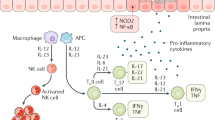
Abstract
Nearly all patients with systemic sclerosis (SSc) are negatively affected by dysfunction in the gastrointestinal tract, and the severity of gastrointestinal disease in SSc correlates with high mortality. The clinical complications of this dysfunction are heterogeneous and include gastro-oesophageal reflux disease, gastroparesis, small intestinal bacterial overgrowth, intestinal pseudo-obstruction, malabsorption and the requirement for total parenteral nutrition. The abnormal gastrointestinal physiology that promotes the clinical manifestations of SSc gastrointestinal disease throughout the gastrointestinal tract are diverse and present a range of therapeutic targets. Furthermore, the armamentarium of medications and non-pharmacological interventions that can benefit affected patients has substantially expanded in the past 10 years, and research is increasingly focused in this area. Here, we review the details of the gastrointestinal complications in SSc, tie physiological abnormalities to clinical manifestations, detail the roles of standard and novel therapies and lay a foundation for future investigative work.
Key points
-
Gastrointestinal disease in systemic sclerosis (SSc) is complex in its clinical presentation, physiology and mechanisms.
-
Data suggest that the neuromuscular pathways that control gastrointestinal motility are dysfunctional in SSc and are a target of the autoimmune response in some patients.
-
Identifying the part or parts of the gut affected and the type of gastrointestinal abnormality that is present can help to guide therapy.
-
The microbiome is probably an important contributor to gastrointestinal symptoms in SSc, although the extent of its involvement and the role of dysbiosis in diagnosis and guiding therapy are unclear.
-
Research is underway to better understand the mechanisms of gastrointestinal disease in SSc, and to optimize the approach to management.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Change history
15 February 2023
A Correction to this paper has been published: https://doi.org/10.1038/s41584-023-00929-1
References
Steen, V. D. & Medsger, T. A. Severe organ involvement in systemic sclerosis with diffuse scleroderma. Arthritis Rheum. 43, 2437–2444 (2000).
Richard, N. et al. Severe gastrointestinal disease in very early systemic sclerosis is associated with early mortality. Rheumatology 58, 636–644 (2019).
Gyger, G. & Baron, M. Systemic sclerosis: gastrointestinal disease and its management. Rheum. Dis. Clin. North. Am. 41, 459–473 (2015).
Kaniecki, T., Abdi, T. & McMahan, Z. H. A practical approach to the evaluation and management of gastrointestinal symptoms in patients with systemic sclerosis. Best Pract. Res. Clin. Rheumatol. 35, 101666 (2021).
Jaovisidha, K., Csuka, M. E., Almagro, U. A. & Soergel, K. H. Severe gastrointestinal involvement in systemic sclerosis: report of five cases and review of the literature. Semin. Arthritis Rheum. 34, 689–702 (2005).
McMahan, Z. H. et al. Relationship between gastrointestinal transit, Medsger gastrointestinal severity, and University of California-Los Angeles scleroderma clinical trial consortium gastrointestinal tract 2.0 symptoms in patients with systemic sclerosis. Arthritis Care Res. 74, 442–450 (2022).
Kawaguchi, Y. et al. Muscarinic-3 acetylcholine receptor autoantibody in patients with systemic sclerosis: contribution to severe gastrointestinal tract dysmotility. Ann. Rheum. Dis. 68, 710–714 (2009).
Lock, G. et al. Association of autonomic nervous dysfunction and esophageal dysmotility in systemic sclerosis. J. Rheumatol. 25, 1330–1335 (1998).
Adler, B. L., Russell, J. W., Hummers, L. K. & McMahan, Z. H. Symptoms of autonomic dysfunction in systemic sclerosis assessed by the COMPASS-31 questionnaire. J. Rheumatol. 45, 1145–1152 (2018).
Furness, J. B., Callaghan, B. P., Rivera, L. R. & Cho, H. J. The enteric nervous system and gastrointestinal innervation: integrated local and central control. Adv. Exp. Med. Biol. 817, 39–71 (2014).
Kulkarni, S. et al. Adult enteric nervous system in health is maintained by a dynamic balance between neuronal apoptosis and neurogenesis. Proc. Natl Acad. Sci. USA 114, E3709–E3718 (2017).
Kulkarni, S. et al. Advances in enteric neurobiology: the “brain” in the gut in health and disease. J. Neurosci. 38, 9346–9354 (2018).
Kulkarni, S. & Pasricha, P. J. Decoding the enteric nervous system: the beginning of our understanding of enteric neuromuscular disorders. Gastroenterology 160, 651–652 (2021).
McMahan, Z. H. et al. Anti-RNPC-3 (U11/U12) antibodies in systemic sclerosis in patients with moderate-to-severe gastrointestinal dysmotility. Arthritis Care Res. 71, 1164–1170 (2019).
McMahan, Z. H., Paik, J. J., Wigley, F. M. & Hummers, L. K. Determining the risk factors and clinical features associated with severe gastrointestinal dysmotility in systemic sclerosis. Arthritis Care Res. 70, 1385–1392 (2018).
