Abstract
Human immunodeficiency virus 1 (HIV-1) infection is associated with heightened inflammation and excess risk of cardiovascular disease, cancer and other complications. These pathologies persist despite antiretroviral therapy. In two independent cohorts, we found that innate lymphoid cells (ILCs) were depleted in the blood and gut of people with HIV-1, even with effective antiretroviral therapy. ILC depletion was associated with neutrophil infiltration of the gut lamina propria, type 1 interferon activation, increased microbial translocation and natural killer (NK) cell skewing towards an inflammatory state, with chromatin structure and phenotype typical of WNT transcription factor TCF7-dependent memory T cells. Cytokines that are elevated during acute HIV-1 infection reproduced the ILC and NK cell abnormalities ex vivo. These results show that inflammatory cytokines associated with HIV-1 infection irreversibly disrupt ILCs. This results in loss of gut epithelial integrity, microbial translocation and memory NK cells with heightened inflammatory potential, and explains the chronic inflammation in people with HIV-1.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the manuscript and its Supplementary Information, and from the corresponding author upon request. Source data for Figs. 1, 2 and 6–8 and Extended Data Figs. 1–3 and 5–7 are provided with the paper. Bulk and single-cell RNA-seq, CUT&RUN and ATAC-seq datasets can be found under SuperSeries GSE122326 at https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE97727 (CD94– and CD94+ NK cell bulk and single-cell RNA-seq (GSE97727); CD94–CD56dim, CD94+CD56dim and CD94+CD56hi NK cell RNA-seq (GSE122324); CD94–CD56dim NK cells, 1° stim and 5-d culture RNA-seq (GSE122325); CD94–CD56dim, CD94+CD56dim and CD94+CD56hi NK cell ATAC-seq (GSE122548); and CD94–CD56dim, CD94+CD56dim and CD94+CD56hi NK cell CUT&RUN (GSE122549)).
References
Deeks, S. G., Tracy, R. & Douek, D. C. Systemic effects of inflammation on health during chronic HIV infection. Immunity 39, 633–645 (2013).
Brenchley, J. M. et al. Microbial translocation is a cause of systemic immune activation in chronic HIV infection. Nat. Med. 12, 1365–1371 (2006).
Vivier, E. et al. Innate lymphoid cells: 10 years on. Cell 174, 1054–1066 (2018).
Kløverpris, H. N. et al. Innate lymphoid cells are depleted irreversibly during acute HIV-1 infection in the absence of viral suppression. Immunity 44, 391–405 (2016).
Mudd, J. C. et al. Hallmarks of primate lentiviral immunodeficiency infection recapitulate loss of innate lymphoid cells. Nat. Commun. 9, 3967 (2018).
Bruel, T. et al. Elimination of HIV-1-infected cells by broadly neutralizing antibodies. Nat. Commun. 7, 10844 (2016).
Alter, G. et al. HIV-1 adaptation to NK-cell-mediated immune pressure. Nature 476, 96–100 (2011).
Cerwenka, A. & Lanier, L. L. Natural killer cell memory in infection, inflammation and cancer. Nat. Rev. Immunol. 16, 112–123 (2016).
Lim, A. I. et al. Systemic human ILC precursors provide a substrate for tissue ILC differentiation. Cell 168, 1086–1100.e10 (2017).
Colonna, M. Innate lymphoid cells: diversity, plasticity, and unique functions in immunity. Immunity 48, 1104–1117 (2018).
Mazzucchelli, R. & Durum, S. K. Interleukin-7 receptor expression: intelligent design. Nat. Rev. Immunol. 7, 144–154 (2007).
Leonard, W. J. & O’Shea, J. J. JAKS and STATs: biological implications. Annu. Rev. Immunol. 16, 293–322 (1998).
Mavilio, D. et al. Characterization of CD56–/CD16+ natural killer (NK) cells: a highly dysfunctional NK subset expanded in HIV-infected viremic individuals. Proc. Natl Acad. Sci. USA 102, 2886–2891 (2005).
Fang, M. et al. CD94 is essential for NK cell-mediated resistance to a lethal viral disease. Immunity 34, 579–589 (2011).
Jeevan-Raj, B. et al. The transcription factor Tcf1 contributes to normal NK cell development and function by limiting the expression of granzymes. Cell Rep. 20, 613–626 (2017).
Wendel, M., Galani, I. E., Suri-Payer, E. & Cerwenka, A. Natural killer cell accumulation in tumors is dependent on IFN-γ and CXCR3 ligands. Cancer Res. 68, 8437–8445 (2008).
