Abstract
A wealth of specialized neuroendocrine command systems intercalated within the hypothalamus control the most fundamental physiological needs in vertebrates1,2. Nevertheless, we lack a developmental blueprint that integrates the molecular determinants of neuronal and glial diversity along temporal and spatial scales of hypothalamus development3. Here we combine single-cell RNA sequencing of 51,199 mouse cells of ectodermal origin, gene regulatory network (GRN) screens in conjunction with genome-wide association study-based disease phenotyping, and genetic lineage reconstruction to show that nine glial and thirty-three neuronal subtypes are generated by mid-gestation under the control of distinct GRNs. Combinatorial molecular codes that arise from neurotransmitters, neuropeptides and transcription factors are minimally required to decode the taxonomical hierarchy of hypothalamic neurons. The differentiation of γ-aminobutyric acid (GABA) and dopamine neurons, but not glutamate neurons, relies on quasi-stable intermediate states, with a pool of GABA progenitors giving rise to dopamine cells4. We found an unexpected abundance of chemotropic proliferation and guidance cues that are commonly implicated in dorsal (cortical) patterning5 in the hypothalamus. In particular, loss of SLIT–ROBO signalling impaired both the production and positioning of periventricular dopamine neurons. Overall, we identify molecular principles that shape the developmental architecture of the hypothalamus and show how neuronal heterogeneity is transformed into a multimodal neural unit to provide virtually infinite adaptive potential throughout life.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Raw, processed and supplementary datasets have been deposited in GEO (accession number: GSE132730). GEO files include: 1) raw fastq files for every sequencing run; 2) filtered matrices for every sample in RDS file format including Seurat 3 objects with all processed cells; 3) original integrated dataset in RDS file format including Seurat 3 objects with all processed cells as well as all used commands; 4) integrated dataset used for dynamics analysis (which passed filtering of RNA velocity analysis); 5) AUCell matrices from pySCENIC pipeline; 6) full regulon hypothalamic network in GraphML file format; 7) metadata protocol describing all experimental, computational procedures and quality control. An interactive view of the integrated dataset (for processing in Pagoda2) can be accessed at https://doi.org/10.6084/m9.figshare.11867889 (~1.1 GB). All data presented (for example, imaging) will be made available by T. Harkany (tibor.harkany@ki.se or tibor.harkany@meduniwien.ac.at) upon reasonable request.
Code availability
The code used is available at https://doi.org/10.6084/m9.figshare.11867889.
References
Saper, C. B. & Lowell, B. B. The hypothalamus. Curr. Biol. 24, R1111–R1116 (2014).
Dulac, C., O’Connell, L. A. & Wu, Z. Neural control of maternal and paternal behaviors. Science 345, 765–770 (2014).
Xie, Y. & Dorsky, R. I. Development of the hypothalamus: conservation, modification and innovation. Development 144, 1588–1599 (2017).
Romanov, R. A. et al. Molecular interrogation of hypothalamic organization reveals distinct dopamine neuronal subtypes. Nat. Neurosci. 20, 176–188 (2017).
Andrews, W. et al. The role of Slit-Robo signaling in the generation, migration and morphological differentiation of cortical interneurons. Dev. Biol. 313, 648–658 (2008).
Mayer, C. et al. Developmental diversification of cortical inhibitory interneurons. Nature 555, 457–462 (2018).
Rakic, P. Evolution of the neocortex: a perspective from developmental biology. Nat. Rev. Neurosci. 10, 724–735 (2009).
Zeisel, A. et al. Molecular architecture of the mouse nervous system. Cell 174, 999–1014.e1022 (2018).
Campbell, J. N. et al. A molecular census of arcuate hypothalamus and median eminence cell types. Nat. Neurosci. 20, 484–496 (2017).
Toda, C., Santoro, A., Kim, J. D. & Diano, S. POMC neurons: from birth to death. Annu. Rev. Physiol. 79, 209–236 (2017).
Burbridge, S., Stewart, I. & Placzek, M. Development of the neuroendocrine hypothalamus. Compr. Physiol. 6, 623–643 (2016).
Wolf, F. A. et al. PAGA: graph abstraction reconciles clustering with trajectory inference through a topology preserving map of single cells. Genome Biol. 20, 59 (2019).
La Manno, G. et al. RNA velocity of single cells. Nature 560, 494–498 (2018).
