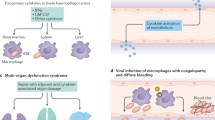
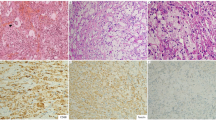
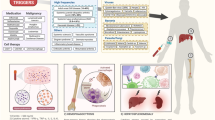
Abstract
Still’s disease is a rare inflammatory syndrome that encompasses systemic juvenile idiopathic arthritis and adult-onset Still’s disease, both of which can exhibit life-threatening complications, including macrophage activation syndrome (MAS), a secondary form of haemophagocytic lymphohistiocytosis. Genetic insights into Still’s disease involve both HLA and non-HLA susceptibility genes, suggesting the involvement of adaptive immune cell-mediated immunity. At the same time, phenotypic evidence indicates the involvement of autoinflammatory processes. Evidence also implicates the type I interferon signature, mechanistic target of rapamycin complex 1 signalling and ferritin in the pathogenesis of Still’s disease and MAS. Pathological entities associated with Still’s disease include lung disease that could be associated with biologic DMARDs and with the occurrence of MAS. Historically, monophasic, recurrent and persistent Still’s disease courses were recognized. Newer proposals of alternative Still’s disease clusters could enable better dissection of clinical heterogeneity on the basis of immune cell profiles that could represent diverse endotypes or phases of disease activity. Therapeutically, data on IL-1 and IL-6 antagonism and Janus kinase inhibition suggest the importance of early administration in Still’s disease. Furthermore, there is evidence that patients who develop MAS can be treated with IFNγ antagonism. Despite these developments, unmet needs remain that can form the basis for the design of future studies leading to improvement of disease management.
Key points
-
Still’s disease, a rare inflammatory syndrome that encompasses systemic juvenile idiopathic arthritis and adult-onset Still’s disease, can exhibit life-threatening macrophage activation syndrome (MAS), a secondary form of haemophagocytic lymphohistiocytosis.
-
Genetic insights into Still’s disease involve both HLA and non-HLA susceptibility genes, suggesting autoimmune involvement, whereas phenotypic evidence suggests the involvement of an autoinflammatory process.
-
Evidence indicates that pathogenic mechanisms involving the type I interferon signature, mechanistic target of rapamycin complex 1 and pro-inflammatory properties of ferritin contribute to the development of Still’s disease and MAS.
-
Pathological conditions now associated with Still’s disease include Still’s disease-related lung involvement, which is potentially associated with the use of biologic DMARDs and the occurrence of MAS.
-
Clusters of patients with Still’s disease have been identified by the characterization of immune cell populations, which might represent diverse endotypes or different phases of disease activity.
-
Therapeutically, evidence is available in relation to IL-1 antagonism and Janus kinase inhibition, including early administration, suggesting that MAS in Still’s disease might be tractable with IFNγ antagonism.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Lee, J. J. Y. & Schneider, R. Systemic juvenile idiopathic arthritis. Pediatr. Clin. North. Am. 65, 691–709 (2018).
Giacomelli, R., Ruscitti, P. & Shoenfeld, Y. A comprehensive review on adult onset Still’s disease. J. Autoimmun. 93, 24–36 (2018).
Nigrovic, P. A. Autoinflammation and autoimmunity in systemic juvenile idiopathic arthritis. Proc. Natl Acad. Sci. USA 112, 15785–15786 (2015).
Gerfaud-Valentin, M., Jamilloux, Y., Iwaz, J. & Sève, P. Adult-onset Still’s disease. Autoimmun. Rev. 13, 708–722 (2014).
Ombrello, M. J. et al. HLA-DRB1*11 and variants of the MHC class II locus are strong risk factors for systemic juvenile idiopathic arthritis. Proc. Natl Acad. Sci. USA 112, 15970–15975 (2015).
McGonagle, D., Ramanan, A. V. & Bridgewood, C. Immune cartography of macrophage activation syndrome in the COVID-19 era. Nat. Rev. Rheumatol. 17, 145–157 (2021).
Nirmala, N. et al. Gene-expression analysis of adult-onset Still’s disease and systemic juvenile idiopathic arthritis is consistent with a continuum of a single disease entity. Pediatr. Rheumatol. Online J. 13, 50 (2015).
Nigrovic, P. A., Raychaudhuri, S. & Thompson, S. D. Review: Genetics and the classification of arthritis in adults and children. Arthritis Rheumatol. 70, 7–17 (2018).
Ruscitti, P. et al. Disparities in the prevalence of clinical features between systemic juvenile idiopathic arthritis and adult-onset Still’s disease. Rheumatology 61, 4124–4129 (2022).
Pay, S. et al. A multicenter study of patients with adult-onset Still’s disease compared with systemic juvenile idiopathic arthritis. Clin. Rheumatol. 25, 639–644 (2006).
Fautrel, B. et al. Proposal for a new set of classification criteria for adult-onset Still disease. Medicine 81, 194–200 (2002).
Petty, R. E. et al. International League of Associations for Rheumatology classification of juvenile idiopathic arthritis: second revision, Edmonton, 2001. J. Rheumatol. 31, 390–392 (2004).
Martini, A. et al. Toward new classification criteria for juvenile idiopathic arthritis: first steps, Pediatric Rheumatology International Trials Organization International Consensus. J. Rheumatol. 46, 190–197 (2019).
Yamaguchi, M. et al. Preliminary criteria for classification of adult Still’s disease. J. Rheumatol. 19, 424–430 (1992).
Ruscitti, P. et al. Advances in immunopathogenesis of macrophage activation syndrome during rheumatic inflammatory diseases: toward new therapeutic targets? Expert Rev. Clin. Immunol. 13, 1041–1047 (2017).
Mitrovic, S. & Fautrel, B. Complications of adult-onset Still’s disease and their management. Expert Rev. Clin. Immunol. 14, 351–365 (2018).
Onel, K. B. et al. 2021 American College of Rheumatology guideline for the treatment of juvenile idiopathic arthritis: therapeutic approaches for oligoarthritis, temporomandibular joint arthritis, and systemic juvenile idiopathic arthritis. Arthritis Rheumatol. 74, 553–569 (2022).
Efthimiou, P. et al. Adult-onset Still’s disease in focus: clinical manifestations, diagnosis, treatment, and unmet needs in the era of targeted therapies. Semin. Arthritis Rheum. 51, 858–874 (2021).
Di Cola, I., Cipriani, P. & Ruscutti, P. Perspectives on the use of non-biological pharmacotherapy for adult-onset Still’s disease. Expert Opin. Pharmacother. 23, 1577–1587 (2022).
Castañeda, S., Blanco, R. & González-Gay, M. A. Adult-onset Still’s disease: advances in the treatment. Best Pract. Res. Clin. Rheumatol. 30, 222–238 (2016).
Nigrovic, P. A. Review: Is there a window of opportunity for treatment of systemic juvenile idiopathic arthritis? Arthritis Rheumatol. 66, 1405–1413 (2014).
Feist, E., Mitrovic, S. & Fautrel, B. Mechanisms, biomarkers and targets for adult-onset Still’s disease. Nat. Rev. Rheumatol. 14, 603–618 (2018).
Jamilloux, Y. et al. Pathogenesis of adult-onset Still’s disease: new insights from the juvenile counterpart. Immunol. Res. 61, 53–62 (2015).