Dein, E. et al. Evaluation of risk factors for pseudo-obstruction in systemic sclerosis. Semin. Arthritis Rheum. 49, 405–410 (2019).
Ziessman, H. A., Jeyasingam, M., Khan, A. U., McMahan, Z. & Pasricha, P. J. Experience with esophagogastrointestinal transit scintigraphy in the initial 229 patients: multiple regions of dysmotility are common. J. Nucl. Med. 62, 115–122 (2021).
Vigone, B. et al. Preliminary safety and efficacy profile of prucalopride in the treatment of systemic sclerosis (SSc)-related intestinal involvement: results from the open label cross-over PROGASS study. Arthritis Res. Ther. 19, 145 (2017).
Karamanolis, G. P. et al. The 5-HT1A receptor agonist buspirone improves esophageal motor function and symptoms in systemic sclerosis: a 4-week, open-label trial. Arthritis Res. Ther. 18, 195 (2016).
McMahan, Z. H. & Khanna, D. Managing gastrointestinal complications in patients with systemic sclerosis. Curr. Treat. Options Gastroenterol. 18, 531–544 (2020).
Rigamonti, C. et al. Clinical features and prognosis of primary biliary cirrhosis associated with systemic sclerosis. Gut 55, 388–394 (2006).
Morrisroe, K. et al. Gastric antral vascular ectasia in systemic sclerosis: a study of its epidemiology, disease characteristics and impact on survival. Arthritis Res. Ther. 24, 103 (2022).
Drokhlyansky, E. et al. The human and mouse enteric nervous system at single-cell resolution. Cell 182, 1606–1622.e1623 (2020).
Morarach, K. et al. Diversification of molecularly defined myenteric neuron classes revealed by single-cell RNA sequencing. Nat. Neurosci. 24, 34–46 (2021).
Jarret, A. et al. Enteric nervous system-derived IL-18 orchestrates mucosal barrier immunity. Cell 180, 813–814 (2020).
Muller, P. A. et al. Crosstalk between muscularis macrophages and enteric neurons regulates gastrointestinal motility. Cell 158, 300–313 (2014).
Nickerson, A. J., Rottgen, T. S. & Rajendran, V. M. Activation of KCNQ (KV7) K+ channels in enteric neurons inhibits epithelial Cl– secretion in mouse distal colon. Am. J. Physiol. Cell Physiol. 320, C1074–C1087 (2021).
Moreno, S. et al. Epithelial propionyl- and butyrylcholine as novel regulators of colonic ion transport. Br. J. Pharmacol. 173, 2766–2779 (2016).
Fung, C. & Vanden Berghe, P. Functional circuits and signal processing in the enteric nervous system. Cell Mol. Life Sci. 77, 4505–4522 (2020).
Phillips, R. J. & Powley, T. L. Innervation of the gastrointestinal tract: patterns of aging. Auton. Neurosci. 136, 1–19 (2007).
Kang, Y. N., Fung, C. & Vanden Berghe, P. Gut innervation and enteric nervous system development: a spatial, temporal and molecular tour de force. Development 148, dev182543 (2021).
Duan, H. et al. Regulation of the autonomic nervous system on intestine. Front. Physiol. 12, 700129 (2021).
Powley, T. L. Brain-gut communication: vagovagal reflexes interconnect the two “brains”. Am. J. Physiol. Gastrointest. Liver Physiol. 321, G576–G587 (2021).
Masliukov, P. M., Emanuilov, A. I. & Budnik, A. F. Sympathetic innervation of the development, maturity, and aging of the gastrointestinal tract. Anat. Rec. https://doi.org/10.1002/ar.25015 (2022).
Mercado-Perez, A. & Beyder, A. Gut feelings: mechanosensing in the gastrointestinal tract. Nat. Rev. Gastroenterol. Hepatol. 19, 283–296 (2022).
Spencer, N. J. & Hu, H. Enteric nervous system: sensory transduction, neural circuits and gastrointestinal motility. Nat. Rev. Gastroenterol. Hepatol. 17, 338–351 (2020).
Kulkarni, S., Kurapati, S. & Bogunovic, M. Neuro-innate immune interactions in gut mucosal immunity. Curr. Opin. Immunol. 68, 64–71 (2021).
Dowling, L. R., Strazzari, M. R., Keely, S. & Kaiko, G. E. Enteric nervous system and intestinal epithelial regulation of the gut-brain axis. J. Allergy Clin. Immunol. 150, 513–522 (2022).
Vaes, N., Idris, M., Boesmans, W., Alves, M. M. & Melotte, V. Nerves in gastrointestinal cancer: from mechanism to modulations. Nat. Rev. Gastroenterol. Hepatol. 19, 768–784 (2022).
Holland, A. M., Bon-Frauches, A. C., Keszthelyi, D., Melotte, V. & Boesmans, W. The enteric nervous system in gastrointestinal disease etiology. Cell Mol. Life Sci. 78, 4713–4733 (2021).