Aujla, P. K., Naratadam, G. T., Xu, L. & Raetzman, L. T. Notch/Rbpjκ signaling regulates progenitor maintenance and differentiation of hypothalamic arcuate neurons. Development 140, 3511–3521 (2013).
McNay, D. E., Pelling, M., Claxton, S., Guillemot, F. & Ang, S.-L. Mash1 is required for generic and subtype differentiation of hypothalamic neuroendocrine cells. Mol. Endocrinol. 20, 1623–1632 (2006).
De Marco García, N. V., Karayannis, T. & Fishell, G. Neuronal activity is required for the development of specific cortical interneuron subtypes. Nature 472, 351–355 (2011).
Cobos, I., Borello, U. & Rubenstein, J. L. Dlx transcription factors promote migration through repression of axon and dendrite growth. Neuron 54, 873–888 (2007).
Wamsley, B. & Fishell, G. Genetic and activity-dependent mechanisms underlying interneuron diversity. Nat. Rev. Neurosci. 18, 299–309 (2017).
Gerstner, J. R. & Landry, C. F. Expression of the transcriptional coactivator CITED1 in the adult and developing murine brain. Dev. Neurosci. 29, 203–212 (2007).
Hökfelt, T., Meister, B., Melander, T. & Everitt, B. Coexistence of classical transmitters and peptides with special reference to the arcuate nucleus–median eminence complex. Adv. Biochem. Psychopharmacol. 43, 21–34 (1987).
Motoike, T. et al. Transient expression of neuropeptide W in postnatal mouse hypothalamus—a putative regulator of energy homeostasis. Neuroscience 301, 323–337 (2015).
Maejima, Y. et al. Oxytocinergic circuit from paraventricular and supraoptic nuclei to arcuate POMC neurons in hypothalamus. FEBS Lett. 588, 4404–4412 (2014).
Aibar, S. et al. SCENIC: single-cell regulatory network inference and clustering. Nat. Methods 14, 1083–1086 (2017).
Boyle, E. A., Li, Y. I. & Pritchard, J. K. An expanded view of complex traits: from polygenic to omnigenic. Cell 169, 1177–1186 (2017).
Popadin, K. et al. Slightly deleterious genomic variants and transcriptome perturbations in Down syndrome embryonic selection. Genome Res. 28, 1–10 (2018).
Cardenas, A. et al. Evolution of cortical neurogenesis in amniotes controlled by Robo signaling levels. Cell 174, 590–606.e521 (2018).
Chédotal, A. & Richards, L. J. Wiring the brain: the biology of neuronal guidance. Cold Spring Harb. Perspect. Biol. 2, a001917 (2010).
Arenas, E., Denham, M. & Villaescusa, J. C. How to make a midbrain dopaminergic neuron. Development 142, 1918–1936 (2015).
Tritsch, N. X., Ding, J. B. & Sabatini, B. L. Dopaminergic neurons inhibit striatal output through non-canonical release of GABA. Nature 490, 262–266 (2012).
Matsushita, N. et al. Dynamics of tyrosine hydroxylase promoter activity during midbrain dopaminergic neuron development. J. Neurochem. 82, 295–304 (2002).
Cowley, M. A. et al. Leptin activates anorexigenic POMC neurons through a neural network in the arcuate nucleus. Nature 411, 480–484 (2001).
Kim, E. J., Ables, J. L., Dickel, L. K., Eisch, A. J. & Johnson, J. E. Ascl1 (Mash1) defines cells with long-term neurogenic potential in subgranular and subventricular zones in adult mouse brain. PLoS One 6, e18472 (2011).
Madisen, L. et al. A robust and high-throughput Cre reporting and characterization system for the whole mouse brain. Nat. Neurosci. 13, 133–140 (2010).
Shu, T., Butz, K. G., Plachez, C., Gronostajski, R. M. & Richards, L. J. Abnormal development of forebrain midline glia and commissural projections in Nfia knock-out mice. J. Neurosci. 23, 203–212 (2003).
Bali, B., Erdélyi, F., Szabó, G. & Kovács, K. J. Visualization of stress-responsive inhibitory circuits in the GAD65-eGFP transgenic mice. Neurosci. Lett. 380, 60–65 (2005).
Tamamaki, N. et al. Green fluorescent protein expression and colocalization with calretinin, parvalbumin, and somatostatin in the GAD67-GFP knock-in mouse. J. Comp. Neurol. 467, 60–79 (2003).