Pascual, V., Allantaz, F., Arce, E., Punaro, M. & Banchereau, J. Role of interleukin-1 (IL-1) in the pathogenesis of systemic onset juvenile idiopathic arthritis and clinical response to IL-1 blockade. J. Exp. Med. 201, 1479–1486 (2005).
Peckham, D., Scambler, T., Savic, S. & McDermott, M. F. The burgeoning field of innate immune-mediated disease and autoinflammation. J. Pathol. 241, 123–139 (2017).
Vastert, S. J., Kuis, W. & Grom, A. A. Systemic JIA: new developments in the understanding of the pathophysiology and therapy. Best Pract. Res. Clin. Rheumatol. 23, 655–664 (2009).
Tang, S. et al. Genetic and pharmacological targeting of GSDMD ameliorates systemic inflammation in macrophage activation syndrome. J. Autoimmun. 133, 102929 (2022).
Evavold, C. L. et al. The pore-forming protein gasdermin D regulates interleukin-1 secretion from living macrophages. Immunity 48, 35–44.e6 (2018).
Wang, C. et al. NLRP3 inflammasome activation triggers gasdermin D-independent inflammation. Sci. Immunol. 6, eabj3859 (2021).
Chen, D. Y. et al. The potential role of advanced glycation end products (AGEs) and soluble receptors for AGEs (sRAGE) in the pathogenesis of adult-onset Still’s disease. BMC Musculoskelet. Disord. 16, 111 (2015).
Kim, H. A. et al. Endogenous ligand S100A8/A9 levels in adult-onset Still’s disease and their association with disease activity and clinical manifestations. Int. J. Mol. Sci. 17, 1342 (2016).
Jung, J. Y., Suh, C.-H. & Kim, H.-A. The role of damage-associated molecular pattern for pathogenesis and biomarkers in adult-onset Still’s disease. Expert Rev. Mol. Diagn. 19, 459–468 (2019).
Cader, M. Z. et al. FAMIN is a multifunctional purine enzyme enabling the purine nucleotide cycle. Cell 180, 278–295.e23 (2020).
Saveljeva, S. et al. A purine metabolic checkpoint that prevents autoimmunity and autoinflammation. Cell Metab. 34, 106–124.e10 (2022).
Wakil, S. M. et al. Association of a mutation in LACC1 with a monogenic form of systemic juvenile idiopathic arthritis. Arthritis Rheumatol. 67, 288–295 (2015).
Kadowaki, T. et al. Haploinsufficiency of A20 causes autoinflammatory and autoimmune disorders. J. Allergy Clin. Immunol. 141, 1485–1488.e11 (2018).
Barnett, K. C., Li, S., Liang, K. & Ting, J. P.-Y. A 360° view of the inflammasome: mechanisms of activation, cell death, and diseases. Cell 186, 2288–2312 (2023).
Brown, R. A. et al. Neutrophils from children with systemic juvenile idiopathic arthritis exhibit persistent proinflammatory activation despite long-standing clinically inactive disease. Front. Immunol. 9, 2995 (2018).
Nam, S. W., Kang, S., Lee, J. H. & Yoo, D. H. Different features of interleukin-37 and interleukin-18 as disease activity markers of adult-onset Still’s disease. J. Clin. Med. 10, 910 (2021).
Aizaki, Y., Yazawa, H., Sato, K. & Mimura, T. Dual effects of interleukin-10 on natural killer cells and monocytes and the implications for adult-onset Still’s disease. Clin. Exp. Rheumatol. 39, 22–29 (2021).
Imbrechts, M. et al. Insufficient IL-10 production as a mechanism underlying the pathogenesis of systemic juvenile idiopathic arthritis. J. Immunol. 201, 2654–2663 (2018).
Serhan, C. N. Pro-resolving lipid mediators are leads for resolution physiology. Nature 510, 92–101 (2014).
Navarini, L. et al. The specialized pro-resolving lipid mediator protectin D1 affects macrophages differentiation and activity in adult-onset Still’s disease and COVID-19, two hyperinflammatory diseases sharing similar transcriptomic profiles. Front. Immunol. 14, 1148268 (2023).
Terkeltaub, R. et al. HLA-Bw35 and prognosis in adult Still’s disease. Arthritis Rheum. 24, 1469–1472 (1981).
Wouters, J. M., Reekers, P. & van de Putte, L. B. Adult-onset Still’s disease. Disease course and HLA associations. Arthritis Rheum. 29, 415–418 (1986).
Asano, T. et al. Effects of HLA-DRB1 alleles on susceptibility and clinical manifestations in Japanese patients with adult onset Still’s disease. Arthritis Res. Ther. 19, 199 (2017).
Li, Z. et al. Both HLA class I and II regions identified as genome-wide significant susceptibility loci for adult-onset Still’s disease in Chinese individuals. Ann. Rheum. Dis. 79, 161–163 (2020).
Teng, J. L. et al. The amino acid variants in HLA II molecules explain the major association with adult-onset Still’s disease in the Han Chinese population. J. Autoimmun. 116, 102562 (2021).
Chen, X. et al. Serum VEGF-C as an evaluation marker of disease activity in adult-onset Still’s disease. Rheumatol. Int. 42, 149–157 (2022).
Sugiura, T. et al. A promoter haplotype of the interleukin-18 gene is associated with juvenile idiopathic arthritis in the Japanese population. Arthritis Res. Ther. 8, R60 (2006).
Wang, F. F. et al. A genetic role for macrophage migration inhibitory factor (MIF) in adult-onset Still’s disease. Arthritis Res. Ther. 15, R65 (2013).
Sighart, R. et al. Evidence for genetic overlap between adult onset Still’s disease and hereditary periodic fever syndromes. Rheumatol. Int. 38, 111–120 (2018).
Wang, M. et al. Association of the leukocyte immunoglobulin-like receptor A3 gene with neutrophil activation and disease susceptibility in adult-onset Still’s disease. Arthritis Rheumatol. 73, 1033–1043 (2021).
Hung, W. T. et al. The association of ATG16L1 variations with clinical phenotypes of adult-onset Still’s disease. Genes 12, 904 (2021).
Jørgensen, S. E. et al. Systemic juvenile idiopathic arthritis and recurrent macrophage activation syndrome due to a CASP1 variant causing inflammasome hyperactivation. Rheumatology 59, 3099–3105 (2020).
Vastert, S. J. et al. Mutations in the perforin gene can be linked to macrophage activation syndrome in patients with systemic onset juvenile idiopathic arthritis. Rheumatology 49, 441–449 (2010).
Rao, S., Tsang, L. S.-L., Zhao, M., Shi, W. & Lu, Q. Adult-onset Still’s disease: a disease at the crossroad of innate immunity and autoimmunity. Front. Med. 9, 881431 (2022).
Manna, R. & Rigante, D. The everchanging framework of autoinflammation. Intern. Emerg. Med. 16, 1759–1770 (2021).
Kim, J. W., Ahn, M.-H., Jung, J.-Y., Suh, C.-H. & Kim, H.-A. An update on the pathogenic role of neutrophils in systemic juvenile idiopathic arthritis and adult-onset Still’s disease. Int. J. Mol. Sci. 22, 13038 (2021).
Liu, Y., Xia, C., Chen, J., Fan, C. & He, J. Elevated circulating pro-inflammatory low-density granulocytes in adult-onset Still’s disease. Rheumatology 60, 297–303 (2021).