Agirman, G., Yu, K. B. & Hsiao, E. Y. Signaling inflammation across the gut-brain axis. Science 374, 1087–1092 (2021).
Zhao, Q. et al. A multidimensional coding architecture of the vagal interoceptive system. Nature 603, 878–884 (2022).
Lagomarsino, V. N., Kostic, A. D. & Chiu, I. M. Mechanisms of microbial-neuronal interactions in pain and nociception. Neurobiol. Pain. 9, 100056 (2021).
Lai, N. Y. et al. Gut-innervating nociceptor neurons regulate Peyer’s patch microfold cells and SFB levels to mediate Salmonella host defense. Cell 180, 33–49 e22 (2020).
Lai, N. Y., Mills, K. & Chiu, I. M. Sensory neuron regulation of gastrointestinal inflammation and bacterial host defence. J. Intern. Med. 282, 5–23 (2017).
Chang, H. Y., Mashimo, H. & Goyal, R. K. IV Current concepts of vagal efferent projections to the gut. Am. J. Physiol. Gastrointest. Liver Physiol. 284, G357–G366 (2003).
Matzel, K. E., Stadelmaier, U., Hohenfellner, M. & Gall, F. P. Electrical stimulation of sacral spinal nerves for treatment of faecal incontinence. Lancet 346, 1124–1127 (1995).
Willemze, R. A. et al. Loss of intestinal sympathetic innervation elicits an innate immune driven colitis. Mol. Med. 25, 1 (2019).
Muller, P. A. et al. Microbiota modulate sympathetic neurons via a gut–brain circuit. Nature 583, 441–446 (2020).
Schirmer, M. et al. Linking the human gut microbiome to inflammatory cytokine production capacity. Cell 167, 1125–1136.e1128 (2016).
Sato, Y. et al. Novel bile acid biosynthetic pathways are enriched in the microbiome of centenarians. Nature 599, 458–464 (2021).
Sjogren, R. W. Gastrointestinal motility disorders in scleroderma. Arthritis Rheum. 37, 1265–1282 (1994).
Roberts, C. G., Hummers, L. K., Ravich, W. J., Wigley, F. M. & Hutchins, G. M. A case-control study of the pathology of oesophageal disease in systemic sclerosis (scleroderma). Gut 55, 1697–1703 (2006).
D’Angelo, W. A., Fries, J. F., Masi, A. T. & Shulman, L. E. Pathologic observations in systemic sclerosis (scleroderma). A study of fifty-eight autopsy cases and fifty-eight matched controls. Am. J. Med. 46, 428–440 (1969).
Howe, S. et al. Antimyenteric neuronal antibodies in scleroderma. J. Clin. Invest. 94, 761–770 (1994).
Singh, J. et al. Effects of scleroderma antibodies and pooled human immunoglobulin on anal sphincter and colonic smooth muscle function. Gastroenterology 143, 1308–1318 (2012).
Singh, J. et al. Immunoglobulins from scleroderma patients inhibit the muscarinic receptor activation in internal anal sphincter smooth muscle cells. Am. J. Physiol. Gastrointest. Liver Physiol. 297, G1206–G1213 (2009).
Amaral, T. N., Peres, F. A., Lapa, A. T., Marques-Neto, J. F. & Appenzeller, S. Neurologic involvement in scleroderma: a systematic review. Semin. Arthritis Rheum. 43, 335–347 (2013).
Thoua, N. M., Schizas, A., Forbes, A., Denton, C. P. & Emmanuel, A. V. Internal anal sphincter atrophy in patients with systemic sclerosis. Rheumatology 50, 1596–1602 (2011).
Thoua, N. M., Abdel-Halim, M., Forbes, A., Denton, C. P. & Emmanuel, A. V. Fecal incontinence in systemic sclerosis is secondary to neuropathy. Am. J. Gastroenterol. 107, 597–603 (2012).
Plichta, D. R. et al. Congruent microbiome signatures in fibrosis-prone autoimmune diseases: IgG4-related disease and systemic sclerosis. Genome Med. 13, 35 (2021).
Kröner, P. T., Tolaymat, O. A., Bowman, A. W., Abril, A. & Lacy, B. E. Gastrointestinal manifestations of rheumatological diseases. Am. J. Gastroenterol. 114, 1441–1454 (2019).
Kaniecki, T., Abdi, T. & McMahan, Z. H. Clinical assessment of gastrointestinal involvement in patients with systemic sclerosis. Med. Res. Arch. 8, 2252 (2020).
Kumar, S. et al. Review article: pathogenesis and clinical manifestations of gastrointestinal involvement in systemic sclerosis. Aliment. Pharmacol. Ther. 45, 883–898 (2017).
McFarlane, I. M. et al. Gastrointestinal manifestations of systemic sclerosis. Rheumatology (Sunnyvale) 8, 235 (2018).
Bajraktari, I. H. et al. Oral manifestations of systemic sclerosis and correlation with anti-topoisomerase I antibodies (SCL-70). Med. Arch. 69, 153–156 (2015).
