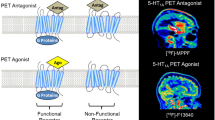
Abstract
Although aberrations in the number and function of glutamate AMPA (α-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid) receptors are thought to underlie neuropsychiatric disorders, no methods are currently available for visualizing AMPA receptors in the living human brain. Here we developed a positron emission tomography (PET) tracer for AMPA receptors. A derivative of 4-[2-(phenylsulfonylamino)ethylthio]-2,6-difluoro-phenoxyacetamide radiolabeled with 11C ([11C]K-2) showed specific binding to AMPA receptors. Our clinical trial with healthy human participants confirmed reversible binding of [11C]K-2 in the brain according to Logan graphical analysis (UMIN000020975; study design: non-randomized, single arm; primary outcome: dynamics and distribution volumes of [11C]K-2 in the brain; secondary outcome: adverse events of [11C]K-2 during the 4–10 d following dosing; this trial met prespecified endpoints). In an exploratory clinical study including patients with epilepsy, we detected increased [11C]K-2 uptake in the epileptogenic focus of patients with mesial temporal lobe epilepsy, which was closely correlated with the local AMPA receptor protein distribution in surgical specimens from the same individuals (UMIN000025090; study design: non-randomized, single arm; primary outcome: correlation between [11C]K-2 uptake measured with PET before surgery and AMPA receptor protein density examined by biochemical study after surgery; secondary outcome: adverse events during the 7 d following PET scan; this trial met prespecified endpoints). Thus, [11C]K-2 is a potent PET tracer for AMPA receptors, potentially providing a tool to examine the involvement of AMPA receptors in neuropsychiatric disorders.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
All requests for raw and analyzed data and materials are promptly reviewed by the Yokohama City University Research Promotion Department to determine whether the request is subject to any intellectual property or confidentiality obligations. Request for patient-related data may need to be inspected by the Institutional Review Board of Yokohama City University Hospital. Any data and materials that can be shared will be released via a material transfer agreement. Source data for Figs. 1 and 3 are presented with the paper.
Code availability
Program codes running LGA analysis are available and requests for them are promptly reviewed by the Yokohama City University Research Promotion Department to determine whether the request is subject to any intellectual property or confidentiality obligations.
References
Malinow, R. & Malenka, R. C. AMPA receptor trafficking and synaptic plasticity. Annu. Rev. Neurosci. 25, 103–126 (2002).
Kessels, H. W. & Malinow, R. Synaptic AMPA receptor plasticity and behavior. Neuron 61, 340–350 (2009).
Zhao, Y., Chen, S., Swensen, A. C., Qian, W. J. & Gouaux, E. Architecture and subunit arrangement of native AMPA receptors elucidated by cryo-EM. Science 364, 355–362 (2019).
Herguedas, B. et al. Architecture of the heteromeric GluA1/2 AMPA receptor in complex with the auxiliary subunit TARPγ8. Science 364, eaav9011 (2019).
Jitsuki, S. et al. Serotonin mediates cross-modal reorganization of cortical circuits. Neuron 69, 780–792 (2011).
Abe, H. et al. CRMP2-binding compound, edonerpic maleate, accelerates motor function recovery from brain damage. Science 360, 50–57 (2018).
Diering, G. H. & Huganir, R. L. The AMPA receptor code of synaptic plasticity. Neuron 100, 314–329 (2018).
Zhao, J. P. & Yoshii, A. Hyperexcitability of the local cortical circuit in mouse models of tuberous sclerosis complex. Mol. Brain 12, 6 (2019).
Chang, P. K., Verbich, D. & McKinney, R. A. AMPA receptors as drug targets in neurological disease—advantages, caveats, and future outlook. Eur. J. Neurosci. 35, 1908–1916 (2012).
Zanos, P. et al. NMDAR inhibition-independent antidepressant actions of ketamine metabolites. Nature 533, 481–486 (2016).
Kondej, M., Stepnicki, P. & Kaczor, A. A. Multi-target approach for drug discovery against schizophrenia. Int. J. Mol. Sci. 19, E3105 (2018).
