Abstract
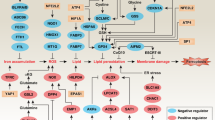
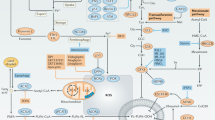
Ferroptosis, a cell death process driven by cellular metabolism and iron-dependent lipid peroxidation, has been implicated in diseases such as ischaemic organ damage and cancer1,2. The enzyme glutathione peroxidase 4 (GPX4) is a central regulator of ferroptosis, and protects cells by neutralizing lipid peroxides, which are by-products of cellular metabolism. The direct inhibition of GPX4, or indirect inhibition by depletion of its substrate glutathione or the building blocks of glutathione (such as cysteine), can trigger ferroptosis3. Ferroptosis contributes to the antitumour function of several tumour suppressors such as p53, BAP1 and fumarase4,5,6,7. Counterintuitively, mesenchymal cancer cells—which are prone to metastasis, and often resistant to various treatments—are highly susceptible to ferroptosis8,9. Here we show that ferroptosis can be regulated non-cell-autonomously by cadherin-mediated intercellular interactions. In epithelial cells, such interactions mediated by E-cadherin suppress ferroptosis by activating the intracellular NF2 (also known as merlin) and Hippo signalling pathway. Antagonizing this signalling axis allows the proto-oncogenic transcriptional co-activator YAP to promote ferroptosis by upregulating several ferroptosis modulators, including ACSL4 and TFRC. This finding provides mechanistic insights into the observations that cancer cells with mesenchymal or metastatic property are highly sensitive to ferroptosis8. Notably, a similar mechanism also modulates ferroptosis in some non-epithelial cells. Finally, genetic inactivation of the tumour suppressor NF2, a frequent tumorigenic event in mesothelioma10,11, rendered cancer cells more sensitive to ferroptosis in an orthotopic mouse model of malignant mesothelioma. Our results demonstrate the role of intercellular interactions and intracellular NF2–YAP signalling in dictating ferroptotic death, and also suggest that malignant mutations in NF2–YAP signalling could predict the responsiveness of cancer cells to future ferroptosis-inducing therapies.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
For western blot source data, see Supplementary Fig. 1. For the gating strategy used for flow cytometry experiments, see Supplementary Fig. 2. Raw data for all experiments are available as Source Data to the relevant figures. ChIP–seq datasets analysed in this article are publicly available in the ENCODE database under the identifiers GSM1010875 and GSM1010868.
Change history
02 August 2019
An Amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Stockwell, B. R. et al. Ferroptosis: a regulated cell death nexus linking metabolism, redox biology, and disease. Cell 171, 273–285 (2017).
Gao, M. & Jiang, X. To eat or not to eat—the metabolic flavor of ferroptosis. Curr. Opin. Cell Biol. 51, 58–64 (2018).
Yang, W. S. et al. Regulation of ferroptotic cancer cell death by GPX4. Cell 156, 317–331 (2014).
Gao, M. et al. Role of mitochondria in ferroptosis. Mol. Cell 73, 354–363.e3 (2019).
Jiang, L. et al. Ferroptosis as a p53-mediated activity during tumour suppression. Nature 520, 57–62 (2015).
Jennis, M. et al. An African-specific polymorphism in the TP53 gene impairs p53 tumor suppressor function in a mouse model. Genes Dev. 30, 918–930 (2016).
Zhang, Y. et al. BAP1 links metabolic regulation of ferroptosis to tumour suppression. Nat. Cell Biol. 20, 1181–1192 (2018).
Viswanathan, V. S. et al. Dependency of a therapy-resistant state of cancer cells on a lipid peroxidase pathway. Nature 547, 453–457 (2017).
Hangauer, M. J. et al. Drug-tolerant persister cancer cells are vulnerable to GPX4 inhibition. Nature 551, 247–250 (2017).
Hmeljak, J. et al. Integrative molecular characterization of malignant pleural mesothelioma. Cancer Discov. 8, 1548–1565 (2018).