Andrews, W. et al. Robo1 regulates the development of major axon tracts and interneuron migration in the forebrain. Development 133, 2243–2252 (2006).
Plump, A. S. et al. Slit1 and Slit2 cooperate to prevent premature midline crossing of retinal axons in the mouse visual system. Neuron 33, 219–232 (2002).
Yang, L. et al. Isl1Cre reveals a common Bmp pathway in heart and limb development. Development 133, 1575–1585 (2006).
Yoshida, M. et al. Evidence that oxytocin exerts anxiolytic effects via oxytocin receptor expressed in serotonergic neurons in mice. J. Neurosci. 29, 2259–2271 (2009).
Bäckman, C. M. et al. Characterization of a mouse strain expressing Cre recombinase from the 3′ untranslated region of the dopamine transporter locus. Genesis 44, 383–390 (2006).
Macosko, E. Z. et al. Highly parallel genome-wide expression profiling of individual cells using nanoliter droplets. Cell 161, 1202–1214 (2015).
Lun, A. T. L. et al. EmptyDrops: distinguishing cells from empty droplets in droplet-based single-cell RNA sequencing data. Genome Biol. 20, 63 (2019).
Petukhov, V. et al. dropEst: pipeline for accurate estimation of molecular counts in droplet-based single-cell RNA-seq experiments. Genome Biol. 19, 78 (2018).
van den Brink, S. C. et al. Single-cell sequencing reveals dissociation-induced gene expression in tissue subpopulations. Nat. Methods 14, 935–936 (2017).
Hinman, M. N. & Lou, H. Diverse molecular functions of Hu proteins. Cell. Mol. Life Sci. 65, 3168–3181 (2008).
Mili, S. & Steitz, J. A. Evidence for reassociation of RNA-binding proteins after cell lysis: implications for the interpretation of immunoprecipitation analyses. RNA 10, 1692–1694 (2004).
Yoo, S. & Blackshaw, S. Regulation and function of neurogenesis in the adult mammalian hypothalamus. Prog. Neurobiol. 170, 53–66 (2018).
Miranda-Angulo, A. L., Byerly, M. S., Mesa, J., Wang, H. & Blackshaw, S. Rax regulates hypothalamic tanycyte differentiation and barrier function in mice. J. Comp. Neurol. 522, 876–899 (2014).
Shimogori, T. et al. A genomic atlas of mouse hypothalamic development. Nat. Neurosci. 13, 767–775 (2010).
Hafemeister, C. & Satija, R. Normalization and variance stabilization of single-cell RNA-seq data using regularized negative binomial regression. Genome Biol. 20, 296 (2019).
Finak, G. et al. MAST: a flexible statistical framework for assessing transcriptional changes and characterizing heterogeneity in single-cell RNA sequencing data. Genome Biol. 16, 278 (2015).
Zeisel, A. et al. Brain structure. Cell types in the mouse cortex and hippocampus revealed by single-cell RNA-seq. Science 347, 1138–1142 (2015).
Alpár, A., Benevento, M., Romanov, R. A., Hökfelt, T. & Harkany, T. Hypothalamic cell diversity: non-neuronal codes for long-distance volume transmission by neuropeptides. Curr. Opin. Neurobiol. 56, 16–23 (2019).
Romanov, R. A., Alpár, A., Hökfelt, T. & Harkany, T. Unified classification of molecular, network, and endocrine features of hypothalamic neurons. Annu. Rev. Neurosci. 42, 1–26 (2019).
Polański, K. et al. BBKNN: fast batch alignment of single cell transcriptomes. Bioinformatics 36, 964–965 (2020).
McInnes, L., Healy, J., Saul, N. & Großberger, L. UMAP: uniform manifold approximation and projection. JOSS 3, 861 (2018).
Johnson, J., Douze, M. & Jegou, H. Billion-scale similarity search with GPUs. IEEE Trans. Big Data https://doi.org/10.1109/TBDATA.2019.2921572 (2019).
Maneewongvatana, S. & Mount, D. in Data Structures, Near Neighbor Searches, and Methodology: Fifth and Sixth DIMACS Implementation Challenges Vol. 59 DIMACS Series in Discrete Mathematics and Theoretical Computer Science (eds. Goldwasser, M., Johnson, D. & McGeoch, C.) 105–123 (American Mathematical Society, 2002).
