosci. 28, 202–208 (2005).
Iadecola, C. The neurovascular unit coming of age: a journey through neurovascular coupling in health and disease. Neuron 96, 17–42 (2017).
Vemuri, P. et al. Vascular and amyloid pathologies are independent predictors of cognitive decline in normal elderly. Brain 138, 761–771 (2015).
Ransohoff, R. M. How neuroinflammation contributes to neurodegeneration. Science 353, 777–783 (2016).
Nation, D. A. et al. Blood–brain barrier breakdown is an early biomarker of human cognitive dysfunction. Nat. Med. 25, 270–276 (2019).
Ringman, J. M. et al. Clinical predictors of severe cerebral amyloid angiopathy and influence of APOE Genotype in persons with pathologically verified Alzheimer disease. JAMA Neurol. 71, 878–883 (2014).
Scheltens, P. & Goos, J. D. Dementia in 2011: microbleeds in dementia—singing a different ARIA. Nat. Rev. Neurol. 8, 68–70 (2012).
Iadecola, C. The pathobiology of vascular dementia. Neuron 80, 844–866 (2013).
Greenberg, S. M. et al. ϵ4 and cerebral hemorrhage associated with amyloid angiopathy. Ann. Neurol. 38, 254–259 (1995).
Premkumar, D. R., Cohen, D. L., Hedera, P., Friedland, R. P. & Kalaria, R. N. Apolipoprotein E-epsilon4 alleles in cerebral amyloid angiopathy and cerebrovascular pathology associated with Alzheimer’s disease. Am. J. Pathol. 148, 2083 (1996).
Shinohara, M. et al. Impact of sex and APOE4 on cerebral amyloid angiopathy in Alzheimer’s disease. Acta Neuropathol. 132, 225–234 (2016).
Janzer, R. C. & Raff, M. C. Astrocytes induce blood–brain barrier properties in endothelial cells. Nature 325, 253–257 (1987).
Armulik, A. et al. Pericytes regulate the blood–brain barrier. Nature 468, 557–561 (2010).
Daneman, R., Zhou, L., Kebede, A. A. & Barres, B. A. Pericytes are required for blood–brain barrier integrity during embryogenesis. Nature 468, 562–566 (2010).
Chow, B. W. & Gu, C. The molecular constituents of the blood–brain barrier. Trends Neurosci. 38, 598–608 (2015).
Qian, T. et al. Directed differentiation of human pluripotent stem cells to blood–brain barrier endothelial cells. Sci. Adv. 3, e1701679 (2017).
Patsch, C. et al. Generation of vascular endothelial and smooth muscle cells from human pluripotent stem cells. Nat. Cell Biol. 17, 994–1003 (2015).
TCW, J. et al. An efficient platform for astrocyte differentiation from human induced pluripotent stem cells. Stem Cell Rep. 9, 600–614 (2017).
Kumar, A. et al. Specification and diversification of pericytes and smooth muscle cells from mesenchymoangioblasts. Cell Rep. 19, 1902–1916 (2017).
Armulik, A., Genové, G. & Betsholtz, C. Pericytes: developmental, physiological, and pathological perspectives, problems, and promises. Dev. Cell 21, 193–215 (2011).
Attwell, D., Mishra, A., Hall, C. N., O’Farrell, F. M. & Dalkara, T. What is a pericyte?. J. Cereb. Blood Flow Metab. 36, 451–455 (2016).
Vanlandewijck, M. et al. A molecular atlas of cell types and zonation in the brain vasculature. Nature 554, 475–480 (2018).
He, L. et al. Single-cell RNA sequencing of mouse brain and lung vascular and vessel-associated cell types. Sci. Data 5, 180160 (2018).
Bonomini, F., Francesca, B. & Rezzani, R. Aquaporin and blood–brain barrier. Curr. Neuropharmacol. 8, 92–96 (2010).
Gautam, J., Zhang, X. & Yao, Y. The role of pericytic laminin in blood–brain barrier integrity maintenance. Sci. Rep. 6, 36450 (2016).
O’Brown, N. M., Pfau, S. J. & Gu, C. Bridging barriers: a comparative look at the blood–brain barrier across organisms. Genes Dev. 32, 466–478 (2018).
Urich, E., Lazic, S. E., Molnos, J., Wells, I. & Freskgård, P.-O. Transcriptional profiling of human brain endothelial cells reveals key properties crucial for predictive in vitro blood–brain barrier models. PLoS ONE 7, e38149 (2012).