Bueno, R. et al. Comprehensive genomic analysis of malignant pleural mesothelioma identifies recurrent mutations, gene fusions and splicing alterations. Nat. Genet. 48, 407–416 (2016).
Seiler, A. et al. Glutathione peroxidase 4 senses and translates oxidative stress into 12/15-lipoxygenase dependent- and AIF-mediated cell death. Cell Metab. 8, 237–248 (2008).
Schneider, M. et al. Absence of glutathione peroxidase 4 affects tumor angiogenesis through increased 12/15-lipoxygenase activity. Neoplasia 12, 254–263 (2010).
Gao, M., Monian, P., Quadri, N., Ramasamy, R. & Jiang, X. Glutaminolysis and transferrin regulate ferroptosis. Mol. Cell 59, 298–308 (2015).
van Roy, F. & Berx, G. The cell–cell adhesion molecule E-cadherin. Cell. Mol. Life Sci. 65, 3756–3788 (2008).
Kim, N. G., Koh, E., Chen, X. & Gumbiner, B. M. E-cadherin mediates contact inhibition of proliferation through Hippo signaling-pathway components. Proc. Natl Acad. Sci. USA 108, 11930–11935 (2011).
Okada, T., Lopez-Lago, M. & Giancotti, F. G. Merlin/NF-2 mediates contact inhibition of growth by suppressing recruitment of Rac to the plasma membrane. J. Cell Biol. 171, 361–371 (2005).
Zhao, B., Lei, Q. Y. & Guan, K. L. The Hippo–YAP pathway: new connections between regulation of n size and cancer. Curr. Opin. Cell Biol. 20, 638–646 (2008).
Pan, D. The Hippo signaling pathway in development and cancer. Dev. Cell 19, 491–505 (2010).
Li, W., Cooper, J., Karajannis, M. A. & Giancotti, F. G. Merlin: a tumour suppressor with functions at the cell cortex and in the nucleus. EMBO Rep. 13, 204–215 (2012).
Li, W. et al. Merlin/NF2 loss-driven tumorigenesis linked to CRL4(DCAF1)-mediated inhibition of the hippo pathway kinases Lats1 and 2 in the nucleus. Cancer Cell 26, 48–60 (2014).
Dupont, S. et al. Role of YAP/TAZ in mechanotransduction. Nature 474, 179–183 (2011).
Falk, M. H. et al. Apoptosis in Burkitt lymphoma cells is prevented by promotion of cysteine uptake. Int. J. Cancer 75, 620–625 (1998).
Varelas, X. & Wrana, J. L. Coordinating developmental signaling: novel roles for the Hippo pathway. Trends Cell Biol. 22, 88–96 (2012).
Zhao, B. et al. Inactivation of YAP oncoprotein by the Hippo pathway is involved in cell contact inhibition and tissue growth control. Genes Dev. 21, 2747–2761 (2007).
Doll, S. et al. ACSL4 dictates ferroptosis sensitivity by shaping cellular lipid composition. Nat. Chem. Biol. 13, 91–98 (2017).
Zhang, Y. et al. Imidazole ketone erastin induces ferroptosis and slows tumor growth in a mouse lymphoma model. Cell Chem. Biol. 26, 623–633.e9 (2019).
Dixon, S. J. et al. Pharmacological inhibition of cystine–glutamate exchange induces endoplasmic reticulum stress and ferroptosis. eLife 3, e02523 (2014).
Dubey, S. et al. A phase II study of sorafenib in malignant mesothelioma: results of Cancer and Leukemia Group B 30307. J. Thorac. Oncol. 5, 1655–1661 (2010).
Papa, S. et al. Phase 2 study of sorafenib in malignant mesothelioma previously treated with platinum-containing chemotherapy. J. Thorac. Oncol. 8, 783–787 (2013).
Acknowledgements
We thank E. De Stanchina and E. Peguero for their help with mouse modelling experiments. We thank members of the Jiang laboratory for critical reading and suggestions. This work is supported by the National Institutes of Health (NIH) R01CA204232 (to X.J.), a Geoffrey Beene Cancer Research fund (to X.J.), a Functional Genomic Initiative fund (to X.J.), a China Scholarship Council fellowship (to J.W.), and NIH T32 fellowship 5T32GM008539-23 (to A.M.M.), National Cancer Institute R35CA209896 and P01CA087497 (to B.R.S.), National Natural Science Foundation of China 31871388 (to M.G.). This work is also supported by NCI cancer centre core grant P30 CA008748 to Memorial Sloan Kettering Cancer Center.