Hie, B., Bryson, B. & Berger, B. Efficient integration of heterogeneous single-cell transcriptomes using Scanorama. Nat. Biotechnol. 37, 685–691 (2019).
Welch, J. D. et al. Single-cell multi-omic integration compares and contrasts features of brain cell identity. Cell 177, 1873–1887.e1817 (2019).
Korsunsky, I. et al. Fast, sensitive, and accurate integration of single cell data with harmony. Nature Methods 16, 1289–1296 (2019).
Haghverdi, L., Lun, A. T. L., Morgan, M. D. & Marioni, J. C. Batch effects in single-cell RNA-sequencing data are corrected by matching mutual nearest neighbors. Nat. Biotechnol. 36, 421–427 (2018).
Barkas, N. et al. Joint analysis of heterogeneous single-cell RNA-seq dataset collections. Nat. Methods 16, 695–698 (2019).
Stuart, T. et al. Comprehensive integration of single-cell data. Cell 177, 1888–1902.e1821 (2019).
Kowalczyk, M. S. et al. Single-cell RNA-seq reveals changes in cell cycle and differentiation programs upon aging of hematopoietic stem cells. Genome Res. 25, 1860–1872 (2015).
Becht, E. et al. Dimensionality reduction for visualizing single-cell data using UMAP. Nat. Biotechnol. 37, 38–44 (2019).
Crow, M., Paul, A., Ballouz, S., Huang, Z. J. & Gillis, J. Characterizing the replicability of cell types defined by single cell RNA-sequencing data using MetaNeighbor. Nat. Commun. 9, 884 (2018).
Chen, R., Wu, X., Jiang, L. & Zhang, Y. Single-cell RNA-seq reveals hypothalamic cell diversity. Cell Rep. 18, 3227–3241 (2017). https://doi.org/10.1016/j.celrep.2017.03.004.
Moffitt, J. R. et al. Molecular, spatial, and functional single-cell profiling of the hypothalamic preoptic region. Science 362, eaau5324 (2018).
Vinh, N. X., Epps, J. & Bailey, J. Information theoretic measures for clusterings comparison: variants, properties, normalization and correction for chance. J. Mach. Learn. Res. 11, 2837–2854 (2010).
Chang, F., Qiu, W., Zamar, R. H., Lazarus, R. & Wang, X. clues: an r package for nonparametric clustering based on local shrinking. J. Stat. Softw. 33, (2010).
Pons, P. & Latapy, M. in Intl Symp. Computer Information Sci. 284–293 (Springer, 2005).
Kurrasch, D. M. et al. The neonatal ventromedial hypothalamus transcriptome reveals novel markers with spatially distinct patterning. J. Neurosci. 27, 13624–13634 (2007).
Zhang, Y. et al. Purification and characterization of progenitor and mature human astrocytes reveals transcriptional and functional differences with mouse. Neuron 89, 37–53 (2016).
Kelley, K. W., Nakao-Inoue, H., Molofsky, A. V. & Oldham, M. C. Variation among intact tissue samples reveals the core transcriptional features of human CNS cell classes. Nat. Neurosci. 21, 1171–1184 (2018).
Lin, Y. et al. Evaluating stably expressed genes in single cells. Gigascience 8, giz106 (2019).
Extra Coordinate Systems. ‘Geoms’, Statistical Transformations, Scales and Fonts for 'ggplot2' [R package ggalt version 0.4.0] v. 0.4.0 https://CRAN.R-project.org/package=ggalt (2017).
Traag, V. A., Waltman, L. & van Eck, N. J. From Louvain to Leiden: guaranteeing well-connected communities. Sci. Rep. 9, 5233 (2019).
Zhang, J. M., Kamath, G. M. & Tse, D. N. Valid post-clustering differential analysis for single-cell RNA-seq. Cell Syst. 9, 383–392.e6 (2019).
Wolf, F. A., Angerer, P. & Theis, F. J. SCANPY: large-scale single-cell gene expression data analysis. Genome Biol. 19, 15 (2018).
Bergen, V., Lange, M., Peidli, S., Wolf, F. A. & Theis, F. J. Generalizing RNA velocity to transient cell states through dynamical modeling. Preprint at https://www.biorxiv.org/content/10.1101/820936v1 (2019).
Moerman, T. et al. GRNBoost2 and Arboreto: efficient and scalable inference of gene regulatory networks. Bioinformatics 35, 2159–2161 (2019).