Sconocchia, G., Titus, J. A. & Segal, D. M. Signaling pathways regulating CD44-dependent cytolysis in natural killer cells. Blood 90, 716–725 (1997).
Liu, L. L. et al. Critical role of CD2 co-stimulation in adaptive natural killer cell responses revealed in NKG2C-deficient humans. Cell Rep. 15, 1088–1099 (2016).
Juelke, K. et al. CD62L expression identifies a unique subset of polyfunctional CD56dim NK cells. Blood 116, 1299–1307 (2010).
Gazit, R. et al. Expression of KIR2DL1 on the entire NK cell population: a possible novel immunodeficiency syndrome. Blood 103, 1965–1966 (2004).
Klein, A. M. et al. Droplet barcoding for single-cell transcriptomics applied to embryonic stem cells. Cell 161, 1187–1201 (2015).
Tibshirani, R. & Walther, G. Cluster validation by prediction strength. J. Comput. Graph. Stat. 14, 511–528 (2005).
Trapnell, C. et al. The dynamics and regulators of cell fate decisions are revealed by pseudotemporal ordering of single cells. Nat. Biotechnol. 32, 381–386 (2014).
Utzschneider, D. T. et al. T cell factor 1-expressing memory-like CD8+ T cells sustain the immune response to chronic viral infections. Immunity 45, 415–427 (2016).
Jeannet, G. et al. Essential role of the Wnt pathway effector Tcf-1 for the establishment of functional CD8 T cell memory. Proc. Natl Acad. Sci. USA 107, 9777–9782 (2010).
Yang, Q. et al. TCF-1 upregulation identifies early innate lymphoid progenitors in the bone marrow. Nat. Immunol. 16, 1044–1050 (2015).
Aksoy, I. et al. Self-renewal of murine embryonic stem cells is supported by the serine/threonine kinases Pim-1 and Pim-3. Stem Cells 25, 2996–3004 (2007).
Baaten, B. J. G. et al. CD44 regulates survival and memory development in Th1 cells. Immunity 32, 104–115 (2010).
Weng, N.-P., Araki, Y. & Subedi, K. The molecular basis of the memory T cell response: differential gene expression and its epigenetic regulation. Nat. Rev. Immunol. 12, 306–315 (2012).
Moretta, L. Dissecting CD56dim human NK cells. Blood 116, 3689–3691 (2010).
Paust, S. et al. Critical role for the chemokine receptor CXCR6 in NK cell-mediated antigen-specific memory of haptens and viruses. Nat. Immunol. 11, 1127–1135 (2010).
Roychoudhuri, R. et al. BACH2 regulates CD8+ T cell differentiation by controlling access of AP-1 factors to enhancers. Nat. Immunol. 17, 851–860 (2016).
Shin, H. M. et al. Epigenetic modifications induced by Blimp-1 regulate CD8+ T cell memory progression during acute virus infection. Immunity 39, 661–675 (2013).
Kamimura, Y. & Lanier, L. L. Homeostatic control of memory cell progenitors in the natural killer cell lineage. Cell Rep. 10, 280–291 (2015).
Brenchley, J. M. et al. Expression of CD57 defines replicative senescence and antigen-induced apoptotic death of CD8+ T cells. Blood 101, 2711–2720 (2003).
Wherry, E. J. et al. Molecular signature of CD8+ T cell exhaustion during chronic viral infection. Immunity 27, 670–684 (2007).
Lin, W.-H. W. et al. CD8+ T lymphocyte self-renewal during effector cell determination. Cell Rep. 17, 1773–1782 (2016).
Vigneau, S., Rohrlich, P.-S., Brahic, M. & Bureau, J.-F. Tmevpg1, a candidate gene for the control of Theiler’s virus persistence, could be implicated in the regulation of gamma interferon. J. Virol. 77, 5632–5638 (2003).
Gomez, J. A. et al. The NeST long ncRNA controls microbial susceptibility and epigenetic activation of the interferon-γ locus. Cell 152, 743–754 (2013).
Walker, W., Aste-Amezaga, M., Kastelein, R. A., Trinchieri, G. & Hunter, C. A. IL-18 and CD28 use distinct molecular mechanisms to enhance NK cell production of IL-12-induced IFN-γ. J. Immunol. 162, 5894–5901 (1999).
Schoenborn, J. R. & Wilson, C. B. Regulation of interferon‐γ during innate and adaptive immune responses. Adv. Immunol. 96, 41–101 (2007).