Zhao, Z., Nelson, A. R., Betsholtz, C. & Zlokovic, B. V. Establishment and dysfunction of the blood–brain barrier. Cell 163, 1064–1078 (2015).
Strickland, L. A. et al. Plasmalemmal vesicle-associated protein (PLVAP) is expressed by tumour endothelium and is upregulated by vascular endothelial growth factor-A (VEGF). J. Pathol. 206, 466–475 (2005).
Srinivasan, B. et al. TEER measurement techniques for in vitro barrier model systems. J. Lab. Autom. 20, 107–126 (2015).
Deli, M. A., Ábrahám, C. S., Kataoka, Y. & Niwa, M. Permeability studies on in vitro blood–brain barrier models: physiology, pathology, and pharmacology. Cell Mol. Neurobiol. 25, 59–127 (2005).
Seetharaman, S., Barrand, M. A., Maskell, L. & Scheper, R. J. Multidrug resistance-related transport proteins in isolated human brain microvessels and in cells cultured from these isolates. J. Neurochem. 70, 1151–1159 (1998).
Zhang, Y. et al. An RNA-sequencing transcriptome and splicing database of glia, neurons, and vascular cells of the cerebral cortex. J. Neurosci. 34, 11929–11947 (2014).
Israel, M. A. et al. Probing sporadic and familial Alzheimer’s disease using induced pluripotent stem cells. Nature 482, 216–220 (2012).
Chambers, S. M. et al. Combined small-molecule inhibition accelerates developmental timing and converts human pluripotent stem cells into nociceptors. Nat. Biotechnol. 30, 715–720 (2012).
Shi, Y., Kirwan, P. & Livesey, F. J. Directed differentiation of human pluripotent stem cells to cerebral cortex neurons and neural networks. Nat. Protoc. 7, 1836–1846 (2012).
Lin, Y. T. et al. APOE4 causes widespread molecular and cellular alterations associated with Alzheimer’s disease phenotypes in human iPSC-derived brain cell types. Neuron 98, 1141–1154 (2018).
Xu, Q. et al. Profile and regulation of apolipoprotein E (ApoE) expression in the CNS in mice with targeting of green fluorescent protein gene to the ApoE locus. J. Neurosci. 26, 4985–4994 (2006).
Mathys, H. et al. Single-cell transcriptomic analysis of Alzheimer’s disease. Nature 1, 1 (2019).
Fryer, J. D. et al. Apolipoprotein E markedly facilitates age-dependent cerebral amyloid angiopathy and spontaneous hemorrhage in amyloid precursor protein transgenic mice. J. Neurosci. 23, 7889–7896 (2003).
Kim, J. et al. Haploinsufficiency of human APOE reduces amyloid deposition in a mouse model of amyloid-β amyloidosis. J. Neurosci. 31, 18007–18012 (2011).
Maloney, B., Ge, Y.-W., Alley, G. M. & Lahiri, D. K. Important differences between human and mouse APOE gene promoters: limitation of mouse APOE model in studying Alzheimer’s disease. J. Neurochem. 103, 1237–1257 (2007).
Reese, L. C. & Taglialatela, G. A role for calcineurin in Alzheimer’s disease. Curr. Neuropharmacol. 9, 685–692 (2011).
Gwack, Y. et al. A genome-wide Drosophila RNAi screen identifies DYRK-family kinases as regulators of NFAT. Nature 441, 646–650 (2006).
Mulero, M. C., Aubareda, A., Schlüter, A. & Pérez-Riba, M. RCAN3, a novel calcineurin inhibitor that down-regulates NFAT-dependent cytokine gene expression. Biochim. Biophys. Acta 330–341, 2007 (1773).
Lee, M. Y., Garvey, S. M., Ripley, M. L. & Wamhoff, B. R. Genome-wide microarray analyses identify the protein C receptor as a novel calcineurin/nuclear factor of activated T cells-dependent gene in vascular smooth muscle cell phenotypic modulation. Arterioscler. Thromb. Vasc. Biol. 31, 2665–2675 (2011).
Orr, A. W., Hastings, N. E., Blackman, B. R. & Wamhoff, B. R. Complex regulation and function of the inflammatory smooth muscle cell phenotype in atherosclerosis. J. Vasc. Res. 47, 168–180 (2010).