Kanamori, M. et al. A genome-wide and nonredundant mouse transcription factor database. Biochem. Biophys. Res. Commun. 322, 787–793 (2004).
Ravasi, T. et al. An atlas of combinatorial transcriptional regulation in mouse and man. Cell 140, 744–752 (2010).
Forrest, A. R. et al. A promoter-level mammalian expression atlas. Nature 507, 462–470 (2014).
Ntranos, V., Yi, L., Melsted, P. & Pachter, L. Identification of transcriptional signatures for cell types from single-cell RNA-seq. Nat. Methods 16, 163–166 (2019).
Bastian, M., Heymann, S. & Jacomy, M. Gephi: An Open Source Software for Exploring and Manipulating Networks https://gephi.org/publications/gephi-bastian-feb09.pdf (2009).
Canela-Xandri, O., Rawlik, K. & Tenesa, A. An atlas of genetic associations in UK Biobank. Nat. Genet. 50, 1593–1599 (2018).
Storey, J. D. & Tibshirani, R. Statistical significance for genomewide studies. Proc. Natl Acad. Sci. USA 100, 9440–9445 (2003).
qvalue: Q-value estimation for false discovery rate control v. 2.18.0 (R package, 2019).
Galili, T., O’Callaghan, A., Sidi, J. & Sievert, C. heatmaply: an R package for creating interactive cluster heatmaps for online publishing. Bioinformatics 34, 1600–1602 (2018).
Setty, M. et al. Characterization of cell fate probabilities in single-cell data with Palantir. Nat. Biotechnol. 37, 451–460 (2019).
Levine, J. H. et al. Data-driven phenotypic dissection of AML reveals progenitor-like cells that correlate with prognosis. Cell 162, 184–197 (2015).
Espana, A. & Clotman, F. Onecut transcription factors are required for the second phase of development of the A13 dopaminergic nucleus in the mouse. J. Comp. Neurol. 520, 1424–1441 (2012).
Miyazaki, T., Fukaya, M., Shimizu, H. & Watanabe, M. Subtype switching of vesicular glutamate transporters at parallel fibre-Purkinje cell synapses in developing mouse cerebellum. Eur. J. Neurosci. 17, 2563–2572 (2003).
Bartesaghi, L. et al. PRDM12 is required for initiation of the nociceptive neuron lineage during neurogenesis. Cell Rep. 26, 3484–3492.e4 (2019).
Wang, F. et al. RNAscope: a novel in situ RNA analysis platform for formalin-fixed, paraffin-embedded tissues. J. Mol. Diagn. 14, 22–29 (2012).
Choi, H. M. T. et al. Mapping a multiplexed zoo of mRNA expression. Development 143, 3632–3637 (2016).
Zhao, S. et al. Cell type–specific channelrhodopsin-2 transgenic mice for optogenetic dissection of neural circuitry function. Nat. Methods 8, 745–752 (2011).
Acknowledgements
A. Reinthaler is acknowledged for her expert laboratory assistance. We thank the Biomedical Sequencing Facility at the CeMM Research Center for Molecular Medicine of the Austrian Academy of Sciences for assistance with next-generation sequencing, G. A. Bazykin for providing access to the 2TB RAM Makarich computational cluster for OMICS data analysis, E. Porcu for advice on GWAS analysis, M. Watanabe for antibodies, and G. Szabó, F. Erdélyi, J. Bunt, L. J. Richards and Y. Yanagawa for transgenic mice. This work was supported by the Swedish Research Council (F.L., I.A., T. Hökfelt, T. Harkany); Novo Nordisk Foundation (T. Hökfelt, T. Harkany); Bertil Hållsten Research Foundation (I.A.); Hjärnfonden (T. Harkany), European Research Council (STEMMING-FROM-NERVE, 2014-CoG-647844; I.A. and SECRET-CELLS, 2015-AdG-695136; T. Harkany), the EMBO Young Investigator Program (I.A.), Åke Wiberg Foundation (I.A.), Wallenberg Academy fellowship (F.L.), a Ming Wai Lau Center investigator grant (F.L.), the Strategic Research program for Brain Sciences (AMED, Japan; K.N.), Fonds spéciaux de recherche of the Université catholique de Louvain (F.C.), Actions de Recherche Concertées (17/22-079) of the Direction générale de l’Enseignement non obligatoire et de la Recherche scientifique–Direction de la Recherche scientifique–Communauté française de Belgique and granted by the Académie universitaire ‘Louvain’ (F.C.), 5 Top 100 Russian Academic Excellence Project at the Immanuel Kant Federal Baltic University and Russian Foundation for Basic Research (project 18-29-13055, K.P.) and intramural funds of the Medical University of Vienna (T. Harkany). M.F. is supported by a special research program of the Austrian Science Fund (FWF-F61). R.A.R. is an EMBO advanced research fellow (ALTF 493-2017). E.O.T. is supported by a scholarship from the Austrian Science Fund (FWF, DOC 33-B27). F.C. is a senior research associate of the F.R.S.-FNRS.