Chen, P. K. et al. Elevated expression of C-type lectin domain family 5-member A (CLEC5A) and its relation to inflammatory parameters and disease course in adult-onset Still’s disease. J. Immunol. Res. 2020, 9473497 (2020).
Liao, T. L. et al. MicroRNA-223 inhibits neutrophil extracellular traps formation through regulating calcium influx and small extracellular vesicles transmission. Sci. Rep. 11, 15676 (2021).
Jia, J. et al. Circulating neutrophil extracellular traps signature for identifying organ involvement and response to glucocorticoid in adult-onset Still’s disease: a machine learning study. Front. Immunol. 11, 563335 (2020).
Backlund, M., Venge, P. & Berntson, L. A cross-sectional cohort study of the activity and turnover of neutrophil granulocytes in juvenile idiopathic arthritis. Pediatr. Rheumatol. Online J. 19, 102 (2021).
Ma, Y. et al. Enhanced type I interferon signature induces neutrophil extracellular traps enriched in mitochondrial DNA in adult-onset Still’s disease. J. Autoimmun. 127, 102793 (2022).
Tsuboi, H. et al. Activation mechanisms of monocytes/macrophages in adult-onset Still disease. Front. Immunol. 13, 953730 (2022).
Chen, P.-K. & Chen, D.-Y. An update on the pathogenic role of macrophages in adult-onset Still’s disease and its implication in clinical manifestations and novel therapeutics. J. Immunol. Res. 2021, 8998358 (2021).
Hu, Q. et al. Increased neutrophil extracellular traps activate NLRP3 and inflammatory macrophages in adult-onset Still’s disease. Arthritis Res. Ther. 21, 9 (2019).
Di Cola, I., Ruscutti, P., Giacomelli, R. & Cipriani, P. The pathogenic role of interferons in the hyperinflammatory response on adult-onset Still’s disease and macrophage activation syndrome: paving the way towards new therapeutic targets. J. Clin. Med. 10, 1164 (2021).
Segawa, S. et al. Placenta specific 8 suppresses IL-18 production through regulation of autophagy and is associated with adult Still disease. J. Immunol. 201, 3534–3545 (2018).
Huang, Z. et al. mTORC1 links pathology in experimental models of Still’s disease and macrophage activation syndrome. Nat. Commun. 13, 6915 (2022).
Concha, S., Rey-Jurado, E., Poli, M. C., Hoyos-Bachiloglu, R. & Borzutzky, A. Refractory systemic juvenile idiopathic arthritis successfully treated with rapamycin. Rheumatology 60, e250–e251 (2021).
Vandenhaute, J., Wouters, C. H. & Matthys, P. Natural killer cells in systemic autoinflammatory diseases: a focus on systemic juvenile idiopathic arthritis and macrophage activation syndrome. Front. Immunol. 10, 3089 (2020).
Lee, S. J. et al. Natural killer T cell deficiency in active adult-onset Still’s disease: correlation of deficiency of natural killer T cells with dysfunction of natural killer cells. Arthritis Rheum. 64, 2868–2877 (2012).
Put, K. et al. Inflammatory gene expression profile and defective interferon-γ and granzyme K in natural killer cells from systemic juvenile idiopathic arthritis patients. Arthritis Rheumatol. 69, 213–224 (2017).
Ohya, T. et al. Impaired interleukin-18 signaling in natural killer cells from patients with systemic juvenile idiopathic arthritis. ACR Open Rheumatol. 4, 503–510 (2022).
Huang, Z. et al. Type I interferon signature and cycling lymphocytes in macrophage activation syndrome. J. Clin. Invest. 26, e165616 (2023).
Domínguez-Andrés, J. et al. Inflammatory Ly6Chigh monocytes protect against candidiasis through IL-15-driven NK cell/neutrophil activation. Immunity 46, 1059–1072.e4 (2017).
Jain, A. et al. T cells instruct myeloid cells to produce inflammasome-independent IL-1β and cause autoimmunity. Nat. Immunol. 21, 65–74 (2020).
Omoyinmi, E. et al. Th1 and Th17 cell subpopulations are enriched in the peripheral blood of patients with systemic juvenile idiopathic arthritis. Rheumatology 51, 1881–1886 (2012).
Chen, D. Y. et al. Potential role of Th17 cells in the pathogenesis of adult-onset Still’s disease. Rheumatology 49, 2305–2312 (2010).
Chen, D. Y. et al. The associations of circulating CD4+CD25high regulatory T cells and TGF-β with disease activity and clinical course in patients with adult-onset Still’s disease. Connect. Tissue Res. 51, 370–377 (2010).
Henderson, L. A. et al. Th17 reprogramming of T cells in systemic juvenile idiopathic arthritis. JCI Insight 5, e132508 (2020).
Avau, A. et al. Systemic juvenile idiopathic arthritis-like syndrome in mice following stimulation of the immune system with Freund’s complete adjuvant: regulation by interferon-γ. Arthritis Rheumatol. 66, 1340–1351 (2014).
Kessel, C. et al. Proinflammatory cytokine environments can drive interleukin-17 overexpression by γ/δ T cells in systemic juvenile idiopathic arthritis. Arthritis Rheumatol. 69, 1480–1494 (2017).
Kuehn, J. et al. Aberrant naive CD4-positive T cell differentiation in systemic juvenile idiopathic arthritis committed to B cell help. Arthritis Rheumatol. 75, 826–841 (2023).
Fang, X. et al. B cell subsets in adult-onset Still’s disease: potential candidates for disease pathogenesis and immunophenotyping. Arthritis Res. Ther. 25, 104 (2023).
Rosário, C., Zandman-Goddard, G., Meyron-Holtz, E. G., D’Cruz, D. P. & Shoenfeld, Y. The hyperferritinemic syndrome: macrophage activation syndrome, Still’s disease, septic shock and catastrophic antiphospholipid syndrome. BMC Med. 11, 185 (2013).
Schulert, G. S. & Canna, S. W. Convergent pathways of the hyperferritinemic syndromes. Int. Immunol. 30, 195–203 (2018).
Ruscitti, P. et al. Expanding the spectrum of the hyperferritinemic syndrome, from pathogenic mechanisms to clinical observations, and therapeutic implications. Autoimmun. Rev. 21, 103114 (2022).
Ruddell, R. G. et al. Ferritin functions as a proinflammatory cytokine via iron-independent protein kinase C zeta/nuclear factor kappaB-regulated signaling in rat hepatic stellate cells. Hepatology 49, 887–900 (2009).
Ruscitti, P. et al. Pro-inflammatory properties of H-ferritin on human macrophages, ex vivo and in vitro observations. Sci. Rep. 10, 12232 (2020).
Ruscitti, P. et al. H-ferritin and CD68+/H-ferritin+ monocytes/macrophages are increased in the skin of adult-onset Still’s disease patients and correlate with the multi-visceral involvement of the disease. Clin. Exp. Immunol. 186, 30–38 (2016).
Ruscitti, P. et al. The CD68+/H-ferritin+ cells colonize the lymph nodes of the patients with adult onset Still’s disease and are associated with increased extracellular level of H-ferritin in the same tissue: correlation with disease severity and implication for pathogenesis. Clin. Exp. Immunol. 183, 397–404 (2016).