Kapturczak, M. H., Meier-Kriesche, H. U. & Kaplan, B. Pharmacology of calcineurin antagonists. Transplant. Proc. 36, 25S–32S (2004).
Boroujerdi, A., Tigges, U., Welser-Alves, J. V. & Milner, R. in Cerebral Angiogenesis 383–392 (Humana Press, 2014).
Stebbins, M. J. et al. Human pluripotent stem cell-derived brain pericyte-like cells induce blood–brain barrier properties. Sci. Adv. 5, eaau7375 (2019).
Taglialatela, G., Rastellini, C. & Cicalese, L. Reduced incidence of dementia in solid organ transplant patients treated with calcineurin inhibitors. J. Alzheimers Dis. 47, 329–333 (2015).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Liao, Y., Smyth, G. K., Bioinformatics, W. S. featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics. 7, 923–930 (2014).
Trapnell, C. et al. Differential gene and transcript expression analysis of RNA-seq experiments with TopHat and Cufflinks. Nat. Protoc. 7, 562–578 (2012).
Bennett, D. A. et al. Religious orders study and rush memory and aging project. J. Alzheimer’s Dis. 64, S161–S189 (2018).
Wang, J. et al. Gene expression distribution deconvolution in single-cell RNA sequencing. Proc. Natl Acad. Sci. USA 115, E6437–E6446 (2018).
Boroujerdi, A. et al. Isolation and Culture of Primary Pericytes from Mouse Brain. Cerebral Angiogenesis. 383–392 (2014).
Acknowledgements
We thank J. Penny, W.C. Huang, Y.M. Yang and P. Narayan for critically reading the manuscript, J.Z. Young, J. Ray, V. Buggia-Prevot, C. Fernandez, the Neurodegenerative Consortium and all Tsai laboratory members for helpful discussions, A. Marco for expertise and support with microscopy, J. Seo for expertise and guidance with western blotting and Y. Zhou, E. McNamara, P. Curtis and T. Garvey for administrative support. This work was supported by The Neurodegeneration Consortium, the Belfer Family Foundation and the Oskar Fisher Project to L.-H.T., Cure Alzheimer’s Fund to L.-H.T. and MK, RF1 AG062377 to L-HT and MK, RF1 AG048056. The National Institutes of Health Common Fund’s Library of Integrated Network-based Cellular Signatures program by U54HG008097 to L.-H.T. and J. Jaffe (Broad Institute), P30AG10161, R01AG15819, R01AG17917, U0161356 to D.A.B., UG3NS115064 to L.-H.T. and J.W.B. M.B.V. is supported by the HHMI Hannah Gray Postdoctoral Fellowship. J.W.B. was supported in part by The Glenn Foundation for Medical Research and American Federation for Aging Research Postdoctoral Fellowship during this work.
Author information
Authors and Affiliations
Contributions
J.W.B. and L.-H.T. conceived the study. J.W.B., M.B., L.A.A., L.Z., A.F., M.B.V. and J.M.B. performed experiments and analyzed results. H.P.C. performed RNA-seq genomic alignments and DEG analysis. J.D.-V. and H.M. performed and analyzed the snRNA-seq experiments. Y.-T.L. generated isogenic and knockout APOE lines. T.K. generated the iPSC lines used in this study. J.W.B, L.-H.T. and H.P.C. wrote and revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
L.-H. Tsai and J. Blanchard filed a patent application on the iBBB technology (PCT/US2020/014572). The authors declare no other competing interests.
Additional information
Peer review information Kate Gao was the primary editor on this article, and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Characterization of human iPSC-derived cells.