Author information
Authors and Affiliations
Contributions
T. Harkany and R.A.R. conceived the project; K.N., I.A., T. Hökfelt and T. Harkany procured funding; R.A.R., I.A., M.F., C.B., T. Hökfelt, E.K. and T. Harkany designed experiments; R.A.R., E.O.T., M.E.K., M.Z., M.H., S.K., K.P., M.B., P.R. and M.F. performed experiments and analysed data; F.L., K.N., F.C., W.D.A. and J.G.P. provided unique reagents and mouse models. R.A.R. and T. Harkany wrote the manuscript with input from all co-authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature thanks Nenad Sestan and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Marker genes to define molecular phenotypes.
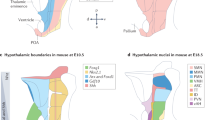
a, Differential gene expression by glia (clusters 1–9) and neurons (clusters 10–45). Because of the integration of six stages, early-expressed TFs and spatially restricted genes amenable to cellular differentiation were identified. For neuronal clusters, fast neurotransmitter specificity is shown to the right. The relative diameter of the solid circles for each cluster is scaled to the fraction of cells that expresses a specific gene. Colour coding and numbering at the top correspond to those in Fig. 1a. b, Top, Differential TF expression in 45 ectoderm-derived cell groups in the hypothalamus. Bottom, subclass-specific TFs recapitulate the UMAP positions of neuronal (left) and glial (right) subtypes. c, Integrated molecular and anatomical annotation of hypothalamic clusters with their specific assignment to hypothalamic areas. ARC-Agrp, arcuate nucleus agouti-related peptide+ neurons; ARC-Sst, arcuate nucleus somatostatin+ neurons; ARC-TIDA, arcuate nucleus–tuberoinfundibular dopamine neurons; Gal, galanin; Ghrh/Vacht, growth hormone-releasing hormone/vesicular acetylcholine transporter+ neurons; LH, lateral hypothalamus; LH-Lhx9, lateral hypothalamus LIM homeobox 9+ cluster; Meis2, meis homeobox 2; MM, mammillary nucleus; MM-Lhx9, mammillary nucleus LIM homeobox 9+ neurons; Pomc, proopiomelanocortin; PH, posterior hypothalamus; PMM, premamillary nucleus; Tbr1, T-box brain transcription factor 1.
Extended Data Fig. 2 Molecular analysis of TFs involved in neurogenesis and neuronal differentiation.
a, Comparative and time-resolved analysis of the cell bridge by MNN, CONOS and Seurat alignment. In UMAP space on separate developmental stages, MNN, CONOS and Seurat algorithms were compared for their ability to specifically resolve the transition of progenitors to immature cells (bridge). Colour codes correspond to those in Fig. 1a. b, RNA velocity at E15.5, E17.5 and P0. Colour codes are consistent with those in Fig. 1a. Note the presence of a bridge (grey background) between progenitor/glial and neuronal compartments at early developmental stages with its rupture being evident by birth. c, Gene expression in UMAP space at E15.5. Note a central role for Notch signalling in neurogenesis. d, Genetic tracing of Ascl1+ cells produced in the developing hypothalamus during the E12.5–E16.5 period. e, In situ hybridization showing the distribution of Tbr1 and Eomes. Image credit: Allen Institute (https://www.brain-map.org). f, Genetic tracing of Ascl1+ cells in the developing hypothalamus of Ascl+/− and Ascl−/− mice. Sox2 was localized by immunohistochemistry. g, Sox2, Ascl1 (Tomato) and Rbfox3 (NeuN) immunolocalization at successive developmental stages. h, Genetic tracing of Ascl+ cells postnatally (as in Fig. 1f). Scale bars, 200 μm (d), 20 μm (f–h).