Björkström, N. K. et al. Expression patterns of NKG2A, KIR, and CD57 define a process of CD56dim NK-cell differentiation uncoupled from NK-cell education. Blood 116, 3853–3864 (2010).
Romee, R. et al. Cytokine activation induces human memory-like NK cells. Blood 120, 4751–4760 (2012).
Cooper, M. A. et al. Cytokine-induced memory-like natural killer cells. Proc. Natl Acad. Sci. USA 106, 1915–1919 (2009).
Henning, A. N., Roychoudhuri, R. & Restifo, N. P. Epigenetic control of CD8+ T cell differentiation. Nat. Rev. Immunol. 18, 340–356 (2018).
Hu, G. & Chen, J. A genome-wide regulatory network identifies key transcription factors for memory CD8+ T-cell development. Nat. Commun. 4, 2830 (2013).
O’Sullivan, T. E., Sun, J. C. & Lanier, L. L. Natural killer cell memory. Immunity 43, 634–645 (2015).
Xing, S. et al. Tcf1 and Lef1 transcription factors establish CD8+ T cell identity through intrinsic HDAC activity. Nat. Immunol. 17, 695–703 (2016).
Thomas, R. et al. NKG2C deletion is a risk factor of HIV infection. AIDS Res. Hum. Retroviruses 28, 844–851 (2012).
Fregni, G. et al. High number of CD56bright NK-cells and persistently low CD4+ T-cells in a hemophiliac HIV/HCV co-infected patient without opportunistic infections. Virol. J. 10, 33 (2013).
Davis, Z. B. et al. A conserved HIV-1-derived peptide presented by HLA-E renders infected T-cells highly susceptible to attack by NKG2A/CD94-bearing natural killer cells. PLoS Pathog. 12, e1005421 (2016).
Neri, S., Mariani, E., Meneghetti, A., Cattini, L. & Facchini, A. Calcein-acetyoxymethyl cytotoxicity assay: standardization of a method allowing additional analyses on recovered effector cells and supernatants. Clin. Diagn. Lab. Immunol. 8, 1131–1135 (2001).
Pertel, T. et al. TRIM5 is an innate immune sensor for the retrovirus capsid lattice. Nature 472, 361–365 (2011).
Poli, A. et al. CD56bright natural killer (NK) cells: an important NK cell subset. Immunology 126, 458–465 (2009).
Hashimshony, T. et al. CEL-Seq2: sensitive highly-multiplexed single-cell RNA-Seq. Genome Biol. 17, 77 (2016).
Skene, P. J. & Henikoff, S. An efficient targeted nuclease strategy for high-resolution mapping of DNA binding sites. Elife 6, e21856 (2017).
Hainer, S. J., Boskovic, A., Rando, O. J. & Fazzio, T. G. Profiling of pluripotency factors in individual stem cells and early embryos. Cell 177, 1319–1329.e11 (2019).
Skene, P. J., Henikoff, J. G. & Henikoff, S. Targeted in situ genome-wide profiling with high efficiency for low cell numbers. Nat. Protoc. 13, 1006–1019 (2018).
Buenrostro, J. D., Wu, B., Chang, H. Y. & Greenleaf, W. J. ATAC-Seq: a method for assaying chromatin accessibility genome-wide. Curr. Protoc. Mol. Biol. 109, 21.29.1–21.29.9 (2015).
Buenrostro, J. D., Giresi, P. G., Zaba, L. C., Chang, H. Y. & Greenleaf, W. J. Transposition of native chromatin for fast and sensitive epigenomic profiling of open chromatin, DNA-binding proteins and nucleosome position. Nat. Methods 10, 1213–1218 (2013).
Kim, D. et al. TopHat2: accurate alignment of transcriptomes in the presence of insertions, deletions and gene fusions. Genome Biol. 14, R36 (2013).
Derr, A. et al. End Sequence Analysis Toolkit (ESAT) expands the extractable information from single-cell RNA-Seq data. Genome Res. 26, 1397–1410 (2016).
Love, M., Anders, S. & Huber, W. Differential analysis of count data—the DESeq2 package. Genome Biol. 15, 550 (2014).
Robinson, M. D. & Oshlack, A. A scaling normalization method for differential expression analysis of RNA-seq data. Genome Biol. 11, R25 (2010).
Hyvärinen, A. & Oja, E. Independent component analysis: algorithms and applications. Neural Netw. 13, 411–430 (2000).