a and b iPSC-derived brain endothelial cells stained with CD144 (VE-Cadherin), CD31 (PECAM), ZO-1 and GLUT1. These experiments were repeated at least 4 times with similar results. c and d, iPSC-derived astrocytes stained with GFAP, S100β and AQP4. e and f Comparative expression analysis of genes in iPSC-derived astrocytes from RNA-sequencing that are reported to be the most differentially upregulated in These experiments were repeated at least 4 times with similar results. e, fibroblasts and f, oligodendrocytes when compared to astrocytes from In vivo data in Vanlandewijck et al., 2018. Astrocytes are from 6 independent bulk RNA-sequencing g-i iPSC-derived mural cells stained with CD13, SM22, NG2, and SMA. These experiments were repeated at least 2 times with similar results. j. Comparative expression analysis of the top differentially upregulated genes in pericytes compared to smooth muscle cells (SMCs) from In vivo data in Vanlandewijck et al., 2018. iMCs are from 6 independent bulk RNA-sequencing. Expression is represented as FPKM values from bulk RNA-sequencing. Center values are mean and error bars are SD k, Comparative expression analysis of the top differentially upregulated genes in SMCs compared to pericytes from In vivo data in Vanlandewijck et al., 2018. iMCs are from 6 independent bulk RNA-sequencing. Expression is represented as FPKM values from bulk RNA-sequencing Center values are mean and error bars are SD l, Expression of the top three differentially upregulated genes in pericytes compared to fibroblasts. m, Expression of the top three differentially upregulated genes in fibroblasts compared to pericytes from In vivo data in Vanlandewijck et al., 2018. iMCs are from 6 independent bulk RNA-sequencing. Center values are mean and error bars are SD. n, Expression of mural cells and mesenchymal marker genes in iPSC-derived mural cells. For e, f, j, k, l, m, differential gene lists are based on analysis provided shown as average counts compared to FPKM from bulk RNA-sequencing of iPSC-derived astrocytes and mural cells. Center value and error bars are means and SD from RNA prepared from 6 independent wells. o, Identification of In vivo Brain Endothelial, Pericyte, and SMC cluster via marker gene expression from single-nucleus RNA-sequencing of the human post-mortem hippocampus from 82 individuals. p, iPSC-derived mural cells express markers of in vivo human brain pericytes. Center values and error bars are means and SD from independent RNA prepared from 3 separate wells. q, Global hierarchical clustering of transcriptomes (13,338 genes) demonstrates that iPSC-derived mural cells cluster with in vivo human hippocampal pericytes. Clustering was performed by average correlation with simple linkage.
Extended Data Fig. 2 Characterization of human iBBB.
a, Three-dimensional vascular network of endothelial cells stained with CD144 scale bar = 200 μm. b, one week after formation iMural cells labeled with SM22 are homogeneously dispersed and rudimentary vessels started forming. After two weeks endothelial vessels formed and iMural cells have homed to perivascular space. c, Astrocytes are dispersed throughout iBBB cultures. Experiments in a-c were repeated at least 3 times. d, mRNA expression of AQP4 in each cell type alone, pair-wise and combined. One-way ANOVA with Bonferroni’s multiple comparison. **, p = 0.0013, 0.002, 0.0035, and 0.0076 for BECs, iMCs, Astrocytes, and BECs + iMCs respectively. ****, p < 0.0001. Center values and error bars are means and SDs from 6 independent RNA isolations. e, iBBB without astrocytes do not stain for AQP4 (green). In iBBBs with astrocytes AQP4 densely stains along endothelial vessels. f, Immunostaining for LAMA4 showing that Matrigel does not contain LAMA4 however iBBB cultures remodel basement membrane surrounding endothelial vessels to contain LAMA4. Experiments in e-f were repeated at least 3 times. g, PLVAP mRNA expression is upregulated in BECs from iBBB cultures compared to BECs cultured alone. Center values and error bars are mean and SD from RNA harvested from six independent wells. h, PLVAP mRNA expression is downregulated in BECs from iBBB upon removal of VEGFA from culture media. Center values and error bars are mean and SD from RNA harvested from six independent wells. Two-sided unpaired student t-test p < 0.0001. i, iBBB cultured in trans-well format express high levels of BBB marker CLDN5 (green) and ZO1 (red). Experiments in e-f were repeated at least 2 times. j, Polarization of ABCG2 was measured by Hoechst transport for both a BECs monolayer and the iBBB from the apical to the basolateral surface and vice versa. Samples treated with the ABCG2 specific inhibitor KO143 were normalized to each respective non-inhibitor treated sample. Stars represent significance determined by two-sided multiple student’s t-test (FDR = 0.01; p = 0.0011) Center values and error bars are mean and SD from 6 independent transwells.