Extended Data Fig. 3 Neurotransmitter and neuropeptide specificity and load in the developing hypothalamus.
a–c, Coincident profiling of fast neurotransmitters (a), neuropeptides (b) and neuropeptide receptors (c, top) in 45 cell groups of ectodermal origin. c, Bottom left, given their abundance, Ntrk2 and Adcyap1r1 were plotted separately along the developmental timeline studied with appropriate scaling. Bottom right, likewise, the distribution of both receptors per cell cluster was mapped and scaled separately. d, Coincident profiling of neuropeptides in neuronal clusters distinguished as GABA (blue) and glutamate (grey) phenotypes. e, Map of Th expression in GABA and glutamate neurons. Colour coding as in d. f, Developmental mapping of hypothalamic Oxtr expression in OxtrVenus/+ mice. Low-magnification image surveys are shown (see also Fig. 2e). Scale bars, 200 μm (f). Data shown as dot plots and scaled as previously described6,51,65.
Extended Data Fig. 4 Hierarchical relationship of GRNs (regulons).
An AUC separability plot was used to assign regulons that determine cell cluster identities identified in SCENIC23. GRNs were reconstructed individually for each cell and then assigned as ‘regulon representation’ (Logreg test) to each cell group. TFs to the left are representative for each regulon. Marked dendrogram branchpoints were estimated by both the Wilcoxon and Logreg tests (see also https://doi.org/10.6084/m9.figshare.11867889).
Extended Data Fig. 5 Relationships between regulons and disease phenotypes in humans.
a, Complete heat map of associations between regulon activity and clinical disease phenotype. Left: classifications of diseases as per phenotypic criteria of the UK biobank registry (https://www.ukbiobank.ac.uk). Top, master genes for each regulon. Genes presented in Fig. 3 are in red and highlighted in b. Colour coding from deep blue to bright yellow shows increasing correlation probability. b, Scatter plot reflecting the ratios of mutability in master genes versus all downstream target genes per regulon. Mutability and the constrains of TFs were expressed as the total number of mutations. Colours represent four quadrants that were separated on the basis of the total number of mutations per master gene (medians, y-axis) versus target genes (medians, x-axis). Horizontal line corresponds to the median of SNPs in all genes. Dot size reflects the median influence of a given regulon on its targets as per SCENIC output.
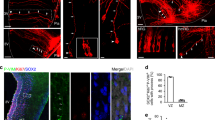
Extended Data Fig. 6 Molecular complexity and function of the Onecut3 regulon.
a, Interlinked Onecut2 and Onecut3 regulons in hypothalamic neurons. Genes that were biologically validated (see below) are shown in black. b, Co-expression of Onecut2 and Onecut3 along the rostrocaudal axis of the hypothalamus. c, Co-localization of Onecut3 and its target genes (from a). d, Overexpression of Onecut3 (OC3) and ATP-binding cassette D2 (Abcd2, to control promoter activity) in Neuro2A cells. Left, representative images by multiple fluorescence labelling-differential interference microscopy. Right, quantification of Hoechst+ and phospho-histone H3 (pHH3)+ Neuro2A cells revealed significantly reduced proliferation upon Onecut3 overexpression. No significant cell death was induced by either overexpressed plasmid or the transfection reagent alone. e, qPCR analysis of genes regulated by Onecut3: Cxxc5, Tmprss9 and Th. All data were normalized to samples transfected with Abcd2, which were taken as technical controls. Scale bars, 50 μm (d), 20 μm (b, f), 10 μm (g).
Extended Data Fig. 7 Experimental validation of ventricle-restricted genes identified by scRNA-seq.
a, Left, expressional dynamics of ventricle-associated marker genes: Slc1a3, Rax and Dll3 on UMAP embedding (top) and trend lines (bottom). Right, validation by in situ hybridization. b, In situ hybridization for the co-existence of Slit2 and Rax in ventricular progenitors and consequential medial-to-lateral Slit1–Dll1–Dll3 patterns during neuronal differentiation and migration by E15.5 (left, top right). Left-to-right orientation corresponds to medial-to-lateral hypothalamic positions. Bottom right, localization of Slit1 and Slit2 mRNAs in the VMH at E18.5. Scale bars, 200 μm (a), 20 μm (b).