Author information
Authors and Affiliations
Contributions
J.W., A.M.M. and X.J. conceived the original idea and designed the study. J.W. and A.M.M. performed most experiments. M.G., H.B. and Y.L. generated several reagents and the inducible GPX4 knockout (GPX4-iKO) used in mouse experiments. B.R.S. supplied IKE and protocols for IKE administration to mice. Z.-N.C. and X.J. supervised the research. J.W., A.M.M., Z.-N.C. and X.J. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
B.R.S. holds equity in and serves as a consultant to Inzen Therapeutics, consults with GLG and Guidepoint Global, and is an inventor on patents and patent applications related to IKE and ferroptosis.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Intercellular contact suppresses ferroptosis.
a, b, HCT116 cells were seeded at the indicated density in 6-well plates and cultured for 24 h. a, Ferroptosis was measured by SYTOX Green staining after cystine starvation for 30 h. Phase contrast and fluorescent images are overlaid (original magnification, ×100). b, Cell death was measured in HCT116 cells at different densities treated with 30 μM erastin for 30 h, quantified by SYTOX Green staining followed by flow cytometry. c, Lipid ROS production of cells in b was assessed by C11-BODIPY staining followed by flow cytometry after 24 h of erastin treatment d, Cell death was measured in HCT116 cells cultured at the indicated cell densities and treated with 5 μM RSL3 for 24 h. e, Lipid ROS production in HCT116 cells cultured at the indicated cell densities and treated with 5 μM RSL3 for 16 h. f, HCT116 cells were seeded at 5 × 104 cells per well, grown for 24 h, and treated with: 1 µM Fer-1; 50 µg ml−1 of the iron chelator deferoxamine (DFO); 20 µM of the pan-caspase inhibitor Z-VAD-FMK; or 10 µM of the RIPK3 inhibitor GSK’872, in complete medium or cysteine-free medium for 30 h, followed by cell death measurement. n.s., P = 0.9999, 0.1995 (left to right). **P = 0.0070, 0.0050; one-way ANOVA. g, Cell death was measured in HCT116 cells seeded at 5 × 104 cells per well, grown for 24 h and treated with 5 µM RSL3 or DMSO and inhibitors as in f for 24 h. n.s., P = 0.4989. *P = 0.0366, ****P < 0.0001; one-way ANOVA. h, Cell death analysis in HCT116 cells seeded at 8 × 105 cells per well, grown for 24 h and treated with cystine-free medium containing the indicated amounts of glutamine for 30 h. Cell death was measured by SYTOX Green staining followed by flow cytometry. n.s., P = 0.5156; one-way ANOVA. All data are mean ± s.d. from n = 3 biological replicates.
Extended Data Fig. 2 ECAD-mediated intercellular interaction regulates ferroptosis in a density-dependent manner.
a, ECAD expression increases with cell density in HCT116 (top) and H1650 (bottom) cells. b, BT474 cells express high levels of ECAD regardless of cell density. c, MDA231 cells express low levels of ECAD regardless of cell density. d, Immunofluorescence of ECAD at increasing cell density in HCT116 cells (original magnification, ×200). e, Tumour spheroids generated from HCT116 or MDA231 cells were fixed, sectioned and stained for ECAD expression by immunohistochemistry (original magnification, ×100). f, HCT116 cells were treated with either IgG or an anti-ECAD antibody that blocks dimerization. Cell death was measured by propidium iodide staining followed by flow cytometry after cystine starvation for 30 h. ***P = 0.0003; two-tailed t-test. g, Western blot analysis of the expression of ECAD and NCAD in HCT116 cells after CRISPR–Cas9-mediated ECAD depletion (ΔECAD). h, ECAD depletion in HCT116 cells was further confirmed by immunofluorescence (magnification 200×). i, Cell death measurement of ΔECAD and the parental cell line seeded at a density of 4 × 105 cells per well after cystine starvation for 30 h. Original magnification, ×100. ****P < 0.0001; two tailed t-test. j, Western blot analysis confirming reconstitution of ECAD or ectodomain-truncated ECAD (ECADΔecto) into ECAD-depleted HCT116 cells. k, ΔECAD cells or ΔECAD cells re-expressing full-length ECAD or ECADΔecto were cultured into spheroids and treated with erastin for 30 h, followed by SYTOX Green staining to monitor cell death (magnification 100×). All data are mean ± s.d. from n = 3 biological replicates.