Jung, S., Martin, T., Schmittbuhl, M. & Huck, O. The spectrum of orofacial manifestations in systemic sclerosis: a challenging management. Oral. Dis. 23, 424–439 (2017).
Crincoli, V. et al. Orofacial manifestations and temporomandibular disorders of systemic scleroderma: an observational study. Int. J. Mol. Sci. 17, 1189 (2016).
Chu, C. H., Yeung, C. M., Lai, I. A., Leung, W. K. & Mok, M. Y. Oral health of Chinese people with systemic sclerosis. Clin. Oral. Investig. 15, 931–939 (2011).
Dagenais, M. et al. The Canadian Systemic Sclerosis Oral Health Study IV: oral radiographic manifestations in systemic sclerosis compared with the general population. Oral. Surg. Oral. Med. Oral. Pathol. Oral. Radiol. 120, 104–111 (2015).
Auluck, A., Pai, K. M., Shetty, C. & Shenoi, S. D. Mandibular resorption in progressive systemic sclerosis: a report of three cases. Dentomaxillofac Radiol. 34, 384–386 (2005).
Abdouh, I. et al. Measuring the impact of systemic sclerosis on oral health-related quality of life in a UK population. J. Oral. Pathol. Med. 50, 812–819 (2021).
Baron, M. et al. The Canadian systemic sclerosis oral health study: orofacial manifestations and oral health-related quality of life in systemic sclerosis compared with the general population. Rheumatology 53, 1386–1394 (2014).
Raja, J., Ng, C. T., Sujau, I., Chin, K. F. & Sockalingam, S. High-resolution oesophageal manometry and 24-hour impedance-pH study in systemic sclerosis patients: association with clinical features, symptoms and severity. Clin. Exp. Rheumatol. 34, 115–121 (2016).
Lock, G., Holstege, A., Lang, B. & Scholmerich, J. Gastrointestinal manifestations of progressive systemic sclerosis. Am. J. Gastroenterol. 92, 763–771 (1997).
Lock, G. et al. Association of esophageal dysfunction and pulmonary function impairment in systemic sclerosis. Am. J. Gastroenterol. 93, 341–345 (1998).
Weston, S., Thumshirn, M., Wiste, J. & Camilleri, M. Clinical and upper gastrointestinal motility features in systemic sclerosis and related disorders. Am. J. Gastroenterol. 93, 1085–1089 (1998).
Henry, M. A., Harbermann, M. C. & Rocha, O. M. Esophageal motor disturbances in progressive systemic sclerosis. Dis. Esophagus 12, 51–53 (1999).
Arif, T., Masood, Q., Singh, J. & Hassan, I. Assessment of esophageal involvement in systemic sclerosis and morphea (localized scleroderma) by clinical, endoscopic, manometric and pH metric features: a prospective comparative hospital based study. BMC Gastroenterol. 15, 24 (2015).
Kahrilas, P. J. et al. The Chicago Classification of esophageal motility disorders, v3.0. Neurogastroenterol. Motil. 27, 160–174 (2015).
Yadlapati, R. et al. Esophageal motility disorders on high-resolution manometry: Chicago classification version 4.0. Neurogastroenterol. Motil. 33, e14058 (2021).
Aggarwal, N. et al. Spectrum of esophageal dysmotility in systemic sclerosis on high-resolution esophageal manometry as defined by Chicago classification. Dis. Esophagus 30, 1–6 (2017).
Roman, S. et al. Esophageal dysmotility associated with systemic sclerosis: a high-resolution manometry study. Dis. Esophagus 24, 299–304 (2011).
Adler, B., Hummers, L. K., Pasricha, P. J. & McMahan, Z. H. Gastroparesis in systemic sclerosis: a detailed analysis using whole-gut scintigraphy. Rheumatology 61, 4503–4508 (2022).
Wegener, M., Adamek, R. J., Wedmann, B., Jergas, M. & Altmeyer, P. Gastrointestinal transit through esophagus, stomach, small and large intestine in patients with progressive systemic sclerosis. Dig. Dis. Sci. 39, 2209–2215 (1994).
Vettori, S. et al. Esophageal high-resolution impedance manometry alterations in asymptomatic patients with systemic sclerosis: prevalence, associations with disease features, and prognostic value. Clin. Rheumatol. 37, 1239–1247 (2018).
Ahuja, N. K. & Clarke, J. O. Scleroderma and the esophagus. Gastroenterol. Clin. North. Am. 50, 905–918 (2021).
Christmann, R. B., Wells, A. U., Capelozzi, V. L. & Silver, R. M. Gastroesophageal reflux incites interstitial lung disease in systemic sclerosis: clinical, radiologic, histopathologic, and treatment evidence. Semin. Arthritis Rheum. 40, 241–249 (2010).
Orringer, M. B., Dabich, L., Zarafonetis, C. J. & Sloan, H. Gastroesophageal reflux in esophageal scleroderma: diagnosis and implications. Ann. Thorac. Surg. 22, 120–130 (1976).