Arstad, E. et al. Closing in on the AMPA receptor: synthesis and evaluation of 2-acetyl-1-(4'-chlorophenyl)-6-methoxy-7-[11C]methoxy-1,2,3,4-tetrahydroisoquinoline as a potential PET tracer. Bioorg. Med. Chem. 14, 4712–4717 (2006).
Gao, M., Kong, D., Clearfield, A. & Zheng, Q. H. Synthesis of carbon-11 and fluorine-18 labeled N-acetyl-1-aryl-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline derivatives as new potential PET AMPA receptor ligands. Bioorg. Med. Chem. Lett. 16, 2229–2233 (2006).
Lee, H. G., Milner, P. J., Placzek, M. S., Buchwald, S. L. & Hooker, J. M. Virtually instantaneous, room-temperature [11C]-cyanation using biaryl phosphine Pd(0) complexes. J. Am. Chem. Soc. 137, 648–651 (2015).
Oi, N. et al. Development of novel PET probes for central 2-amino-3-(3-hydroxy-5-methyl-4-isoxazolyl)propionic acid receptors. J. Med. Chem. 58, 8444–8462 (2015).
Yuan, G., Jones, G. B., Vasdev, N. & Liang, S. H. Radiosynthesis and preliminary PET evaluation of 18F-labeled 2-(1-(3-fluorophenyl)-2-oxo-5-(pyrimidin-2-yl)-1,2-dihydropyridin-3-yl)benzonitrile for imaging AMPA receptors. Bioorg. Med. Chem. Lett. 26, 4857–4860 (2016).
Takahata, K. et al. A human PET study of [11C]HMS011, a potential radioligand for AMPA receptors. EJNMMI Res. 7, 63 (2017).
Fleming, J. J. & England, P. M. Developing a complete pharmacology for AMPA receptors: a perspective on subtype-selective ligands. Bioorg. Med. Chem. 18, 1381–1387 (2010).
Sekiguchi, M. et al. A novel allosteric potentiator of AMPA receptors: 4-[2-(phenylsulfonylamino)ethylthio]-2,6-difluoro-phenoxyaceta mide. J. Neurosci. 17, 5760–5771 (1997).
Koga, K. et al. Development of TASP0410457 (TASP457), a novel dihydroquinolinone derivative as a PET radioligand for central histamine H3 receptors. EJNMMI Res. 6, 11 (2016).
Chernet, E. et al. Use of LC/MS to assess brain tracer distribution in preclinical, in vivo receptor occupancy studies: dopamine D2, serotonin 2A and NK-1 receptors as examples. Life Sci. 78, 340–346 (2005).
Toth, E., Sershen, H., Hashim, A., Vizi, E. S. & Lajtha, A. Effect of nicotine on extracellular levels of neurotransmitters assessed by microdialysis in various brain regions: role of glutamic acid. Neurochem. Res. 17, 265–271 (1992).
Tossman, U., Segovia, J. & Ungerstedt, U. Extracellular levels of amino acids in striatum and globus pallidus of 6-hydroxydopamine-lesioned rats measured with microdialysis. Acta Physiol. Scand. 127, 547–551 (1986).
Curras, M. C. & Dingledine, R. Selectivity of amino acid transmitters acting at N-methyl-d-aspartate and amino-3-hydroxy-5-methyl-4-isoxazolepropionate receptors. Mol. Pharmacol. 41, 520–526 (1992).
Stein, E., Cox, J. A., Seeburg, P. H. & Verdoorn, T. A. Complex pharmacological properties of recombinant α-amino-3-hydroxy-5-methyl-4-isoxazole propionate receptor subtypes. Mol. Pharmacol. 42, 864–871 (1992).
Innis, R. B. et al. Consensus nomenclature for in vivo imaging of reversibly binding radioligands. J. Cereb. Blood Flow Metab. 27, 1533–1539 (2007).
Logan, J. et al. Distribution volume ratios without blood sampling from graphical analysis of PET data. J. Cereb. Blood Flow Metab. 16, 834–840 (1996).
Citraro, R., Aiello, R., Franco, V., De Sarro, G. & Russo, E. Targeting α-amino-3-hydroxyl-5-methyl-4-isoxazole-propionate receptors in epilepsy. Expert Opin. Ther. Targets 18, 319–334 (2014).