Extended Data Fig. 3 Cell density, ECAD and NF2 converge on the transcriptional co-regulator YAP.
a, HCT116 cells were cultured at different cell densities and YAP localization was assessed by immunofluorescence. Original magnification, ×200. Bottom images are enlarged to show localization. b, Western blot analysis of phosphorylated YAP (p-YAP; at Ser127) and YAP in whole-cell or cytosolic fractions of HCT116 cells cultured at different densities. PARP was used as a marker for nuclear protein. c, Western blot analysis of ECAD, YAP and p-YAP in parental and ΔECAD HCT116 cells. d, Immunofluorescence of YAP (green) and ECAD (red) in parental and ΔECAD HCT116 cells. Original magnification, ×400. e, Western blot analysis confirming knockdown efficiency of ECAD (shECAD #1 and #2), NF2 (shNF2 #1 and #2), or LATS1 or LATS2 (shLATS1/2 #1 and #2, and shLATSs1 #1) in HCT116 cells. f, Western blot analysis of NF2, p-YAP and YAP in HCT116 cells transfected with shNT and shNF2. g, Knockdown of NF2 in HCT116 cells induced the nuclear accumulation of YAP in dense cell cultures, as assessed by immunofluorescence. Original magnification, ×200. h, Transcriptional levels of the canonical YAP targets CTGF and CYR61 by qPCR in HCT116 cells seeded at 1 × 105 (sparse) or 8 × 105 (confluent) cells per well in 6-well dishes and grown for 24 h. ***P = 0.0002, ****P < 0.0001; two-tailed t-test. i, Transcription levels of CTGF and CYR61 measured by qPCR in parental and ΔECAD HCT116 cells plated at high density. **P = 0.0013, ****P < 0.0001; two-tailed t-test. j, YAP/TEAD transcriptional activity in HCT116 and ΔECAD cells was measured by a luciferase assay using the 8xGTIIC-luciferase reporter. ****P < 0.0001; two-tailed t-test. k, Transcription levels of CTGF and CYR61 measured by qPCR in HCT116 cells plated at high density and transfected with shNT or shNF2. ***P = 0.0007, 0.0005 (left to right); two-tailed t-test. l, YAP/TEAD transcriptional activity in HCT116 cells transfected with shNT and shNF2 cells was measured by a luciferase assay using the 8xGTIIC-luciferase reporter. ***P = 0.0002; two-tailed t-test. All data are mean ± s.d. from n = 3 biological replicates.
Extended Data Fig. 4 The Hippo pathway links cell density and intercellular contact to ferroptosis.
a, Confluent cells were subjected to cystine starvation for 30 h. Cell death was determined by propidium iodide staining. b, HCT116 cells expressing shNT, shECAD, shNF2 or shLATS1/2 as indicated were treated with 5 µM RSL3 with or without 2 µM Fer-1. Cell death was measured at 18 h. ****P < 0.0001; one-way ANOVA. c, Lipid ROS production of cells as in b was assessed at 12 h after treatment. ****P < 0.0001; one-way ANOVA. d, Cumulative cell growth curve expressed as the total cell count of HCT116 cells transfected with shNT, shECAD, shNF2 or shLATS1/2. n.s., P = 0.9497; two-way ANOVA. e, Western blot analysis of the expression and phosphorylation of NF2 in HCT116 cells transfected with enhanced green fluorescent protein (eGFP)-tagged PAK containing a prenylation (CAAX) motif (thus constitutively active), or an inactive mutant form of PAK (K298R). f, YAP/TEAD transcriptional activity was measured by a luminescence assay in HCT116 cells expressing activated or inactive PAK and transfected with the 8xGTIIC-luciferase reporter. ****P < 0.0001; one-way ANOVA. g, Cell death was measured in HCT116 cells plated at high density expressing activated or inactive PAK, and treated with cystine-free medium with or without 1 µM Fer-1 for 30 h. ****P < 0.0001; one-way ANOVA. h, Cells were prepared as in g and treated with DMSO or 5 µM RSL3 with or without 1 µM Fer-1. Cell death was measured at 24 h. ****P < 0.0001; one-way ANOVA. All data are mean ± s.d. from n = 3 biological replicates.