Hechenbichler, K. & Schliep, K. Weighted k-Nearest-Neighbor Techniques and Ordinal Classification Discussion Paper 399 (Ludwig-Maximilians-Universität München Collaborative Reseach Center 386, 2004).
Robinson, M. D., McCarthy, D. J. & Smyth, G. K. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26, 139–140 (2010).
Van Der Maaten, L. Accelerating t-SNE using tree-based algorithms. J. Mach. Learn. Res. 15, 3221–3245 (2014).
Zhang, Y. et al. Model-based analysis of ChIP-Seq (MACS). Genome Biol. 9, R137 (2008).
Robinson, J. T. et al. Integrative genomics viewer. Nat. Biotechnol. 29, 24–26 (2011).
Acknowledgements
We thank the study participants who provided blood and colon biopsy samples, as well as their caretakers, J. Daly, S. Cheeseman and M. Wessolossky of the UMMS. C. Mannarino, A. Foley, M. McManus (UMMS) and M. Krone (UCSF) provided Institutional Review Board regulatory assistance, sample preparation and record keeping. K. Luzuriaga (UMMS) supported the patient sample database and repository. A. Ratner, S. Boswell and A. Klein (Harvard Medical School) contributed technical assistance and barcoded hydrogel beads. T. Fazzio and T. Wu provided technical support and protein A-MNase for CUT&RUN. D. Artis, L. Berg, M. Colonna, J. Huh, J. Kang, R. Rutishauser and S. Swain offered invaluable advice. This research was supported by NIH grants U01HG007910 (to M.G. and J.L.), R37AI147868 (to J.L.), R01AI111809 (to J.L.), DP1DA034990 (to J.L.), R21AI119885 (to M.G.), R01DK105837 (to M.G.) and P51OD01192 (to J.D.E. at the Oregon National Primate Research Center), and the Translational Medicine Core of the University of Massachusetts Center for AIDS Research (P30 AI042845). The UCSF-based SCOPE cohort was supported by the UCSF/Gladstone Institute of Virology and Immunology CFAR (P30 AI027763) and the CFAR Network of Integrated Systems (R24 AI067039). Additional support was provided by the Delaney AIDS Research Enterprise (AI096109 and AI127966). Funding for this study was provided in part by the Division of Intramural Research/NIAID/NIH (to J.M.B.). The content of this publication does not necessarily reflect the views or policies of DHHS, nor does the mention of trade names, commercial products, or organizations imply endorsement by the US Government.
Author information
Authors and Affiliations
Contributions
Y.W. and J.L. designed the experiments. Y.W. performed the experiments with assistance from C.L.V., K.B.-S., S.J., K.G., A.D., L.L., S.M., K.K., P.V., P.W.H., S.G.D., J.M.B., J.D.E. and P.M. Y.W. and J.L. analyzed the experimental data. Y.W., L.L., K.G., P.V., A.D., A.K., M.G. and J.L. analyzed the expression data. T.G., J.H., M.S. and S.G.D. obtained and provided the clinical samples. Y.W. and J.L. wrote the manuscript, which was revised and approved by all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Zoltan Fehervari was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
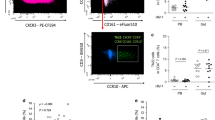
Extended Data Fig. 1 ILC_gating_and_effect_of_HIV-1.
a, Lymphoid, singlet, live, CD45+ PBMCs from HIV-1– individuals were stained with lineage antibodies (see Methods), CD56, and CD16, and the percent ILCs were Lin–CD56–CD16–CD127+ cells. b, CD117 and CRTH2 on Lin–CD56–CD16–CD127+ cells from HIV-1– PBMCs (n = 11). c, ILCs from HIV-1–, HIV-1+ viremic, HIV-1+ on ART, and HIV-1+ spontaneous controllers, as in a (Supplementary Table 1). d, Correlation of ILCs (Lin–CD56–CD16–CD127+PBMCs) with CD4+ nadir (Supplementary Table 1). Correlation coefficient (R) by Pearson, zero slope p value determined by F-test (n = 80). e, CD127, CD117, and RORγt in Lin– colon lamina propria from HIV-1–. f, IL-22 and CD127 on Lin– colon lamina propria from HIV-1–, 3 hrs PMA/ionomycin. g, Lin– colon lamina propria, CD127 versus RORγT from HIV-1– and HIV-1+ (Supplementary Table 2). h, Colon lamina propria CD4+, 3 HIV-1– and 5 HIV-1+ (Supplementary Table 2). i, Percent CD3+CD117– cells in rectosigmoid tissue from 8 HIV-1– and 16 HIV-1+ on ART (Supplementary Table 1). j, Trimmed mean of M-values (DEBrowser), RNA-Seq from sorted Lin–CD127+ILCs of 4 HIV-1–. k, Percent ILCs within Lin–CD56–CD16– PBMCs from HIV-1– donors after incubation with IL-2 or IL-4 for 16 hrs (n = 5). l, IL-22 produced by gut lamina propria ILC3s maintains epithelium integrity (left). Irreversible decrease in ILC3s with HIV-1 infection (right). Data are mean ± s.e.m.; h,i, two tailed unpaired t-test; b,k, two tailed paired t-test. ns, not significant, *p < 0.05, **p < 0.001.