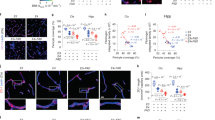
Extended Data Fig. 3 Validation of the iBBB as a model for CAA.
a, iBBBs generated from a familial AD patient iPSC with duplication of the APP gene (APP1.1) do not inherently have higher amyloid levels than non-AD controls (AG09173). b, iBBBs generated from iPSCs with a familial AD-associated mutation (M146I) in the PSEN1 gene do not inherently have higher amyloid levels than its non-AD isogenic control. Experiments in a and b were repeated at least 3 times. c, Media conditioned by neuronal cells derived from familial AD patient has significantly higher Aβ(1-42). Two-sided Student t-test (p = 0.0022) Center values and error bars are mean and SD from 3 independent wells. d, Representative images depicting that iBBBs derived from APOE3/4 individuals exhibit high levels of amyloid accumulation relative to iBBBs generated from APOE3/3 individuals. e and f, Representative images depicting that iBBBs derived from isogenic APOE3/3 and APOE4/4 individuals exhibit high levels of amyloid accumulation assay with anti-amyloid antibody Thioflavin T (f) and 12F4 (e). Experiments in d-f were repeated at least 3 times. g and h, Representative images and quantification of amyloid accumulation in isogenic iBBBs exposed to 20 nM Aβ-FITC for 1–40 and 1–42 isoforms. The total area positive for amyloid was divided by total nuclei and then normalized to the mean amyloid/nuclei from all E3/3 samples such that the mean of E3/E3 is set to 100% for each isoform. Students t-test, 1–40 p = 0.0044; 1–42 p > 0.00001. Experiments in were repeated at least 3 times. Center values and error bars are means and SD from 12 independent iBBBs. i and j, Normalized amyloid accumulation in isogenic iMural cells and BECs mono-culture for each APOE genotype. Two-sided Students t-test, iMural cells, p = 0.0002; BECs p = 0.0118. Center values and error bars are means and SD from 12 independent wells.
Extended Data Fig. 4 APOE4 Pericytes Increase CAA pathology in iBBB.
a, Quantification of Aβ accumulation in deconstructed iBBBs. B/iMC/A3 and B/iMC/A4 indicate all APOE3/3 and APOE4/4 iBBBs respectively where B = BECs only, B/A = BECs and astrocytes, and B/iMC = BECs and iMural cells. Analysis was performed by One-way ANOVA with Bonferroni’s post-hoc analysis (p < 0.0001). Center values and error bars are means and SD from 5 independent iBBBs. B/iMCA3 v B/iMCA4, p = 0.0005; B/iMCA4 v- B3, p = 0.0001; B4, p = 0.0001; B/A3, p = 0.0064; B/A4, p = 0.0001; B/iMC3, p = 0.0026; B/iMC/A3 v B/iMC4, p < 0.0001. b, Exposing APOE4/4 astrocytes to APOE4/4 iMural cell conditioned media significantly increases amyloid accumulation compared APOE3/3 pericyte conditioned media. Unpaired two-sided Student t test, p = 0.0001. Center values and error bars are means and SD from 4 iBBBs. c, GO analysis from Toppfun (statistics described at https://toppgene.cchmc.org/enrichment.jsp) depicting biological processes associated with up-regulated and down-regulated genes. From RNA extracted from 3 independent wells of iMCs for each genotype.
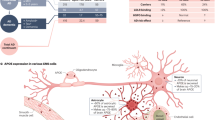
Extended Data Fig. 5 APOE expression is selectively elevated in APOE4 pericytes.
a, Quantification and representative image of APOE protein expression in brain vascular pericytes (NG2-positive cells) and non-pericytes (NG2-negative) cells in APOE4 knock-in mouse. Two-sided Student t test, p < 0.0001 Scale bar = 50 um. Center values and error bars are means from 150 APOE-positive cells for each genotype. b, anti-APOE antibody (Abcam, ab52607) is specific for human APOE protein and does not react with mouse Apoe protein or other proteins present in wild-type mouse hippocampus as reported by the manufacturer. The staining was repeated using 3 different mice c and d, Expression of APOE in isogenic iMural cells (c) and astrocyte (d) measured by RNA sequencing each condition represents three biological replicates pericyte, q = 0.0003 astrocyte, q = 0.0006 statistics performed by Dseq2, Center values and error bars are mean and SD from RNA prepared from 3 independent wells for each genotype. e Violin plots depicting APOE expression in pericytes/endothelial cells isolated from post-mortem prefrontal cortex of APOE4-carriers (n = 7) compared to non-carriers (n = 18). Differential expression was measured using a two-tailed Wilcoxon rank sum test, considering cells with detected expression of APOE (p = 0.0026). f, Images and quantification of APOE protein expression in post-mortem human prefrontal cortex from APOE4 carriers and non-carriers. Unpaired two-tailed t test (p = 0.023). Center values and error bars are mean and SD from staining of 4 prefrontal cortex sections from 4 different individuals. g, Differential plot of representative maker genes showing that pericytes and endothelial cells isolated from human hippocampus segregated into distinct cellular clusters (n = 82 individuals). h, Violin plots depicting APOE expression in endothelial cells isolated from post-mortem hippocampus APOE4-carriers (n = 16) compared to non-carriers (n = 46). Differential expression was measured using a two-tailed Wilcoxon rank sum test, considering cells with detected expression of APOE. Center lines are mean and dashed line are SD. i, Images and quantification of APOE protein expression α-SMA positive SMC in post-mortem human hippocampus from APOE4 carriers and non-carriers. Unpaired two-tailed t test (p = 0.5814). Center values are mean and SD of hippocampal sections from 6 individuals for each genotype. j, Western blot of APOE and GAPDH protein in a isogenic iPSC line in which the APOE gene was truncated via CRISPR-editing to yield a line that does not express of APOE protein and therefore is termed APOEKO line. The experiment was repeated at least 3 time with similar results.