Extended Data Fig. 5 NF2 expression correlates with sensitivity to ferroptosis in mesothelioma cell lines.
a, Western blot analysis of the expression of LATS1 and LATS2 in the indicated mesothelioma cell lines. b, Spheroids were treated with 10 μM erastin for 24 h before SYTOX Green staining. Original magnification, ×40. c, Western blot analysis confirming knockdown efficiency of NF2 shRNA in 211H cells. d, Confluent 211H cells transfected with shNT or shNF2 were treated with 1 µM RSL3, with or without 2 µM Fer-1. Cell death (left, 24 h after treatment) and lipid ROS production (right, 16 h) were measured. ****P < 0.0001; one-way ANOVA. e, NF2-mutant Meso33 cells were reconstituted with wild-type NF2, and the expression of NF2 was confirmed by western blot. f, Localization of YAP (green) under sparse or confluent conditions in Meso33 cells expressing wild-type NF2 was determined by immunofluorescence. Original magnification, ×200. g, Meso33 cells expressing wild-type NF2 were cultured under sparse or confluent conditions and stimulated with cystine-free medium. Cell death was measured by SYTOX Green staining coupled with flow cytometry after 24 h of treatment. Original magnification, ×100. n.s., P = 0.1874. *P = 0.0104; two-tailed t-test. h, Meso33 cells expressing wild-type NF2 were cultured as in g and the production of lipid ROS was measured after cystine starvation for 16 h. n.s., P = 0.4860. *P = 0.0201; two-tailed t-test. i, Meso33 spheroids containing Dox-inducible NF2 were grown in the presence or absence of 1 µg ml−1 Dox for 72 h, at which point 10 µM erastin was added. Cell death was measured after 24 h by SYTOX Green staining of spheroids. Original magnification, ×100. All data are mean ± s.d. from n = 3 biological replicates.
Extended Data Fig. 6 NCAD suppresses ferroptosis in MSTO-211H cells in a density-dependent manner.
a, Western blot analysis of the levels of NCAD, p-YAP and total YAP in 211H cells cultured at different cell densities. b, Knockdown efficiency of NCAD shRNA (shNcad #1 and #2) was assessed by western blot analysis in 211H cells infected with lentiviruses expressing shNCAD. c, Confluent or sparse shNT or shNCAD 211H cells, as indicated, were subjected to cystine starvation for 24 h, at which point cell death was monitored by SYTOX Green staining. Original magnification, ×100. d, Flow cytometric quantification of cell death in c. n.s., P = 0.8426. ***P = 0.0056, 0.0015 (left to right), ****P < 0.0001; one-way ANOVA. e, Confluent or sparse shNT or shNCAD 211H cells, as indicated, were treated with 1 µM RSL3 for 16 h, at which point cell death was measured by SYTOX Green staining followed by flow cytometry. n.s., P = 0.3012, *P = 0.0315, ****P < 0.0001; one-way ANOVA. f, Spheroids generated from shNT and shNCAD 211H cells were treated with 10 μM erastin for 24 h, and cell death was determined by SYTOX Green staining. Original magnification, ×40. g, Cell viability of spheroids described in f was assayed by measuring cellular ATP levels. n.s., P = 0.4365, ***P = 0.0006, ****P < 0.0001; two-tailed t-test. h, shNT or shNCAD 211H cells were plated at high density and YAP localization was assessed by immunofluorescence. Original magnification, ×400. i, Transcription levels of CTGF and CYR61 measured by qPCR 211H cells plated at high density and transfected with shNT or shNCAD. *P = 0.0108, **P = 0.0016, ***P = 0.0007, ****P < 0.0001; one-way ANOVA. j, YAP/TEAD transcriptional activity in 211H cells transfected with shNT or shNCAD was measured by a luciferase assay using the 8xGTIIC-luciferase reporter. ***P = 0.0002, ****P < 0.0001; one-way ANOVA. All data are mean ± s.d. from n = 3 biological replicates.