Ruscitti, P. et al. H-ferritin and proinflammatory cytokines are increased in the bone marrow of patients affected by macrophage activation syndrome. Clin. Exp. Immunol. 191, 220–228 (2018).
Zarjou, A. et al. Ferritin light chain confers protection against sepsis-induced inflammation and organ injury. Front. Immunol. 10, 131 (2019).
Jia, J. et al. Ferritin triggers neutrophil extracellular trap-mediated cytokine storm through Msr1 contributing to adult-onset Still’s disease pathogenesis. Nat. Commun. 13, 6804 (2022).
Chen, P. K. et al. Increased lipid peroxidation may be linked to ferritin levels elevation in adult-onset Still’s disease. Biomedicines 9, 1508 (2021).
Fujita, Y. et al. Clinical relevance for circulating cold-inducible RNA-binding protein (CIRP) in patients with adult-onset Still’s disease. PLoS ONE 16, e0255493 (2021).
Berardicurti, O. et al. The similar expression of both ferritin and scavenger receptors activation genes in patients with COVID19 and AOSD support their role in the pathogenesis of these diseases and identify a common mechanism at the basis of the “hyperferritinemic syndromes”. Autoimmun. Rev. 22, 103309 (2023).
Ruscitti, P. et al. The hyper-expression of NLRP4 characterizes the occurrence of macrophage activation syndrome assessing STING pathway in adult-onset Still’s disease. Clin. Exp. Immunol. 208, 95–102 (2022).
Fan, J., Jiang, T. & He, D. Emerging insights into the role of ferroptosis in the pathogenesis of autoimmune diseases. Front. Immunol. 14, 1120519 (2023).
Zhou, G. et al. Retrospective analysis of 1,641 cases of classic fever of unknown origin. Ann. Transl. Med. 8, 690 (2020).
Yachie, A. Clinical perspectives and therapeutic strategies: pediatric autoinflammatory disease-a multi-faceted approach to fever of unknown origin of childhood. Inflamm. Regen. 42, 21 (2022).
Bilgin, E. et al. Proposal for a simple algorithm to differentiate adult-onset Still’s disease with other fever of unknown origin causes: a longitudinal prospective study. Clin. Rheumatol. 38, 1699–1706 (2019).
Ying, S. et al. Development and validation of the AF score for diagnosis of adult-onset Still’s disease in fever of unknown origin. J. Transl. Autoimmun. 6, 100184 (2022).
Daghor-Abbaci, K. et al. Proposal of a new diagnostic algorithm for adult-onset Still’s disease. Clin. Rheumatol. 42, 1125–1135 (2023).
Fautrel, B. Adult-onset Still disease. Best Pract. Res. Clin. Rheumatol. 22, 773–792 (2008).
Ruscitti, P. et al. The joint involvement in adult onset Still’s disease is characterised by a peculiar magnetic resonance imaging and a specific transcriptomic profile. Sci. Rep. 11, 12455 (2021).
Barile, A. et al. Computed tomography and MR imaging in rheumatoid arthritis. Radiol. Clin. North Am. 55, 997–1007 (2017).
Zuelgaray, E. et al. Increased severity and epidermal alterations in persistent versus evanescent skin lesions in adult-onset Still disease. J. Am. Acad. Dermatol. 279, 969–971 (2018).
Ding, Y. et al. Risk of macrophage activation syndrome in patients with adult-onset Still’s disease with skin involvement: a retrospective cohort study. J. Am. Acad. Dermatol. 85, 1503–1509 (2021).
Ruscitti, P. et al. Adult-onset Still’s disease: evaluation of prognostic tools and validation of the systemic score by analysis of 100 cases from three centers. BMC Med. 14, 194 (2016).
Tibaldi, J. et al. Development and initial validation of a composite disease activity score for systemic juvenile idiopathic arthritis. Rheumatology 59, 3505–3514 (2020).
Zhu, D. et al. Performance of the modified Systemic Manifestation Score for systemic juvenile idiopathic arthritis in adult-onset Still’s disease. Clin. Rheumatol. 42, 187–195 (2023).
Di Benedetto, P. et al. Ferritin and C-reactive protein are predictive biomarkers of mortality and macrophage activation syndrome in adult onset Still’s disease. Analysis of the multicentre Gruppo Italiano di Ricerca in Reumatologia Clinica e Sperimentale (GIRRCS) cohort. PLoS ONE 15, e0235326 (2020).
Cron, R. Q., Davi, S., Minoia, F. & Ravelli, A. Clinical features and correct diagnosis of macrophage activation syndrome. Expert Rev. Clin. Immunol. 11, 1043–1053 (2015).
Eloseily, E. M. A. et al. Ferritin to erythrocyte sedimentation rate ratio: simple measure to identify macrophage activation syndrome in systemic juvenile idiopathic arthritis. ACR Open Rheumatol. 1, 345–349 (2019).
Zou, L. X. et al. Clinical and laboratory features, treatment, and outcomes of macrophage activation syndrome in 80 children: a multi-center study in China. World J. Pediatr. 16, 89–98 (2020).
Cush, J. J., Medsger, T. A., Christy, W. C., Herbert, D C. & Cooperstein, L. A. Adult-onset Still’s disease. Clinical course and outcome. Arthritis Rheum. 30, 186–194 (1987).
Maria, A. T. et al. Adult onset Still’s disease (AOSD) in the era of biologic therapies: dichotomous view for cytokine and clinical expressions. Autoimmun. Rev. 13, 1149–1159 (2014).
Vercruysse, F. et al. Adult-onset Still’s disease biological treatment strategy may depend on the phenotypic dichotomy. Arthritis Res. Ther. 21, 53 (2019).
Neau, P. A. et al. The spectrum of Still’s disease: a comparative analysis of phenotypic forms in a cohort of 238 patients. J. Clin. Med. 11, 6703 (2022).
Mitrovic, S. & Fautrel, B. Clinical phenotypes of adult-onset Still’s disease: new insights from pathophysiology and literature findings. J. Clin. Med. 10, 2633 (2021).
Webster, A. J., Gaitskell, K., Turnbull, I., Cairns, B. J. & Clarke, R. Characterisation, identification, clustering, and classification of disease. Sci. Rep. 11, 5405 (2021).
Sugiyama, T. et al. Latent class analysis of 216 patients with adult-onset Still’s disease. Arthritis Res. Ther. 24, 7 (2022).
Li, R. et al. Clinical phenotypes and prognostic factors of adult-onset Still’s disease: data from a large inpatient cohort. Arthritis Res. Ther. 23, 300 (2021).
Wahbi, A. et al. Catastrophic adult-onset Still’s disease as a distinct life-threatening clinical subset: case-control study with dimension reduction analysis. Arthritis Res. Ther. 23, 256 (2021).
Di Cola, I. et al. Adult-onset Still’s disease with elderly onset: results from a multicentre study. Clin. Exp. Rheumatol. 40, 1517–1525 (2022).
Kishida, D. et al. Clinical characteristics and treatment of elderly onset adult-onset Still’s disease. Sci. Rep. 12, 6787 (2022).
Berardicurti, O. et al. Dissecting the clinical heterogeneity of adult-onset Still’s disease: results from a multi-dimensional characterization and stratification. Rheumatology 60, 4844–4849 (2021).
Ruscitti, P. et al. Derivation and validation of four patient clusters in Still’s disease, results from GIRRCS AOSD-study group and AIDA Network Still Disease Registry. RMD Open 9, e003419 (2023).