Takemoto, K. et al. Optical inactivation of synaptic AMPA receptors erases fear memory. Nat. Biotechnol. 35, 38–47 (2017).
Zilles, K., Qu, M. S., Kohling, R. & Speckmann, E. J. Ionotropic glutamate and GABA receptors in human epileptic neocortical tissue: quantitative in vitro receptor autoradiography. Neuroscience 94, 1051–1061 (1999).
Krauss, G. L. et al. Randomized phase III study 306: adjunctive perampanel for refractory partial-onset seizures. Neurology 78, 1408–1415 (2012).
Finnema, S. J. et al. Imaging synaptic density in the living human brain. Sci. Transl. Med. 8, 348ra396 (2016).
Holmes, S. E. et al. Lower synaptic density is associated with depression severity and network alterations. Nat. Commun. 10, 1529 (2019).
Ward, S. E., Bax, B. D. & Harries, M. Challenges for and current status of research into positive modulators of AMPA receptors. Br. J. Pharmacol. 160, 181–190 (2010).
Tracy, T. E., Yan, J. J. & Chen, L. Acute knockdown of AMPA receptors reveals a trans-synaptic signal for presynaptic maturation. EMBO J. 30, 1577–1592 (2011).
Hammers, A. et al. Three-dimensional maximum probability atlas of the human brain, with particular reference to the temporal lobe. Hum. Brain Mapp. 19, 224–247 (2003).
Logan, J. et al. Graphical analysis of reversible radioligand binding from time–activity measurements applied to [N-11C-methyl]-(–)-cocaine PET studies inhuman subjects. J. Cereb. Blood Flow Metab. 10, 740–747 (1990).
Logan, J. Graphical analysis of PET data applied to reversible and irreversible tracers. Nucl. Med. Biol. 27, 661–670 (2000).
Acknowledgements
We gratefully thank T. Inoue for comprehensive support. This project was supported by Special Coordination Funds for Promoting Science and Technology (T.T.) and AMED under grants JP19dm0107124 (T.T.), JP18dm0207023 (T.T.), JP19lm0203007 (T.T.) and 19dm0107125 (M.M.).
Author information
Authors and Affiliations
Contributions
T.T. and T.M. designed the project and experiments, interpreted and analyzed data and wrote the manuscript. M. Higuchi and M.M. interpreted data. M.Y. and H.U. wrote the manuscript. M. Taguri performed statistical analysis. Y.S., Y. Kuroki, A. Serizawa, A. Sano, S.K., T.Y., M. Tokunaga, C.S., Y.N., T. Minamimoto and K. Kawamura performed and analyzed rodent experiments. W.N., M. Hatano, Y. Kimura, Y.I., F.Y. and H.K. analyzed human imaging data. H.T., N.N., T.K. and S.N. interpreted human imaging data. M.-R.Z., T.A., Y.T., K. Kimura and Y.H. synthesized [11C]K-2 and related compounds. N.I., M.S., M.I. and N.K. performed ATL. All authors contributed to and have approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
T.T. and T.M. are the inventors on a patent application claiming a novel compound that specifically binds to the AMPA receptor, which includes K-2 and K-2OH. The patent used in this work is “Novel compound that specifically binds to AMPA receptor” (WO/2017/006931), filed with the Shobayashi International Patent and Trademark Office.
Additional information
Peer review information Kate Gao was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 [11C] K-2 and [99mTc] HMPAO uptake in shRNA-injected rats.
(a) [11C] K-2 representative image obtained 20–40 min after radiotracer injection in the shRNA (shRNA targeting AMPARs and scramble construct)-injected rat. This experiment was repeated independently with similar results using 6 animals. (b) Representative SPECT image of the brain with [99mTc] HMPAO obtained 15–60 min after radiotracer injection in the shRNA-injected rat presented in (a). This experiment was repeated independently with similar results using 3 animals. (c) [99mTc] HMPAO uptake in the shRNA-injected rats. Data are presented after normalizing the radiotracer retention in the shRNA targeting to AMPARs-injected sides, with values from the scramble construct-injected sides. n = 3 animals. *P = 0.50 (Wilcoxon signed-rank test with two-tailed). Data are shown as the mean ± SEM.