Katzka, D. A. et al. Barrett’s metaplasia and adenocarcinoma of the esophagus in scleroderma. Am. J. Med. 82, 46–52 (1987).
Anilkumar, M., Alkhayyat, M., Grewal, U. S., Sanaka, M. R. & Thota, P. N. Higher risk of neoplastic progression of Barrett’s esophagus in patients with systemic sclerosis. Gastroenterol. Rep. 9, 595–596 (2021).
Snyder, D. L. et al. Prevalence of Barrett’s esophagus in female patients with scleroderma. Am. J. Gastroenterol. 116, 517–521 (2021).
Hunt, R. H. et al. The stomach in health and disease. Gut 64, 1650–1668 (2015).
Chen, J. D. & McCallum, R. W. Clinical applications of electrogastrography. Am. J. Gastroenterol. 88, 1324–1336 (1993).
Savarino, E. et al. Gastrointestinal motility disorder assessment in systemic sclerosis. Rheumatology 52, 1095–1100 (2013).
Gemignani, L. et al. Lactulose breath test to assess oro-cecal transit delay and estimate esophageal dysmotility in scleroderma patients. Semin. Arthritis Rheum. 42, 522–529 (2013).
Marie, I. et al. Delayed gastric emptying determined using the 13C-octanoic acid breath test in patients with systemic sclerosis. Arthritis Rheum. 64, 2346–2355 (2012).
Franck-Larsson, K., Hedenström, H., Dahl, R. & Rönnblom, A. Delayed gastric emptying in patients with diffuse versus limited systemic sclerosis, unrelated to gastrointestinal symptoms and myoelectric gastric activity. Scand. J. Rheumatol. 32, 348–355 (2003).
Chen, J. D., Schirmer, B. D. & McCallum, R. W. Serosal and cutaneous recordings of gastric myoelectrical activity in patients with gastroparesis. Am. J. Physiol. 266, G90–G98 (1994).
McNearney, T., Lin, X., Shrestha, J., Lisse, J. & Chen, J. D. Characterization of gastric myoelectrical rhythms in patients with systemic sclerosis using multichannel surface electrogastrography. Dig. Dis. Sci. 47, 690–698 (2002).
McNearney, T. A. et al. Gastric slow waves, gastrointestinal symptoms and peptides in systemic sclerosis patients. Neurogastroenterol. Motil. 21, 1269–e1120 (2009).
Tack, J. Functional dyspepsia: impaired fundic accommodation. Curr. Treat. Options Gastroenterol. 3, 287–294 (2000).
Sridhar, K. R., Lange, R. C., Magyar, L., Soykan, I. & McCallum, R. W. Prevalence of impaired gastric emptying of solids in systemic sclerosis: diagnostic and therapeutic implications. J. Lab. Clin. Med. 132, 541–546 (1998).
Ghrénassia, E. et al. Prevalence, correlates and outcomes of gastric antral vascular ectasia in systemic sclerosis: a EUSTAR case-control study. J. Rheumatol. 41, 99–105 (2014).
Harrison, E., Herrick, A. L., McLaughlin, J. T. & Lal, S. Malnutrition in systemic sclerosis. Rheumatology 51, 1747–1756 (2012).
Caserta, L. et al. Assessment of intestinal permeability and orocecal transit time in patients with systemic sclerosis: analysis of relationships with epidemiologic and clinical parameters. Rheumatol. Int. 23, 226–230 (2003).
Medsger, T. A. et al. A disease severity scale for systemic sclerosis: development and testing. J. Rheumatol. 26, 2159–2167 (1999).
Savarino, E. et al. Possible connection between gastroesophageal reflux and interstitial pulmonary fibrosis in patients with systemic sclerosis. Recent. Prog. Med. 100, 512–516 (2009).
Parodi, A. et al. Small intestinal bacterial overgrowth in patients suffering from scleroderma: clinical effectiveness of its eradication. Am. J. Gastroenterol. 103, 1257–1262 (2008).
Simrén, M. & Stotzer, P. O. Use and abuse of hydrogen breath tests. Gut 55, 297–303 (2006).
Quigley, E. M. Small intestinal bacterial overgrowth: what it is and what it is not. Curr. Opin. Gastroenterol. 30, 141–146 (2014).
Sachdev, A. H. & Pimentel, M. Gastrointestinal bacterial overgrowth: pathogenesis and clinical significance. Ther. Adv. Chronic Dis. 4, 223–231 (2013).
Kaye, S. A. et al. Small bowel bacterial overgrowth in systemic sclerosis: detection using direct and indirect methods and treatment outcome. Br. J. Rheumatol. 34, 265–269 (1995).
Feng, X., Li, X. Q. & Jiang, Z. Prevalence and predictors of small intestinal bacterial overgrowth in systemic sclerosis: a systematic review and meta-analysis. Clin. Rheumatol. 40, 3039–3051 (2021).