Ruscitti, P., Berardicurti, O., Giacomelli, R. & Cipriani, P. The clinical heterogeneity of adult onset Still’s disease may underlie different pathogenic mechanisms. Implications for a personalised therapeutic management of these patients. Semin. Immunol. 58, 101632 (2021).
Guo, R. et al. AOSD endotypes based on immune cell profiles: patient stratification with hierarchical clustering analysis. Rheumatology 62, 1636–1644 (2023).
Grom, A. A., Horne, A. & De Benedetti, F. Macrophage activation syndrome in the era of biologic therapy. Nat. Rev. Rheumatol. 12, 259–268 (2016).
Bracaglia, C. et al. Elevated circulating levels of interferon-γ and interferon-γ-induced chemokines characterise patients with macrophage activation syndrome complicating systemic juvenile idiopathic arthritis. Ann. Rheum. Dis. 76, 166–172 (2017).
Chaturvedi, V. et al. T-cell activation profiles distinguish hemophagocytic lymphohistiocytosis and early sepsis. Blood 137, 2337–2346 (2021).
Lin, H. et al. IFN-γ signature in the plasma proteome distinguishes pediatric hemophagocytic lymphohistiocytosis from sepsis and SIRS. Blood Adv. 5, 3457–3467 (2021).
De Benedetti, F., Prencipe, G., Bracaglia, C., Marasco, E. & Grom, A. A. Targeting interferon-γ in hyperinflammation: opportunities and challenges. Nat. Rev. Rheumatol. 17, 678–691 (2021).
Prencipe, G. et al. Neutralization of IFN-γ reverts clinical and laboratory features in a mouse model of macrophage activation syndrome. J. Allergy Clin. Immunol. 141, 1439–1449 (2018).
Girard-Guyonvarc’h, C. et al. Unopposed IL-18 signaling leads to severe TLR9-induced macrophage activation syndrome in mice. Blood 131, 1430–1441 (2018).
Ruscitti, P. et al. Cytokine profile, ferritin and multi-visceral involvement characterize macrophage activation syndrome during adult-onset Still’s disease. Rheumatology 62, 321–329 (2022).
Shimizu, M. et al. Interleukin-18 for predicting the development of macrophage activation syndrome in systemic juvenile idiopathic arthritis. Clin. Immunol. 160, 277–281 (2015).
Inoue, N. et al. Cytokine profile in adult-onset Still’s disease: comparison with systemic juvenile idiopathic arthritis. Clin. Immunol. 169, 8–13 (2016).
Yagishita, M. et al. Clinical features and serum cytokine profiles of elderly-onset adult-onset Still’s disease. Sci. Rep. 12, 21334 (2022).
Saper, V. E. et al. Emergent high fatality lung disease in systemic juvenile arthritis. Ann. Rheum. Dis. 78, 1722–1731 (2019).
Schulert, G. S. et al. Systemic juvenile idiopathic arthritis-associated lung disease: characterization and risk factors. Arthritis Rheumatol. 71, 1943–1954 (2019).
Ruscitti, P. et al. Parenchymal lung disease in adult onset Still’s disease: an emergent marker of disease severity-characterisation and predictive factors from Gruppo Italiano di Ricerca in Reumatologia Clinica e Sperimentale (GIRRCS) cohort of patients. Arthritis Res. Ther. 22, 151 (2020).
Saper, V. E. et al. Severe delayed hypersensitivity reactions to IL-1 and IL-6 inhibitors link to common HLA-DRB1*15 alleles. Ann. Rheum. Dis. 81, 406–415 (2022).
Wobma, H. et al. Incidence and risk factors for eosinophilia and lung disease in biologic-exposed children with systemic juvenile idiopathic arthritis. Arthritis Care Res. 75, 2063–2072 (2023).
Lerman, A. M. et al. HLA-DRB1*15 and eosinophilia are common among patients with systemic juvenile idiopathic arthritis. Arthritis Care Res. 75, 2082–2087 (2023).
Huang, Y. et al. Disease course, treatments, and outcomes of children with systemic juvenile idiopathic arthritis associated lung disease (SJIA-LD). Arthritis Care Res., https://doi.org/10.1002/acr.25234 (2023).
Binstadt, B. A. & Nigrovic, P. A. The conundrum of lung disease and drug hypersensitivity-like reactions in systemic juvenile idiopathic arthritis. Arthritis Rheumatol. 74, 1122–1131 (2022).
Chen, G. et al. Identification of distinct inflammatory programs and biomarkers in systemic juvenile idiopathic arthritis and related lung disease by serum proteome analysis. Arthritis Rheumatol. 74, 1271–1283 (2022).
Malengier-Devlies, B. et al. Lung functioning and inflammation in a mouse model of systemic juvenile idiopathic arthritis. Front. Immunol. 12, 642778 (2021).
Ruscitti, P. et al. Managing adult-onset Still’s disease: the effectiveness of high-dosage of corticosteroids as first-line treatment in inducing the clinical remission. Results from an observational study. Medicine 98, e15123 (2019).
Kondo, F. et al. Associated factors with poor treatment response to initial glucocorticoid therapy in patients with adult-onset Still’s disease. Arthritis Res. Ther. 24, 92 (2022).
Cipriani, P., Ruscitti, P., Carubbi, F., Liakouli, V. & Giacomelli, R. Methotrexate: an old new drug in autoimmune disease. Expert Rev. Clin. Immunol. 10, 1519–1530 (2014).
Nakamura, H. et al. Calcineurin inhibitors for adult-onset Still’s disease: a multicentre retrospective cohort study. Clin. Exp. Rheumatol. 38, 11–16 (2020).
Erkens, R., Esteban, Y., Towe, C., Schulert, G. & Vastert, S. Pathogenesis and treatment of refractory disease courses in systemic juvenile idiopathic arthritis: refractory arthritis, recurrent macrophage activation syndrome and chronic lung disease. Rheum. Dis. Clin. North Am. 47, 585–606 (2021).
Fautrel, B. et al. Systematic review on the use of biologics in adult-onset Still’s disease. Semin. Arthritis Rheum. 58, 152139 (2023).
Vastert, S. J. et al. Anakinra in children and adults with Still’s disease. Rheumatology 58, vi9–vi22 (2019).
Mejbri, M., Theodoropoulou, K., Hofer, M. & Cimaz, R. Interleukin-1 blockade in systemic juvenile idiopathic arthritis. Paediatr. Drugs 22, 251–262 (2020).
Giacomelli, R. et al. The treatment of adult-onset Still’s disease with anakinra, a recombinant human IL-1 receptor antagonist: a systematic review of literature. Clin. Exp. Rheumatol. 39, 187–195 (2021).
Giancane, G. et al. Anakinra in patients with systemic juvenile idiopathic arthritis: long-term safety from the Pharmachild Registry. J. Rheumatol. 49, 398–407 (2022).
Ruscitti, P. et al. The reduction of concomitant glucocorticoids dosage following treatment with IL-1 receptor antagonist in adult onset Still’s disease. A systematic review and meta-analysis of observational studies. Ther. Adv. Musculoskelet. Dis. 12, 1759720X20933133 (2020).
Pardeo, M. et al. Anakinra in systemic juvenile idiopathic arthritis: a single-center experience. J. Rheumatol. 42, 1523–1527 (2015).