Extended Data Fig. 8 Physiological and morphological subtypes of hypothalamic dopamine neurons.
a, Action potential waveforms of dopamine neurons within the A12–A14 groups. Note the diversification of A14 dopamine cells into subgroups A–D with clearly different action potential signatures. Morphological reconstruction of biocytin-filled neurons is shown with each group. b, Distribution of tdTomato+ neurons in the hypothalamus of Slc6a3-Ires-cre::Ai14 mice. Scale bars, 50 μm (b), 20 μm (a).
Extended Data Fig. 9 Transcriptional and physiological features of dopamine neurons in the developing hypothalamus.
a, Ascl1-creERT2/+::Ai14 (control) versus Ascl1-creERT2/ERT2::Ai14 mice (a knock-in mouse line with Cre disrupting the Ascl1 gene, referred to as Ascl1 ko), injected with tamoxifen at E11.5 and analysed at E13.5. Note the accumulation of tdTomato+ cells in the KO relative to controls. b, Genetic tracing in Ascl1-creERT2::Ai14 reporter mice identified Ascl1+/Th+ neurons within the preoptic and periventricular nuclei. Meanwhile, Ascl1−/Th+ neurons populated the Arc and zona incerta (ZI) by E18.5. c, Isl1 and Meis2 transcriptional trends of differentiation for trajectories in Th+ groups (clusters 1–9). Amplitudes are shown in log10 scale. Line shading corresponds to mean ± s.e.m. d, Genetic lineage tracing using Isl1-cre::Ai14 mice. e, In situ hybridization for Gad1 and Th revealed anti-parallel expressional load for these genes as a factor of medial-to-lateral positioning. Scatter plots show the number of fluorescent puncta per cell (threshold >2). f, In situ hybridization for Meis2, Th and Ddc in the hypothalami of E18.5 and P2 mice. Scale bars, 120 μm (a, f (left)), 12 μm (b, d–f (right)).
Extended Data Fig. 10 GABA origin of hypothalamic dopamine neurons.
a, Immunohistochemical analysis of TH and ONECUT3 protein expression in the hypothalamus of (BAC)GAD65–eGFP and GAD67–GFP mice at the developmental time-points indicated. Note a gradual GABA-to-dopamine transition as a factor of advancing age with ONECUT3 expression preceding that of TH. Dashed rectangles denote the positions of high-resolution insets. b, Expression patterns of regulon-forming TFs that directly drive Th transcription in the developing hypothalamus. Meis2, Pbx3 and Dlx1 were visualized on UMAP embedding for neuronal lineages. c, Histochemical localization of the migratory route of prospective PeVN dopamine neurons (cluster 9) through the coincident localization of TH and ONECUT3 during embryonic development. Dashed lines denote the ventricular surface. d, Localization of Onecut2 and Pmfbp1a target genes within the Onecut3 regulon to PeVN dopamine neurons by a combination of immunohistochemistry and in situ hybridization. e, Sst expression in PeVN dopamine neurons. f, Post hoc reconstruction of A14 Onecut3+ dopamine neurons after patch-clamp recordings. Scale bars, 200 μm (a, overviews), 50 μm (a, insets), 20 μm (f), 12 μm (c–e).
Supplementary information
Supplementary Information
This file contains Supplementary Sections 1-3. Section 1 contains integrated data analysis - quality control, data filtration, integration and clustering, and includes Supplementary Figures S1-S11. Section 2 includes identification of dopamine cell groups and reconstruction of their developmental trajectories, including Supplementary Figures S12 and S13. Section 3 contains additional remarks to the source data on Figshare.
Rights and permissions
About this article
Cite this article
Romanov, R.A., Tretiakov, E.O., Kastriti, M.E. et al. Molecular design of hypothalamus development. Nature 582, 246–252 (2020). https://doi.org/10.1038/s41586-020-2266-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-020-2266-0
This article is cited by
-
Maternal dietary fat during lactation shapes single nucleus transcriptomic profile of postnatal offspring hypothalamus in a sexually dimorphic manner in mice
Nature Communications (2024)
-
Reciprocal activity of AgRP and POMC neurons governs coordinated control of feeding and metabolism
Nature Metabolism (2024)
-
Transient expression of the neuropeptide galanin modulates peripheral‑to‑central connectivity in the somatosensory thalamus during whisker development in mice
Nature Communications (2024)
-
Drug targeting in psychiatric disorders — how to overcome the loss in translation?
Nature Reviews Drug Discovery (2024)
-
Single cell tracing of Pomc neurons reveals recruitment of ‘Ghost’ subtypes with atypical identity in a mouse model of obesity
Nature Communications (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.