Extended Data Fig. 2 Identification of a metabolized form of K-2 (K-2OH).
(a) Radio-HPLC analysis of [11C] K-2 metabolites in the rat brain homogenate (incubation: 37 °C, 60 min). The HPLC chart shows K-2 and major metabolite (K-2 metabolite) peaks. (b) HPLC analysis (UV, 254 nm) of a non-labelled K-2 metabolite in the rat brain homogenate (incubation: 37 °C, 30 min). The HPLC chart shows K-2 and major metabolite (K-2 metabolite) peaks. (c) HPLC analysis (UV, 254 nm) of K-2. (d) MS spectra of a major metabolite of K-2 in the rat brain homogenate (incubation: 37 °C, 60 min). Red arrowheads indicate peaks at 418, 440 and 456 m/z corresponding to M+H+, M+Na+ and M+K+, respectively. (e) The structure of K-2OH (major metabolite of K-2) was determined by HPLC, radio-HPLC and LC-MS analysis of the rat brain homogenate. (f) Synthetic scheme for the chemical conversion from K-2 to K-2OH. (g) HPLC analysis (UV, 254 nm) of synthesized K-2OH. (h) MS spectra of synthesized K-2OH. Red arrowheads indicate peaks at 418, 440 and 456 m/z, corresponding to M+H+, M+Na+ and M+K+, respectively. The HPLCs for (a) to (c) and (g) were as follows: column: Waters Atlantis T3 Analytical Columns 5 µm 4.6 × 250 mm, mobile phase A: 0.1% formic acid water, mobile phase B: 0.1% formic acid acetonitrile, gradient: 0 min 20% of B, 8 min 80% of B, 13 min 80% of B, 16 min 20% of B, 25 min 20% of B. All experiments in these figures were performed once.
Extended Data Fig. 3 Identification of structures of K-2 and K-2OH with NMR.
(a) 1H NMR chart of K-2. (b) 13C NMR chart of K-2. (c) 1H NMR chart of K-2OH. (d) 13C NMR chart of K-2OH. These chemical shifts of (a) to (d) are described in the Methods section. All experiments in these figures were performed once.
Extended Data Fig. 4 Temporal metabolic rate of K-2 and affinity of K-2 and K-2OH.
(a) Fractions of non-metabolized (black line) and metabolized (grey line) K-2 in the rat brain 3-20 min after intravenous injection. n = 4 animals at each time point. Data are shown as the mean ± SEM. (b) Non- metabolized (black line) and metabolized (grey line) [11C] K-2 in the human plasma 0–60 min after radiotracer injection for 6 participants. Data are shown as the mean ± SEM. (c) Typical saturation curve for [11C] K-2OH binding to acute striatum slices. Nonspecific binding was calculated in the presence of 400 µM K-2OH. This experiment was repeated independently with similar results twice. (d) Typical saturation curve for [11C] K-2 binding to acute striatum slices. Nonspecific binding was calculated in the presence of 100 µM PEPA. This experiment was repeated independently with similar results twice.
Extended Data Fig. 5 The effect of K-2 on NMDA and Kainate receptor-mediated synaptic currents.
(a) Averaged evoked NMDAR-mediated EPSCs (pA) of pre and post K-2 application at 150 μM. n = 7cells. t (7) =0.5411, *P = 0.6052 (paired two-tailed t-test). (b) Averaged evoked Kainate receptor-mediated EPSCs of pre-and post K-2 application at 150 μM. For n= 6 cells. t (6) =0.6008, *P = 0.5700 (paired two-tailed t-test).
Extended Data Fig. 6 In vitro binding affinity of [11C] K-2 to AMPARs.
Typical saturation curve for [11C] K-2 binding to recombinant GluA1 (a), GluA2 (b), GluA3 (c), GluA4 (d), GluA1/2 heteromer (e), GluA2/3 heteromer (f) expressed in COS cells and (g) comparative table showing relative KD value among these subunits. (h) Typical saturation curve for [11C] K-2 binding to recombinant GluA2/3 heteromer expressed in COS cells under 2 µM glutamate. Nonspecific binding was calculated in the presence of 100 µM K-2. All experiment in these figures were repeated independently with similar results twice.