Nigrovic, P. A. et al. Anakinra as first-line disease-modifying therapy in systemic juvenile idiopathic arthritis: report of forty-six patients from an international multicenter series. Arthritis Rheum. 63, 545–555 (2011).
Pardeo, M. et al. Early treatment and IL1RN single-nucleotide polymorphisms affect response to anakinra in systemic juvenile idiopathic arthritis. Arthritis Rheumatol. 73, 1053–1061 (2021).
Vastert, S. J. et al. Effectiveness of first-line treatment with recombinant interleukin-1 receptor antagonist in steroid-naive patients with new-onset systemic juvenile idiopathic arthritis: results of a prospective cohort study. Arthritis Rheumatol. 66, 1034–1043 (2014).
Ter Haar, N. M. et al. Treatment to target using recombinant interleukin-1 receptor antagonist as first-line monotherapy in new-onset systemic juvenile idiopathic arthritis: results from a five-year follow-up study. Arthritis Rheumatol. 71, 1163–1173 (2019).
Erkens, R. G. A. et al. Recombinant interleukin-1 receptor antagonist is an effective first-line treatment strategy in new-onset systemic Juvenile Idiopathic Arthritis, irrespective of HLA-DRB1 background and IL1RN variants. Arthritis Rheumatol., https://doi.org/10.1002/art.42656 (2023).
Arthur, V. L. et al. IL1RN variation influences both disease susceptibility and response to recombinant human interleukin-1 receptor antagonist therapy in systemic juvenile idiopathic arthritis. Arthritis Rheumatol. 70, 1319–1330 (2018).
Kedor, C., Tomaras, S., Baeumer, D. & Feist, E. Update on the therapy of adult-onset Still’s disease with a focus on IL-1-inhibition: a systematic review. Ther. Adv. Musculoskelet. Dis. 13, 1759720X211059598 (2021).
Ruperto, N. et al. Two randomized trials of canakinumab in systemic juvenile idiopathic arthritis. N. Engl. J. Med. 367, 2396–2406 (2012).
Ruperto, N. et al. Canakinumab in patients with systemic juvenile idiopathic arthritis and active systemic features: results from the 5-year long-term extension of the phase III pivotal trials. Ann. Rheum. Dis. 77, 1710–1719 (2018).
Alexeeva, E. et al. Efficacy and safety of canakinumab as a second line biologic after tocilizumab treatment failure in children with systemic juvenile idiopathic arthritis: a single-centre cohort study using routinely collected health data. Front. Pediatr. 11, 1114207 (2023).
Quartier, P. et al. Tapering canakinumab monotherapy in patients with systemic juvenile idiopathic arthritis in clinical remission: results from a phase IIIb/IV open-label, randomized study. Arthritis Rheumatol. 73, 336–346 (2021).
Kedor, C. et al. Canakinumab for treatment of adult-onset Still’s disease to achieve reduction of arthritic manifestation (CONSIDER): phase II, randomised, double-blind, placebo-controlled, multicentre, investigator-initiated trial. Ann. Rheum. Dis. 79, 1090–1097 (2020).
Laskari, K. et al. Outcome of refractory to conventional and/or biologic treatment adult Still’s disease following canakinumab treatment: countrywide data in 50 patients. Semin. Arthritis Rheum. 51, 137–143 (2021).
Vitale, A. et al. Canakinumab as first-line biological therapy in Still’s disease and differences between the systemic and the chronic-articular courses: real-life experience from the international AIDA registry. Front. Med. 9, 1071732 (2022).
Ghannam, K. et al. Distinct effects of interleukin-1β inhibition upon cytokine profile in patients with adult-onset Still’s disease and active articular manifestation responding to canakinumab. J. Clin. Med. 10, 4400 (2021).
Brachat, A. et al. Early changes in gene expression and inflammatory proteins in systemic juvenile idiopathic arthritis patients on canakinumab therapy. Arthritis Res. Ther. 19, 13 (2017).
Verweyen, E. L., Pickering, A., Grom, A. A. & Schulert, G. S. Distinct gene expression signatures characterize strong clinical responders versus nonresponders to canakinumab in children with systemic juvenile idiopathic arthritis. Arthritis Rheumatol. 73, 1334–1340 (2021).
Galozzi, P., Bindoli, S., Doria, A. & Sfriso, P. The revisited role of interleukin-1 alpha and beta in autoimmune and inflammatory disorders and in comorbidities. Autoimmun. Rev. 20, 102785 (2021).
Choy, E. H. et al. Translating IL-6 biology into effective treatments. Nat. Rev. Rheumatol. 16, 335–345 (2020).
De Benedetti, F. et al. Randomized trial of tocilizumab in systemic juvenile idiopathic arthritis. N. Engl. J. Med. 367, 2385–2395 (2012).
Kaneko, Y. et al. Tocilizumab in patients with adult-onset still’s disease refractory to glucocorticoid treatment: a randomised, double-blind, placebo-controlled phase III trial. Ann. Rheum. Dis. 77, 1720–1729 (2018).
Pacharapakornpong, T., Vallibhakara, S. A.-O., Lerkvaleekul, B. & Vilaiyuk, S. Comparisons of the outcomes between early and late tocilizumab treatment in systemic juvenile idiopathic arthritis. Rheumatol. Int. 37, 251–255 (2017).
Yan, X. et al. Tocilizumab in systemic juvenile idiopathic arthritis: response differs by disease duration at medication initiation and by phenotype of disease. Front. Pediatr. 9, 735846 (2021).
Nada, D. W., Moghazy, A., El-Sayed Allam, A., Alunno, A. & Ibrahim, A. M. Short-term outcomes and predictors of effectiveness of tocilizumab in systemic juvenile idiopathic arthritis: a prospective cohort study. Front. Med. 8, 665028 (2021).
Kaneko, Y. & Takeuchi, T. Interleukin-6 inhibition: a therapeutic strategy for the management of adult-onset Still’s disease. Expert Opin. Biol. Ther. 22, 79–85 (2022).
Sota, J. et al. Efficacy and safety of tocilizumab in adult-onset Still’s disease: real-life experience from the international AIDA registry. Semin. Arthritis Rheum. 57, 152089 (2022).
Locatelli, F. et al. Emapalumab in children with primary hemophagocytic lymphohistiocytosis. N. Engl. J. Med. 382, 1811–1822 (2020).
De Benedetti, F. et al. Efficacy and safety of emapalumab in macrophage activation syndrome. Ann. Rheum. Dis. 82, 857–865 (2023).
Tanaka, Y., Luo, Y., O’Shea, J. J. & Nakayamada, S. Janus kinase-targeting therapies in rheumatology: a mechanisms-based approach. Nat. Rev. Rheumatol. 18, 133–145 (2022).
Favoino, E. et al. Working and safety profiles of JAK/STAT signaling inhibitors. Are these small molecules also smart? Autoimmun. Rev. 20, 102750 (2021).
He, T., Xia, Y., Luo, Y. & Yang, J. JAK inhibitors in systemic juvenile idiopathic arthritis. Front. Pediatr. 11, 1134312 (2023).
Hu, Q. et al. Tofacitinib in refractory adult-onset Still’s disease: 14 cases from a single centre in China. Ann. Rheum. Dis. 79, 842–844 (2020).
Gillard, L. et al. JAK inhibitors in difficult-to-treat adult-onset Still’s disease and systemic-onset juvenile idiopathic arthritis. Rheumatology 62, 1594–1604 (2023).