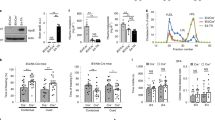
Extended Data Fig. 6 NFAT/Calcineurin signaling is selectively elevated in APOE4 pericytes.
a, Increasing the soluble APOE concentration through the addition of recombinant APOE protein to iBBB culture increases amyloid accumulation. One-way ANOVA with Bonferroni’s post-hoc analysis. Center values and error bars are mean and SD from 4 independent iBBBs for each condition. One-way ANOVA with Bonferroni’s multiple comparison test. APOE3/3 (Parental) V: APOE4/4 (Isogenic), p = 0.02; E3/3 + rAPOE3, p = 0.0034; E3/3 + rAPOE4, p = 0.0144. b and c, Representative western blot and quantification depicting nuclear NFATc1 expression in isogenic APOE3 and 4 iMural cells. Unpaired student t test, p = 0.0254. Experiment was repeated 3 times with similar results. (c) Center values and error bars are mean and SD from 3 independent lysate preparations for each genotype. d, Expression of calcineurin catalytic subunits measured by RNAseq. PPP3CA (q = 0.0003); PPP3CC (q = 0.0188). Analysis from DSEQ2. Center values and error bars are mean expression and SD of RNA prepared from 3 independent wells for each genoptype. e, Expression of negative Regulators of Calcineurin genes (RCANs) measured by RNAseq. RCAN2 (q = 0.0003); RCAN3 (q = 0.0123).). Analysis from DSEQ2. Center values and error bars are mean expression and SD of RNA prepared from 3 independent wells for each genoptype. f, Expression of DYRKs kinases known to phosphorylate NFAT measured by RNAseq. DYRK4 (q = 0.0003). Analysis from DSEQ2. Center values and error bars are mean expression and SD of RNA prepared from 3 independent wells for each genoptype. g, Expression of predicted NFAT response gene, VCAM1 and ACTG2, in iMural cells. Expression is quantified by qRT-PCR and normalized to the average of E3/3 cells. Significance determined by One-way ANOVA (p < 0.0001) with Bonferroni’s multiple comparison. For VCAM1 E3/3 (Parental) v- E4/4 (Isogenic), p < 0.0001; H9, p < 0.0001; 231, p = 0.0152; 332, p < 0.0001. For ACTG2 E3/3 (Parental) v- E4/4 (Isogenic), p = 0.0006; H9, p = 0.0108; 231, p = 0.0076; 332, p < 0.0001. Center values and error bars are mean expression and SD of RNA prepared from 4 independent wells for each cell line. h and i, Violin plots depicting NFATC1 (h) and NFATC2 (i) expression in pericytes isolated from post-mortem prefrontal cortex of APOE4-carriers (n = 16) compared to non-carriers (n = 46). Differential expression was measured using a two-tailed Wilcoxon rank sum test, considering cells with detected expression of APOE. Center lines are mean and dashed line are SD. j and k, Violin plots depicting NFATC2 expression in endothelial cells isolated from post-mortem prefrontal cortex of APOE4-carriers (n = 7 compared to non-carriers (n = 18). Differential expression was measured using a two-tailed Wilcoxon rank sum test, considering cells with detected expression of APOE (p = 0.035). Center lines are mean and dashed line are SD.