Marie, I., Ducrotté, P., Denis, P., Menard, J. F. & Levesque, H. Small intestinal bacterial overgrowth in systemic sclerosis. Rheumatology 48, 1314–1319 (2009).
Harrison, E., Herrick, A. L., McLaughlin, J. T. & Lal, S. An assessment of the nutritional status of patients with systemic sclerosis. Clin. Nutr. ESPEN 10, e177–e178 (2015).
Valenzuela, A. et al. Intestinal pseudo-obstruction in patients with systemic sclerosis: an analysis of the Nationwide Inpatient Sample. Rheumatology 55, 654–658 (2016).
Mecoli, C., Purohit, S., Sandorfi, N. & Derk, C. T. Mortality, recurrence, and hospital course of patients with systemic sclerosis-related acute intestinal pseudo-obstruction. J. Rheumatol. 41, 2049–2054 (2014).
Cheah, J. X. et al. Slow colonic transit in systemic sclerosis: an objective assessment of risk factors and clinical phenotype. Arthritis Care Res. https://doi.org/10.1002/acr.24767 (2021).
Nagasako, K., Ota, Y., Sasaki, H. & Hamano, K. Progressive systemic sclerosis: report of a case with colonic involvement. Dis. Colon. Rectum 21, 364–368 (1978).
Trezza, M., Krogh, K., Egekvist, H., Bjerring, P. & Laurberg, S. Bowel problems in patients with systemic sclerosis. Scand. J. Gastroenterol. 34, 409–413 (1999).
Umar, S. B. et al. The impact of pelvic floor and lower gastrointestinal symptoms on quality of life in women with systemic sclerosis. J. Clin. Gastroenterol. 50, e55–e59 (2016).
Engel, A. F., Kamm, M. A. & Talbot, I. C. Progressive systemic sclerosis of the internal anal sphincter leading to passive faecal incontinence. Gut 35, 857–859 (1994).
Jaffin, B. W., Chang, P. & Spiera, H. Fecal incontinence in scleroderma. Clinical features, anorectal manometric findings, and their therapeutic implications. J. Clin. Gastroenterol. 25, 513–517 (1997).
Richard, N. et al. Clinical correlates of faecal incontinence in systemic sclerosis: identifying therapeutic avenues. Rheumatology 56, 581–588 (2017).
Fynne, L., Worsøe, J., Laurberg, S. & Krogh, K. Faecal incontinence in patients with systemic sclerosis: is an impaired internal anal sphincter the only cause. Scand. J. Rheumatol. 40, 462–466 (2011).
Fynne, L. et al. Distensibility of the anal canal in patients with systemic sclerosis: a study with the functional lumen imaging probe. Colorectal Dis. 15, e40–e47 (2013).
Rao, S. S., Rattanakovit, K. & Patcharatrakul, T. Diagnosis and management of chronic constipation in adults. Nat. Rev. Gastroenterol. Hepatol. 13, 295–305 (2016).
Vollebregt, P. F., Burgell, R. E., Hooper, R. L., Knowles, C. H. & Scott, S. M. Clinical impact of rectal hyposensitivity: a cross-sectional study of 2,876 patients with refractory functional constipation. Am. J. Gastroenterol. 116, 758–768 (2021).
Kim, K. C. et al. Anorectal dysfunction in systemic sclerosis. J. Korean Med. Sci. 11, 244–249 (1996).
Sallam, H. S., McNearney, T. A. & Chen, J. Z. Anorectal motility and sensation abnormalities and its correlation with anorectal symptoms in patients with systemic sclerosis: a preliminary study. ISRN Gastroenterol. 2011, 402583 (2011).
Antoniou, A. J. et al. Comprehensive radionuclide esophagogastrointestinal transit study: methodology, reference values, and initial clinical experience. J. Nucl. Med. 56, 721–727 (2015).
Ahuja, N. K., Mische, L., Clarke, J. O., Wigley, F. M. & McMahan, Z. H. Pyridostigmine for the treatment of gastrointestinal symptoms in systemic sclerosis. Semin. Arthritis Rheum. 48, 111–116 (2018).
Johnson, D. A. et al. Metoclopramide response in patients with progressive systemic sclerosis. Effect on esophageal and gastric motility abnormalities. Arch. Intern. Med. 147, 1597–1601 (1987).
Sarosiek, I. et al. Effect of domperidone therapy on gastroparesis symptoms: results of a dynamic cohort study by NIDDK gastroparesis consortium. Clin. Gastroenterol. Hepatol. 20, e452–e464 (2022).
Ou, L. B., Moriello, C., Douros, A. & Filion, K. B. Domperidone and the risks of sudden cardiac death and ventricular arrhythmia: a systematic review and meta-analysis of observational studies. Br. J. Clin. Pharmacol. 87, 3649–3658 (2021).
Sanger, G. J. & Furness, J. B. Ghrelin and motilin receptors as drug targets for gastrointestinal disorders. Nat. Rev. Gastroenterol. Hepatol. 13, 38–48 (2016).