Extended Data Fig. 7 Relationship between VT with pTAC and BPND with reference.
(a) Linear regression analysis between BPND using white matter as a reference and VT of brain regions (except white matter) computed with pTAC in 6 healthy individuals. (b) Comparison of VT value, which corresponds to the zero point of BPND, using white matter as a reference (X intercept of the plot in (a)) and VT of white matter computed with pTAC for n= 6 individuals. t (5) =0.1291, P = 0.9023 (paired two-tailed t-test). (c) Correlation between BPND using white matter as a reference (without pTAC) and BPND computed with pTAC in 11 brain regions per subject. n= 6 participants (Pearson correlation analysis with two-tailed; correlation coefficient = 0.9844, P < 0.0001, Y = 1.033*X + 0.01463). Individual parametric PET images of [11C] K-2 in healthy participants showing BPND (d, n = 6 participants) and SUVR30-50 min (e, n = 6 participants). (f) Representative orthogonal parametric PET images of SUVR30-50 min presented in Fig. 2e with unadjusted color scale (lowest SUVR value set to be 0). n = 6 participants.
Extended Data Fig. 8 PET imaging characteristics of [11C] K-2 in patients with epilepsy.
(a) Logan graphical analysis (LGA) where the ratio between an integrated tTAC in the reference region (white matter; CR) and a tTAC (C), and an integrated C and C itself, are plotted on x- and y-axes, respectively. LGA of a representative patient with MTLE is shown. (b) Correlation between SUVR30-50 min -1 and BPND obtained from LGA in 11 brain regions per subject. n = 6 patients with epilepsy (Pearson correlation analysis with two-tailed; correlation coefficient = 0.9871, P < 0.0001, Y = 1.018*X + 0.01154). (c–f) Individual correlation between SUVR30-50 min for [11C]K-2 and biochemical amount of AMPARs in resected areas of patients with MTLE (Patient 1, n = 10 samples, Pearson correlation analysis with two-tailed; correlation coefficient = 0.6121, P = 0.06, Y = 4.185e-008*X + 1.638: Patient 3, n = 14 samples, Pearson correlation analysis with two-tailed; correlation coefficient = 0.7356, P = 0.0027, Y = 6.532e-008*X + 1.243: Patient 4, n = 13 samples, Pearson correlation analysis with two-tailed; correlation coefficient = 0.5909, P = 0.00334, Y = 6.603e-008*X + 1.45: Patient 8, n = 10 samples, Pearson correlation analysis with two-tailed; correlation coefficient = 0.7577, P = 0.0111, Y = 6.219e-008*X + 1.372).
Extended Data Fig. 9 [11C]K-2 in patients with epilepsy in another color scale.
SUVR30-50 min images of [11C] K-2 were obtained from the six MTLE patients presented in Fig. 2e with unadjusted color scale (lowest SUVR value set to be 0).
Supplementary information
Supplementary Information
Supplementary Tables 1–3 and CONSORT checklist
Source data
Source Data Fig. 1
Unprocessed western blots for Fig. 1i
Source Data Fig. 3
Unprocessed western blots for Fig. 3b
Rights and permissions
About this article
Cite this article
Miyazaki, T., Nakajima, W., Hatano, M. et al. Visualization of AMPA receptors in living human brain with positron emission tomography. Nat Med 26, 281–288 (2020). https://doi.org/10.1038/s41591-019-0723-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-019-0723-9
This article is cited by
-
Enhancement of angiotensin II type 1 receptor-associated protein in the paraventricular nucleus suppresses angiotensin II-dependent hypertension
Hypertension Research (2024)
-
Quantifying AMPARs with 99mTc-omberacetam: a novel diagnostic radiotracer for ischemic stroke
Journal of Umm Al-Qura University for Applied Sciences (2024)
-
The molecular pathology of schizophrenia: an overview of existing knowledge and new directions for future research
Molecular Psychiatry (2023)
-
Neurological update: structural and functional imaging in epilepsy surgery
Journal of Neurology (2023)
-
Glutamatergic dysfunction in Schizophrenia
Translational Psychiatry (2022)