Extended Data Fig. 7 Inhibiting NFAT/Calcineurin signaling reduces APOE expression and CAA pathology.
a, Chemical structures of CsA, FK506, and INCA6 showing highly dissimilar structures. b, Expression of PGK1, HPRT, and GAPDH in pericytes after two weeks with DMSO, Cyclosporine A (CsA), FK506 or INCA6. One-way ANOVA (p < 0.0001) with Bonferroni’s multiple comparison. Center values and error bars are mean expression and SD of RNA prepared from 4 independent wells for each cell line. c and d, Representative immunofluorescence imaging of APOE protein staining in pericytes after two weeks of treatment with chemicals. Scale bar, 50 μm. Experiments were repeated at least 3 times with similar results. e DEGs and associated GO terms for up-regulated and down-regulated genes in E3 and E4 CsA-treated pericyte from RNA-sequencing of RNA prepared from 3 independent wells for each condition. DEGs were determined by DSEQ2 and GO analysis was performed with Toppfun. f and g, Representative imaging and quantification depicting APOE protein expression in the APOE4KI mouse cortical slices following treatment with cyclosporine A (CsA) for one week. Unpaired, two tailed t test (p = 0.0009). Experiments were repeated with similar results using at least 3 slice preparations for each condition. Center values and error bars are mean intensity and SD from 12 independent measurements. h, Quantification of amyloid APOE4KI mouse cortical slices treated with either CsA or FK506 for one week and then exposed to 20 nM Ab for 48 hours. One-way ANOVA (p = 0.0105) with Bonferroni’s multiple comparison. Control v- CsA, p = 0.0188; FK506, p = 0.0245. Center values and error bars are mean and SD from slices prepared from 3 different mice. i, APOE mRNA expression in primary pericytes isolated from brain microvasculature of APOE4 knock-in mice treated with DMSO, Cyclosporine A, or FK506. One-way ANOVA (p = 0.0139) with Bonferroni’s multiple comparison. For DMSO v- CsA, p = 0.0221; FK506, p = 0.0367. Center values and error bars are mean and SD from pericytes prepared from 3 different mice. j, Representative image of immunostaining for APOE in hippocampal pericytes from APOE4 KI x 5xFAD mice treated with cyclosporine A or vehicle for one week. k, Representative images of vascular amyloid in the hippocampus following treatment of 6-month-old APOE4KI x 5XFAD female mice with either vehicle or CsA. Amyloid was detected and quantified with two independent anti-amyloid antibodies (6e10 and 12F4). These experiments were repeated 2 times with similar results.
Supplementary information
Supplementary Table 1
Supplementary Table 1
Supplementary Video 1
iPSC-derived BECs in the iBBB stained for VE-cadherin shown in green.
Supplementary Video 2
The iBBB with endothelial cells stained for ZO-1 shown in red and iMCs stained with SM22 shown in green.
Supplementary Video 3
The iBBB with endothelial cells stained with VE-cadherin shown in red, astrocytes stained with S100b in blue and Aquaporin 4 shown in green.
Supplementary Video 4
APOE4/4 iBBBs accumulate more amyloid than isogenic APOE3/3 iBBBs.
Source data
Source Data Fig. 4
Unprocessed western blots.
Source Data Extended Data Fig. 5
Unprocessed western blots.
Source Data Extended Data Fig. 6
Unprocessed western blots.
Rights and permissions
About this article
Cite this article
Blanchard, J.W., Bula, M., Davila-Velderrain, J. et al. Reconstruction of the human blood–brain barrier in vitro reveals a pathogenic mechanism of APOE4 in pericytes. Nat Med 26, 952–963 (2020). https://doi.org/10.1038/s41591-020-0886-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-020-0886-4
This article is cited by
-
Cell type-specific roles of APOE4 in Alzheimer disease
Nature Reviews Neuroscience (2024)
-
A model for secondary traumatic stress following workplace exposure to traumatic material in analytical staff
Communications Psychology (2024)
-
Andrographolide Improves ApoE4-Mediated Blood–Brain Barrier Injury by Alleviating Inflammation
Molecular Neurobiology (2024)
-
The contribution of β-amyloid, Tau and α-synuclein to blood–brain barrier damage in neurodegenerative disorders
Acta Neuropathologica (2024)
-
Multisensory gamma stimulation promotes glymphatic clearance of amyloid
Nature (2024)