Fiorucci, S., Distrutti, E., Gerli, R. & Morelli, A. Effect of erythromycin on gastric and gallbladder emptying and gastrointestinal symptoms in scleroderma patients is maintained medium term. Am. J. Gastroenterol. 89, 550–555 (1994).
Soudah, H. C., Hasler, W. L. & Owyang, C. Effect of octreotide on intestinal motility and bacterial overgrowth in scleroderma. N. Engl. J. Med. 325, 1461–1467 (1991).
Camilleri, M. Effects of somatostatin analogues on human gastrointestinal motility. Digestion 57, 90–92 (1996).
Hansi, N. et al. Consensus best practice pathway of the UK scleroderma study group: gastrointestinal manifestations of systemic sclerosis. Clin. Exp. Rheumatol. 32, S-214–S-221 (2014).
Nagaraja, V., McMahan, Z. H., Getzug, T. & Khanna, D. Management of gastrointestinal involvement in scleroderma. Curr. Treatm. Opt. Rheumatol. 1, 82–105 (2015).
Hughes, M. et al. Proton pump inhibitors in systemic sclerosis: a reappraisal to optimise treatment of gastro-oesophageal reflux disease. Lancet Rheumatol. 4, e795–e803 (2022).
Foocharoen, C. et al. Prevalence and predictors of proton pump inhibitor partial response in gastroesophageal reflux disease in systemic sclerosis: a prospective study. Sci. Rep. 10, 769 (2020).
Lombardo, L., Foti, M., Ruggia, O. & Chiecchio, A. Increased incidence of small intestinal bacterial overgrowth during proton pump inhibitor therapy. Clin. Gastroenterol. Hepatol. 8, 504–508 (2010).
Yadlapati, R., Gyawali, C. P., Pandolfino, J. E. et al. AGA clinical practice update on the personalized approach to the evaluation and management of GERD: expert review. Clin. Gastroenterol. Hepatol. 20, 984–994.e981 (2022).
Gyawali, C. P. et al. Modern diagnosis of GERD: the Lyon Consensus. Gut 67, 1351–1362 (2018).
Shirai, Y., Kawami, N., Iwakiri, K. & Kuwana, M. Use of vonoprazan, a novel potassium-competitive acid blocker, for the treatment of proton pump inhibitor-refractory reflux esophagitis in patients with systemic sclerosis. J. Scleroderma Relat. Disord. 7, 57–61 (2022).
Shibli, F., Kitayama, Y. & Fass, R. Novel therapies for gastroesophageal reflux disease: beyond proton pump inhibitors. Curr. Gastroenterol. Rep. 22, 16 (2020).
Foocharoen, C. et al. Effectiveness of add-on therapy with domperidone vs alginic acid in proton pump inhibitor partial response gastro-oesophageal reflux disease in systemic sclerosis: randomized placebo-controlled trial. Rheumatology 56, 214–222 (2017).
Koek, G. H., Sifrim, D., Lerut, T., Janssens, J. & Tack, J. Effect of the GABAB agonist baclofen in patients with symptoms and duodeno-gastro-oesophageal reflux refractory to proton pump inhibitors. Gut 52, 1397–1402 (2003).
Leiman, D. A. et al. Alginate therapy is effective treatment for GERD symptoms: a systematic review and meta-analysis. Dis. Esophagus 30, 1–9 (2017).
Malamood, M., Roberts, A., Kataria, R., Parkman, H. P. & Schey, R. Mirtazapine for symptom control in refractory gastroparesis. Drug. Des. Devel. Ther. 11, 1035–1041 (2017).
Jubran, B. B., Bolduc, J., Eng, A., Stapleton, M. & Wilson, B. J. Off-label use of aprepitant for scleroderma-associated nausea and vomiting: a case report. J. Clin. Pharm. Ther. 44, 805–808 (2019).
Jehangir, A. & Parkman, H. P. Cannabinoid use in patients with gastroparesis and related disorders: prevalence and benefit. Am. J. Gastroenterol. 114, 945–953 (2019).
Tack, J., Janssen, P., Masaoka, T., Farré, R. & Van Oudenhove, L. Efficacy of buspirone, a fundus-relaxing drug, in patients with functional dyspepsia. Clin. Gastroenterol. Hepatol. 10, 1239–1245 (2012).
Pittman, N. et al. Treatment of small intestinal bacterial overgrowth in systemic sclerosis: a systematic review. Rheumatology 57, 1802–1811 (2018).
Pazzi, P., Putinati, S., Bagni, B., Govoni, M. & Trotta, F. Bile acid malabsorption in progressive systemic sclerosis. Gut 29, 552–553 (1988).
Vijayvargiya, P. & Camilleri, M. Update on bile acid malabsorption: finally ready for prime time? Curr. Gastroenterol. Rep. 20, 10 (2018).
Corsetti, M. & Tack, J. New pharmacological treatment options for chronic constipation. Expert Opin. Pharmacother. 15, 927–941 (2014).
Rao, S. S. C. Plecanatide: a new guanylate cyclase agonist for the treatment of chronic idiopathic constipation. Ther. Adv. Gastroenterol. 11, 1756284818777945 (2018).