Extended Data Fig. 2 HIV-1_infection_increases_CD94+NK_cells.
a, Fraction of CD94+NK cells among Lin–TBX21+ PBMCs after stimulation with PMA and ionomycin (n = 10) or with IL-15 (n = 10), or with IL-12 + IL15 (n = 4). b, Sorting strategy for CD94– and CD94+NK cells. c, Percent CD107a among CD94– and CD94+NK cells after PBMCs were stimulated with PMA/iono (n = 5). d, Percent specific lysis of K562 or Jurkat cells by sorted CD94–NK cells and CD94+NK cells (n = 8).e, Percent Ki67 and Annexin V among CD94– or CD94+NK cells after the indicated treatment (n = 4). f, Representative flow cytometry for indicated genes as detected in Fig. 2h. Data are mean ± s.e.m. Each dot represents a unique sample. two-tailed paired t-test, lines connect cells from common donor. ns, not significant, *p < 0.05, **p < 0.01, ***p < 0.001. All data are from HIV-1– anonymous blood donors.
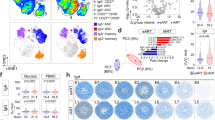
Extended Data Fig. 3 Single_cell_analysis_of_CD94-_and_CD94+NK_cells.
a, Heatmap of 1,729 CD94– (blue) and 1,548 CD94+NK cells (yellow) sorted from 2 donors using all differentially expressed genes based on CD94 positivity. b, Plot of predictive strength as a function of the number of clusters in Fig. 3b shows that 2 clusters yield stable and significant groupings, while separation into additional clusters artificially segregates the cells. The predictive strength based on 1753 single cells was calculated using spectral clustering on the ICA components. c, Heatmap from Fig. 3c was reconstructed utilizing the pseudotime ordering of single cells based on the minimum spanning tree. d, Flow cytometry for CD44, CXCR3 and SELL on TCF7– and TCF7+ NK cells. e, Flow cytometry for GZMK after sorted Lin–CD56+CD94–NK cells were treated as in Fig. 3h. f,g, PBMCs were treated with or without IL-15 for 5 days, Lin–CD56+ cells were gated on CD56 and CD94 (f) and percent CD56hi NK cells (g) (n = 10). Data are mean ± s.e.m. two-tailed paired t-test, *p < 0.001. All data are from HIV-1– anonymous blood donors.
Extended Data Fig. 4 Distinct_chromatin_landscape_of_CD94+CD56hiNK_cells.
a, PCA based on H3K4me3 CUT&RUN of the indicated NK cell subsets (n = 2). b, Correlation between differentially expressed genes and enriched H3K4me3 regions by CUT&RUN (log2 fold change) p < 0.001. The correlation coefficient (R) was determined by Pearson, p value for the slope being zero was determined by the F-test. c, Differential signals for H3K4me3 CUT&RUN and ATAC-Seq at the indicated loci in the indicated NK cell subsets. d, Overlapping signal for TCF7 CUT&RUN and ATAC-Seq at the indicated loci. Data are from HIV-1– blood donors.
Extended Data Fig. 5 Memory_associated_gene_loci_are_accessible_in_the_CD94+CD56hiNK_cells.
a, H3K4me1 and H3K4me3 CUT&RUN and ATAC-Seq signal on genes associated with memory T and NK cells, except for effector marker KLRG1, on the indicated NK cell subsets. b, IFN-γ production among CD56dim and CD56hiNK cells after stimulation with IL-12 + IL-15 for 16 hr (n = 4). mean ± s.e.m.; two tailed paired t-test, *p < 0.01. c, H3K4me1 and H3K4me3 CUT&RUN and ATAC-Seq signal at loci for IFN-γ signaling related genes. Data are from HIV-1– blood donors.