Furuya, M. Y. et al. Tofacitinib inhibits granulocyte-macrophage colony-stimulating factor-induced NLRP3 inflammasome activation in human neutrophils. Arthritis Res. Ther. 20, 196 (2018).
Ahmed, A. et al. Ruxolitinib in adult patients with secondary haemophagocytic lymphohistiocytosis: an open-label, single-centre, pilot trial. Lancet Haematol. 6, e630–e637 (2019).
Wang, J. et al. Ruxolitinib-combined doxorubicin-etoposide-methylprednisolone regimen as a salvage therapy for refractory/relapsed haemophagocytic lymphohistiocytosis: a single-arm, multicentre, phase 2 trial. Br. J. Haematol. 193, 761–768 (2021).
Keenan, C., Nichols, K. E. & Albeituni, S. Use of the JAK inhibitor ruxolitinib in the treatment of hemophagocytic lymphohistiocytosis. Front. Immunol. 12, 614704 (2021).
Yang, L. et al. The signal pathways and treatment of cytokine storm in COVID-19. Signal Transduct. Target. Ther. 6, 255 (2021).
Dessie, Z. G. & Zewotir, T. Mortality-related risk factors of COVID-19: a systematic review and meta-analysis of 42 studies and 423,117 patients. BMC Infect. Dis. 21, 855 (2021).
Ruscitti, P. et al. Severe COVID-19, another piece in the puzzle of the hyperferritinemic syndrome. An immunomodulatory perspective to alleviate the storm. Front. Immunol. 11, 1130 (2020).
Ruscitti, P. et al. Lung involvement in macrophage activation syndrome and severe COVID-19: results from a cross-sectional study to assess clinical, laboratory and artificial intelligence-radiological differences. Ann. Rheum. Dis. 79, 1152–1155 (2020).
GTEx Consortium. Human genomics. The Genotype-Tissue Expression (GTEx) pilot analysis: multitissue gene regulation in humans. Science 348, 648–660 (2015).
McGonagle, D., Sharif, K., O’Regan, A. & Bridgewood, C. The role of cytokines including interleukin-6 in COVID-19 induced pneumonia and macrophage activation syndrome-like disease. Autoimmun. Rev. 19, 102537 (2020).
Sposito, B. et al. The interferon landscape along the respiratory tract impacts the severity of COVID-19. Cell 184, 4953–4968.e16 (2021).
Fajgenbaum, D. C. & June, C. H. Cytokine storm. N. Engl. J. Med. 383, 2255–2273 (2020).
Diamond, M. S., Lambris, J. D., Ting, J. P. & Tsang, J. S. Considering innate immune responses in SARS-CoV-2 infection and COVID-19. Nat. Rev. Immunol. 22, 465–470 (2022).
Vora, S. M., Lieberman, J. & Wu, H. Inflammasome activation at the crux of severe COVID-19. Nat. Rev. Immunol. 21, 694–703 (2021).
McGonagle, D., O’Donnell, J. S., Sharif, K., Emery, P. & Bridgewood, C. Immune mechanisms of pulmonary intravascular coagulopathy in COVID-19 pneumonia. Lancet Rheumatol. 2, e437–e445 (2020).
McGonagle, D., Bridgewood, C., Ramanan, A. V., Meaney, J. F. M. & Watad, A. COVID-19 vasculitis and novel vasculitis mimics. Lancet Rheumatol. 3, e224–e233 (2021).
Giryes, S., Bragazzi, N. L., Bridgewood, C., De Marco, G. & McGonagle, D. COVID-19 vasculitis and vasculopathy-distinct immunopathology emerging from the close juxtaposition of type II pneumocytes and pulmonary endothelial cells. Semin. Immunopathol. 44, 375–390 (2022).
McGonagle, D. et al. Therapeutic implications of ongoing alveolar viral replication in COVID-19. Lancet Rheumatol. 4, e135–e144 (2022).
van de Veerdonk, F. L. et al. A guide to immunotherapy for COVID-19. Nat. Med. 28, 39–50 (2022).
Potere, N. et al. Interleukin-1 and the NLRP3 inflammasome in COVID-19: pathogenetic and therapeutic implications. EBioMedicine 85, 104299 (2022).
Zizzo, G. et al. Immunotherapy of COVID-19: inside and beyond IL-6 signalling. Front. Immunol. 13, 795315 (2022).
Matuozzo, D. et al. Rare predicted loss-of-function variants of type I IFN immunity genes are associated with life-threatening COVID-19. Genome Med. 15, 22 (2023).
Zhang, Q. et al. Inborn errors of type I IFN immunity in patients with life-threatening COVID-19. Science 370, eabd4570 (2020).
Zhang, Q. et al. Human genetic and immunological determinants of critical COVID-19 pneumonia. Nature 603, 587–598 (2022).
Bastard, P. et al. Autoantibodies against type I IFNs in patients with life-threatening COVID-19. Science 370, eabd4585 (2020).
Pfeifer, J. et al. Autoantibodies against interleukin-1 receptor antagonist in multisystem inflammatory syndrome in children: a multicentre, retrospective, cohort study. Lancet Rheumatol. 4, e329–e337 (2022).
Ursini, F. et al. Spectrum of short-term inflammatory musculoskeletal manifestations after COVID-19 vaccine administration: a report of 66 cases. Ann. Rheum. Dis. 81, 440–441 (2022).
Chen, C. C. & Chen, C.-J. New-onset inflammatory arthritis after COVID-19 vaccination: a systematic review. Int. J. Rheum. Dis. 26, 267–277 (2023).
Scholkmann, F. & May, C.-A. COVID-19, post-acute COVID-19 syndrome (PACS, “long COVID”) and post-COVID-19 vaccination syndrome (PCVS, “post-COVIDvac-syndrome”): similarities and differences. Pathol. Res. Pract. 246, 154497 (2023).
Palassin, P. et al. Comprehensive description of adult-onset Still’s disease after COVID-19 vaccination. J. Autoimmun. 134, 102980 (2023).
Tahtinen, S. et al. IL-1 and IL-1ra are key regulators of the inflammatory response to RNA vaccines. Nat. Immunol. 23, 532–542 (2022).
Giacomelli, R. et al. International consensus: what else can we do to improve diagnosis and therapeutic strategies in patients affected by autoimmune rheumatic diseases (rheumatoid arthritis, spondyloarthritides, systemic sclerosis, systemic lupus erythematosus, antiphospholipid syndrome and Sjogren’s syndrome)? the unmet needs and the clinical grey zone in autoimmune disease management. Autoimmun. Rev. 16, 911–924 (2017).
Inoue, N. & Schulert, G. S. Mouse models of systemic juvenile idiopathic arthritis and macrophage activation syndrome. Arthritis Res. Ther. 25, 48 (2023).
Mizuta, M. et al. Distinct roles of IL-18 and IL-1β in murine model of macrophage activation syndrome. J. Allergy Clin. Immunol. 152, 940–948 (2023).
Chi, H. et al. The landscape of innate and adaptive immune cell subsets in patients with adult-onset Still’s disease. Rheumatology, https://doi.org/10.1093/rheumatology/kead507 (2023).
Mitrovic, S. et al. Adult-onset Still’s disease or systemic-onset juvenile idiopathic arthritis and spondyloarthritis: overlapping syndrome or phenotype shift? Rheumatology 61, 2535–2547 (2022).