Extended Data Fig. 7 Ferroptosis can be regulated by the Hippo pathway in non-epithelial cells.
a, Cell death was measured in MEFs after cystine starvation for 12 h. b, Cells were treated as in a and lipid ROS production was measured at 8 h. c, Cell death was measured in MEFs seeded at the indicated densities and treated with 1 µM erastin for 12 h. d, Cells were treated as in c and production of lipid ROS was measured at 8 h. e, MEFs treated with 1 µM RSL3 at the indicated densities were measured for cell death at 8 h. f, Cells were treated as in e and the production of lipid ROS was measured at 5 h. g, Immunofluorescence probing for YAP localization in MEFs seeded at increasing density. Bottom images are enlarged to show localization. Original magnification, ×400. h, MEFs were transfected with NF2 shRNAs (shNF2 #1 and #2), and knockdown efficiency was assessed by western blot. i, Immunofluorescence probing for YAP localization after NF2 knockdown in MEFs. Original magnification, ×200. j, Increased cell death occurred in confluent MEFs after NF2 depletion (shNF2 #2) and cystine starvation, or treatment with erastin (1 µM, 12 h) or RSL3 (1 µM, 8 h), and this increase was blocked by Fer-1 (2 µM). ***P = 0.0007, 0.0007, 0.0006 (left to right); two-tailed t-test. k, Cells were treated as in j and lipid ROS production was assessed at 8 h (cystine starvation, erastin) or 5 h (RSL3). ****P < 0.0001; two-tailed t-test. l, Western blot analysis of expression of YAP and TAZ in CA-46 Burkitt lymphoma cells. m, Cell death measurement of CA-46 cells treated as indicated after 24 h. All data are mean ± s.d. from n = 3 biological replicates.
Extended Data Fig. 8 YAP regulates ferroptosis.
a, Western blot confirming expression of YAP(S127A) in HCT116 cells. b, YAP localization in HCT116 cells expressing YAP(S127A) assessed by immunofluorescence. Original magnification, ×200. c, Transcriptional levels of CTGF and CYR61 measured by qPCR in HCT116 cells expressing YAP(S127A). ***P = 0.0005, ****P < 0.0001; two-tailed t-test. d, YAP/TEAD transcriptional activity in HCT116 cells expressing YAP(S127A) was measured by a luminescence assay using the 8xGTIIC-luciferase reporter. ****P < 0.0001; two-tailed t-test. e, Spheroids generated from parental and YAP(S127A)-overexpressing HCT116 cells were treated with 15 μM erastin for 30 h, followed by SYTOX Green staining. Original magnification, ×40. f, 211H cells were infected with retroviral vectors encoding the Flag–YAP(S127A) mutant, and levels of Flag–YAP, YAP and p-YAP were analysed by western blot. g, Localization of YAP (green) was determined by immunofluorescence in 211H cells expressing constitutively active YAP. Original magnification, ×200. h, Parental and YAP(S127A)-overexpressing 211H cells were cultured under sparse or confluent conditions and cell death was measured after cystine starvation for 24 h. *P = 0.0354, ***P = 0.0003; two-tailed t-test. i, Cells were cultured as in h and the production of lipid ROS was measured after 16 h of cystine starvation. ***P = 0.0006, *** P < 0.0001; two-tailed t-test. j, Spheroids generated from parental and YAP(S127A)-overexpressing 211H cells were treated with 10 μM erastin for 24 h and cell death was measured by SYTOX staining. Original magnification, ×40. k, Cells were cultured as in j and cell viability within spheroids was determined by measuring cellular ATP levels. n.s., P = 0.1534. ***P = 0.0009l; two-tailed