Extended Data Fig. 6 Surface_markers_and_WNT-associated_gene_loci_comparing_CD56dim_and_CD56hiNK_cells.
a,b, Detection of CD16 (n = 4), KIR2DL1 (n = 7), KIR2DL2/3 (n = 8), KIR3DL1 (n = 8) and CD57 (n = 8) in CD56dim and CD56hiNK cells from PBMCs, mean ± s.e.m.; two tailed paired t-test, *p < 0.001. c, Sorted, CFSE labelled CD94+CD56hiNK cells were cultured in IL-12 (10 ng/ml) and IL-15 (10 ng/ml) for 5 days. Proliferation, CXCR6, and CD57 were detected as indicated. CD56dim and CD56hi NK cells from fresh, unstimulated PBMCs were used as control. d, H3K4me3 CUT&RUN and ATAC-Seq signals for gene loci of WNT signaling components and WNT target genes in the indicated NK cell subsets. AXIN1, in contrast, is a WNT inhibitory gene. Data are from HIV-1– blood donors.
Extended Data Fig. 7 WNT_inhibition_blocks_cytokine_induced_NK_cell_memory.
a, PBMCs were treated with or without LGK974 for 16 hrs. Percentage of Lin–TBX21+ cells, and the CD94– and CD94+ cells among the Lin–TBX21+ population, are indicated (left); data are representative of 4 anonymous HIV-1– blood donors. PBMCs were stimulated with IL-12 and IL-15 for 16 hrs in the absence or presence of LGK974. Live cells and Lin–CD56+ cells were examined (right); data are representative of 10 anonymous HIV-1– blood donors. b, NK cells without primary stimulation as in Fig. 7a were stimulated with IL-12 + IL-15, then IFN-γ production of cells with or without LGK974 was detected (n = 8), samples are from HIV-1– donors. c, Magnetic beads enriched NK cells from HIV-1– donors were transduced with lentivectors expressing GFP and shRNAs targeting either TCF7 or control, the TCF7 level in GFP+ cells was detected by flow cytometry. d, Control or TCF7 knockdown NK cell in c were treated as in Fig. 7a, percent IFN-γ+ among GFP+ cell were detected after secondary stimulation (n = 3). e, Correlation of ILCs with TCF7+NK cells. Samples are from HIV-1+ viremic individuals, ART suppressed HIV-1+ individuals (ART), and HIV-1+ individuals who spontaneously control viremia without ART. Cohort characteristics are described in Supplementary Table 1 (n = 53). The correlation coefficient (R) was determined by Pearson, p value for the slope being zero was determined by the F-test. Data are mean ± s.e.m; b,d, two tailed paired t-test, ns, not significant, *p < 0.01.
Supplementary information
Supplementary Tables
Supplementary Tables 1–10.
Source data
Source Data Fig. 1
Statistical Source Data
Source Data Fig. 2
Statistical Source Data
Source Data Fig. 6
Statistical Source Data
Source Data Fig. 7
Statistical Source Data
Source Data Fig. 8
Statistical Source Data
Source Data Extended Data Fig. 1
Statistical Source Data
Source Data Extended Data Fig. 2
Statistical Source Data
Source Data Extended Data Fig. 3
Statistical Source Data
Source Data Extended Data Fig. 5
Statistical Source Data
Source Data Extended Data Fig. 6
Statistical Source Data
Source Data Extended Data Fig. 7
Statistical Source Data
Rights and permissions
About this article
Cite this article
Wang, Y., Lifshitz, L., Gellatly, K. et al. HIV-1-induced cytokines deplete homeostatic innate lymphoid cells and expand TCF7-dependent memory NK cells. Nat Immunol 21, 274–286 (2020). https://doi.org/10.1038/s41590-020-0593-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41590-020-0593-9
This article is cited by
-
Identification of the shared hub gene signatures and molecular mechanisms between HIV-1 and pulmonary arterial hypertension
Scientific Reports (2024)
-
Immune mechanisms in the pathophysiology of hypertension
Nature Reviews Nephrology (2024)
-
Autologous dendritic cell vaccination against HIV-1 induces changes in natural killer cell phenotype and functionality
npj Vaccines (2023)
-
Direct intranodal tonsil vaccination with modified vaccinia Ankara vaccine protects macaques from highly pathogenic SIVmac251
Nature Communications (2023)
-
Natural killer cell responses during SARS-CoV-2 infection and vaccination in people living with HIV-1
Scientific Reports (2023)