Giacomelli, R. et al. Guidelines for biomarkers in autoimmune rheumatic diseases — evidence based analysis. Autoimmun. Rev. 18, 93–106 (2019).
Mitrovic, S. & Fautrel, B. New markers for adult-onset Still’s disease. Jt Bone Spine 85, 285–293 (2018).
Colafrancesco, S., Priori, R. & Valsini, G. Presentation and diagnosis of adult-onset Still’s disease: the implications of current and emerging markers in overcoming the diagnostic challenge. Expert Rev. Clin. Immunol. 11, 749–761 (2015).
Chen, P. K., Wey, S. J. & Chen, D. Y. Interleukin-18: a biomarker with therapeutic potential in adult-onset Still’s disease. Expert Rev. Clin. Immunol. 18, 823–833 (2022).
Manfredi, M. et al. Circulating calprotectin (cCLP) in autoimmune diseases. Autoimmun. Rev. 22, 103295 (2023).
Leavis, H. L. et al. Management of adult-onset Still’s disease: evidence- and consensus-based recommendations by experts. Rheumatology, https://doi.org/10.1093/rheumatology/kead461 (2023).
Giacomelli, R. et al. Expert consensus on the treatment of patients with adult-onset still’s disease with the goal of achieving an early and long-term remission. Autoimmun. Rev. 22, 103400 (2023).
Schett, G. et al. Tapering biologic and conventional DMARD therapy in rheumatoid arthritis: current evidence and future directions. Ann. Rheum. Dis. 75, 1428–1437 (2016).
Giacomelli, R. et al. The growing role of precision medicine for the treatment of autoimmune diseases; results of a systematic review of literature and Experts’ Consensus. Autoimmun. Rev. 20, 102738 (2021).
Quartier, P. Systemic juvenile idiopathic arthritis/pediatric Still’s disease, a syndrome but several clinical forms: recent therapeutic approaches. J. Clin. Med. 11, 1357 (2022).
Rood, J. E. et al. Improvement of refractory systemic juvenile idiopathic arthritis-associated lung disease with single-agent blockade of IL-1β and IL-18. J. Clin. Immunol. 43, 101–108 (2023).
Girard-Guyonvarc’h, C., Harel, M. & Gabay, C. The role of interleukin 18/interleukin 18-binding protein in adult-onset Still’s disease and systemic juvenile idiopathic arthritis. J. Clin. Med. 11, 430 (2022).
Ambler, W. G., Nanda, K., Onel, C. B. & Shenoi, S. Refractory systemic onset juvenile idiopathic arthritis: current challenges and future perspectives. Ann. Med. 54, 1839–1850 (2022).
Ma, Y. et al. Current and emerging biological therapy in adult-onset Still’s disease. Rheumatology 60, 3986–4000 (2021).
Ruscitti, P. et al. Macrophage activation syndrome in patients affected by adult-onset still disease: analysis of survival rates and predictive factors in the Gruppo Italiano di Ricerca in Reumatologia Clinica e Sperimentale Cohort. J. Rheumatol. 45, 864–872 (2018).
Ruscitti, P. et al. Macrophage activation syndrome in Still’s disease: analysis of clinical characteristics and survival in paediatric and adult patients. Clin. Rheumatol. 36, 2839–2845 (2017).
Ruscitti, P. et al. Prognostic factors of macrophage activation syndrome, at the time of diagnosis, in adult patients affected by autoimmune disease: analysis of 41 cases collected in 2 rheumatologic centers. Autoimmun. Rev. 16, 16–21 (2017).
Ruscitti, P. et al. Impact of smoking habit on adult-onset Still’s disease prognosis, findings from a multicentre observational study. Clin. Rheumatol. 41, 641–647 (2022).
Di Cola, I. et al. Clinical characteristics of obese patients with adult-onset Still’s disease. Data from a large multicentre cohort. Jt. Bone Spine 18, 105576 (2023).
Lenert, A., Oh, G., Ombrello, M. J. & Kim, S. Clinical characteristics and comorbidities in adult-onset Still’s disease using a large US administrative claims database. Rheumatology 59, 1725–1733 (2020).
Meng, J. et al. Characteristics and risk factors of relapses in patients with adult-onset Still’s disease: a long-term cohort study. Rheumatology 60, 4520–4529 (2021).
Mitrovic, S. et al. Clinical and biological characteristics of children and adults affected with Still’s disease: a systematic review and meta-analysis informing the 2023 EULAR/PReS recommendations for the diagnosis and management of systemic juvenile idiopathic arthritis and adult-onset Still’s disease. Arthritis Rheumatol. 75 (Suppl. 9), Abstr. 0758 (2023).
Bindoli, S. et al. Efficacy and safety of therapeutic interventions for the treatment of Still’s disease: a systematic review and meta-analysis informing the EULAR/PReS recommendations for the diagnosis and management of systemic juvenile idiopathic arthritis and adult-onset Still’s disease? Arthritis Rheumatol. 75 (Suppl. 9), Abstr. 2569 (2023).
Fautrel, B. et al. EULAR/PreS recommendations for the diagnosis and management of systemic juvenile idiopathic arthritis (sJIA) and adult onset Still’s disease (AOSD). Arthritis Rheumatol. 75 (Suppl. 9), Abstr. 0761 (2023).
Fardet, L. et al. Development and validation of the HScore, a score for the diagnosis of reactive hemophagocytic syndrome. Arthritis Rheumatol. 66, 2613–2620 (2014).
Minoia, F. et al. Development and initial validation of the MS score for diagnosis of macrophage activation syndrome in systemic juvenile idiopathic arthritis. Ann. Rheum. Dis. 78, 1357–1362 (2019).
Wobma, H. et al. Development of a screening algorithm for lung disease in systemic juvenile idiopathic arthritis. ACR Open Rheumatol. 5, 556–562 (2023).
Vitale, A. et al. Still’s disease continuum from childhood to elderly: data from the international AIDA Network Still’s disease registry. RMD Open 9, e003578 (2023).
Colafrancesco, S. et al. Management of adult-onset Still’s disease with interleukin-1 inhibitors: evidence- and consensus-based statements by a panel of Italian experts. Arthritis Res. Ther. 21, 275 (2019).
Ursini, F. et al. Care pathway analysis and evidence gaps in adult-onset Still’s disease: interviews with experts from the UK, France, Italy, and Germany. Front. Med. 10, 1257413 (2023).
Acknowledgements
The authors thank M. Vomero for her assistance in creating the figures. P.A.N. was funded by NIH grants R01AR073201 and P30AR070253.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
P.A.N. discloses consulting for Apollo, BMS, Exo Therapeutics, Fresh Tracks Therapeutics, Merck, Novartis, Pfizer, Qiagen and Sobi; investigator-initiated research grants from BMS and Pfizer; and authorship royalties from UpToDate, the American Academy of Paediatrics and Arthritis & Rheumatology. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Rheumatology thanks Qiongyi Hu, Masaki Shimizu, Chang Hee Suh and Chengde Yang for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ruscitti, P., Cantarini, L., Nigrovic, P.A. et al. Recent advances and evolving concepts in Still’s disease. Nat Rev Rheumatol 20, 116–132 (2024). https://doi.org/10.1038/s41584-023-01065-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41584-023-01065-6