t-test. l, YAP was knocked out by CRISPR–Cas9 (sgYAP) in shNF2 HCT116 cells. m, HCT116 cells were transduced with retroviral particles containing mCherry–ACSL4 and/or transfected with TFRC. Expression levels were assayed by western blot. Two bands were detected for mCherry–ACSL4, representing the full-length mCherry–ACSL4 and that with the mCherry tag truncated. n, HCT116 cells treated as in m were plated at the indicated density and treated with 2 µM RSL3 for 24 h. Cell death was measured by SYTOX Green staining coupled with flow cytometry. *P = 0.0158, **P = 0.0012, ****P < 0.0001; one-way ANOVA. o, Western blot analysis confirming knockdown of TFRC in HCT116 shNF2 cells. p, Western blot confirming knockdown of TFRC in HCT116 ΔECAD (sgECAD) cells. q, Cells as in p were treated with medium lacking cystine for 30 h. Cell death was measured by SYTOX Green staining coupled with flow cytometry. ****P < 0.0001; one-way ANOVA. r, Western blot analysis of HCT116 cells after CRISPR–Cas9-mediated knockout of ACSL4 (sgACSL4) and/or transfection with shNF2. All data are mean ± s.d. from n = 3 biological replicates.
Extended Data Fig. 9 NF2, LATS1 and LATS2 regulate cancer cell sensitivity to ferroptosis in vivo.
a, Top, western blot analysis confirming knockout of GPX4 (GPX4 iKO) in 211H cells after treatment with 1 µg ml−1 Dox for 5 days (top). Bottom, cells were infected with the indicated hairpins. b, Spheroids formed by shNT-GPX4-iKO or shNF2-GPX4-iKO 211H cells were treated with or without Dox for five days. Cell death and viability, respectively, were determined by SYTOX staining (top) and cellular ATP levels (bottom). Original magnification, ×40. n.s., P = 0.3523, ***P = 0.0007; two-tailed t-test. c, shNT-GPX4-iKO and shNF2-GPX4-iKO cells were subcutaneously injected into nude mice. The effect of NF2 knockdown on xenografted tumours was validated by immunostaining of NF2, ACSL4, TFRC and YAP, all counterstained with haematoxylin (blue), on sections of tumours bearing shNT and shNF2 as indicated. Inset images are enlarged to show TFRC expression at plasma membranes and increase in nuclear localization of YAP. d, shNT-GPX4-iKO cells and shNF2-GPX4-iKO cells were subcutaneously injected into nude mice followed by treatment with or without Dox (to induce GPX4 knockout; n = 8 mice per group). Representative haematoxylin and eosin (H&E) and immunostaining images of GPX4, PTGS2 and Ki67, all counterstained with haematoxylin (blue), are shown from sections of xenografted tumours. e, Images of resected MSTO-211H subcutaneous tumours. Scale bar, 1 cm. f, Mice injected subcutaneously with HCT116 cells expressing shNT or shLATS1/2 as indicated were treated with or without IKE. Top, images of resected HCT116 shNT or shLATS1/2 tumours. Bottom, mass of resected tumours. n = 6 mice per group. n.s., P = 0.8677. *P = 0.0486; two-tailed t-test. g, Representative BLI showing the tumour growth of indicated cells in an orthotopic model of mesothelioma in nonobese diabetic/severe combined immunodeficiency (NOD/SCID) mice. Dox treatment started when the average total flux reached 108 photons per second (time point 0). h, Tumour spheroids of 211H cells expressing shNT or shNF2 were grown in Matrigel, and invasion was monitored. In the representative images, arrows show protrusions extruded from the main body of spheroids. Original magnification, ×400. All data are mean ± s.d. from n = 3 biological replicates.
Extended Data Fig. 10 The Hippo pathway as a potential biomarker for predicting cancer cell sensitivity to ferroptosis.
a, Cell death was measured in HCT116 cells seeded at 0.5 × 105 cells per 3.5 cm2 well (sparse) or 4 × 105 cells per 3.5 cm2 well (confluent) and grown for 24 h. Cells were treated with DMSO, 10 µM sorafenib or 10 µM sorafenib plus 2 µM Fer-1 as indicated for 24 h. ****P < 0.0001; two-tailed t-test. b, Cell death was measured in parental or ΔECAD HCT116 cells seeded at 4 × 105 cells per 3.5 cm2 well and grown for 24 h. Cells were treated as in a. *P = 0.0394; two-tailed t-test. c, d, Cell death was measured in HCT116 (c) or 211H (d) shNT or shNF2 cells seeded at high density and treated as in a. *P = 0.0167, ***P = 0.0004; two-tailed t-test. e, f, Cell death was measured in HCT116 (e) or 2211H (f) cells expressing parental or YAP(S127A) cells seeded at high density and treated as in a. *P = 0.0143, **P = 0.0014; two-tailed t-test. g, Cell death was measured in HCT116 shNT or shLATS1/2 cells seeded at high density and treated as in a. **P = 0.0017; two-tailed t-test. h, NF639 cells, derived from mouse mammary tumours containing MMTV-neu, were treated with various concentrations of TGFβ for 48 h. mRNA expression of a panel of EMT-related genes was assayed by qPCR. i, NF639 cells were treated with or without 6 ng µl−1 TGFβ for 48 h, at which point they were plated at low density (0.8 × 105 cells per 3.5 cm2 well), grown overnight and treated with medium containing or lacking cystine, with or without 1 µM Fer-1 for 12 h, followed by cell death measurement. n.s., P = 0.0777; two-tailed t-test. j, NF639 cells were plated at 3.2 × 105 cells per 3.5 cm2 well, grown overnight and treated as described in a. ****P < 0.0001; two-tailed t-test. k, 211H cells were infected with YAP(S127A) or the activated mutant PIK3CA(H1047R). Lysates were probed for overexpression and phosphorylated AKT (p-AKT; S473) to confirm the activity of PIK3CA(H1047R). l, Approximately 50,000 211H cells were seeded in 3.5-cm2 plates and grown for 5 days. Cells were counted daily. ***P = 0.0007, ****P < 0.0001; two-way ANOVA. m, Cell death was measured by flow cytometry in 211H cells seeded at high density (8 × 105 cells per 3.5-cm2 well) after cystine starvation for 24 h. n.s., P = 0.8838. **P = 0.0041; one-way ANOVA. All data are mean ± s.d.; n = 3 biological replicates.
Supplementary information
Supplementary Figures
This file contains Supplementary Figure 1, the gel source data and Supplementary Figure 2, the gating strategy for flow cytometry experiments.
Supplementary Table 1
A list of shRNAs.
Supplementary Table 2
A list of oligonucleotides used for q-PCR and ChIP analysis, and subcloning.
Source data
Rights and permissions
About this article
Cite this article
Wu, J., Minikes, A.M., Gao, M. et al. Intercellular interaction dictates cancer cell ferroptosis via NF2–YAP signalling. Nature 572, 402–406 (2019). https://doi.org/10.1038/s41586-019-1426-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-1426-6
This article is cited by
-
Role of ferroptosis and ferroptosis-related long non'coding RNA in breast cancer
Cellular & Molecular Biology Letters (2024)
-
KCNA1 promotes the growth and invasion of glioblastoma cells through ferroptosis inhibition via upregulating SLC7A11
Cancer Cell International (2024)
-
Low-dose hypomethylating agents cooperate with ferroptosis inducers to enhance ferroptosis by regulating the DNA methylation-mediated MAGEA6-AMPK-SLC7A11-GPX4 signaling pathway in acute myeloid leukemia
Experimental Hematology & Oncology (2024)
-
Harnessing ferroptosis for enhanced sarcoma treatment: mechanisms, progress and prospects
Experimental Hematology & Oncology (2024)
-
Synergistic suppression of ovarian cancer by combining NRF2 and GPX4 inhibitors: in vitro and in vivo evidence
Journal of Ovarian Research (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.