Fukudo, S. et al. High-dose linaclotide is effective and safe in patients with chronic constipation: a phase III randomized, double-blind, placebo-controlled study with a long-term open-label extension study in Japan. Neurogastroenterol. Motil. 31, e13487 (2019).
Farmer, A. D., Ruffle, J. K. & Hobson, A. R. Linaclotide increases cecal pH, accelerates colonic transit, and increases colonic motility in irritable bowel syndrome with constipation. Neurogastroenterol. Motil. 31, e13492 (2019).
Andresen, V. et al. Effect of 5 days linaclotide on transit and bowel function in females with constipation-predominant irritable bowel syndrome. Gastroenterology 133, 761–768 (2007).
Lembo, A. J. et al. Long-term safety and effectiveness of lubiprostone, a chloride channel (ClC-2) activator, in patients with chronic idiopathic constipation. Dig. Dis. Sci. 56, 2639–2645 (2011).
Drossman, D. A. et al. Clinical trial: lubiprostone in patients with constipation-associated irritable bowel syndrome — results of two randomized, placebo-controlled studies. Aliment. Pharmacol. Ther. 29, 329–341 (2009).
Miner, P. B. et al. A randomized phase III clinical trial of plecanatide, a uroguanylin analog, in patients with chronic idiopathic constipation. Am. J. Gastroenterol. 112, 613–621 (2017).
Bassotti, G., Usai Satta, P. & Bellini, M. Plecanatide for the treatment of chronic idiopathic constipation in adult patients. Expert. Rev. Clin. Pharmacol. 12, 1019–1026 (2019).
Dein, E. J., Wigley, F. M. & McMahan, Z. H. Linaclotide for the treatment of refractory lower bowel manifestations of systemic sclerosis. BMC Gastroenterol. 21, 174 (2021).
Rao, S. S. Biofeedback therapy for constipation in adults. Best. Pract. Res. Clin. Gastroenterol. 25, 159–166 (2011).
Butt, S. K. et al. Lack of effect of sacral nerve stimulation for incontinence in patients with systemic sclerosis. Colorectal Dis. 17, 903–907 (2015).
Yin, J., Abell, T. D., McCallum, R. W. & Chen, J. D. Gastric neuromodulation with Enterra system for nausea and vomiting in patients with gastroparesis. Neuromodulation 15, 224–231 (2012).
Zhu, Y. et al. Transcutaneous auricular vagal nerve stimulation improves functional dyspepsia by enhancing vagal efferent activity. Am. J. Physiol. Gastrointest. Liver Physiol. 320, G700–G711 (2021).
Gottfried-Blackmore, A. et al. Open-label pilot study: Non-invasive vagal nerve stimulation improves symptoms and gastric emptying in patients with idiopathic gastroparesis. Neurogastroenterol. Motil. 32, e13769 (2020).
Chen, J. D. Z., Ni, M. & Yin, J. Electroacupuncture treatments for gut motility disorders. Neurogastroenterol. Motil. 30, e13393 (2018).
McNearney, T. A., Sallam, H. S., Hunnicutt, S. E., Doshi, D. & Chen, J. D. Prolonged treatment with transcutaneous electrical nerve stimulation (TENS) modulates neuro-gastric motility and plasma levels of vasoactive intestinal peptide (VIP), motilin and interleukin-6 (IL-6) in systemic sclerosis. Clin. Exp. Rheumatol. 31, 140–150 (2013).
Zhang, B. et al. Integrative effects and vagal mechanisms of transcutaneous electrical acustimulation on gastroesophageal motility in patients with gastroesophageal reflux disease. Am. J. Gastroenterol. 116, 1495–1505 (2021).
Liu, Z. et al. Preventive effects of transcutaneous electrical acustimulation on ischemic stroke-induced constipation mediated via the autonomic pathway. Am. J. Physiol. Gastrointest. Liver Physiol. 315, G293–G301 (2018).
Parrado, R. H., Lemus, H. N., Coral-Alvarado, P. X. & Quintana López, G. Gastric antral vascular ectasia in systemic sclerosis: current concepts. Int. J. Rheumatol. 2015, 762546 (2015).
Hoffmann-Vold, A. M. et al. Safety and efficacy of faecal microbiota transplantation by Anaerobic Cultivated Human Intestinal Microbiome (ACHIM) in patients with systemic sclerosis: study protocol for the randomised controlled phase II ReSScue trial. BMJ Open. 11, e048541 (2021).
Raja, J., Nihtyanova, S. I., Murray, C. D., Denton, C. P. & Ong, V. H. Sustained benefit from intravenous immunoglobulin therapy for gastrointestinal involvement in systemic sclerosis. Rheumatology 55, 115–119 (2016).
Hoffmann-Vold, A. M. & Volkmann, E. R. Gastrointestinal involvement in systemic sclerosis: effects on morbidity and mortality and new therapeutic approaches. J. Scleroderma Relat. Disord. 6, 37–43 (2021).
Clark, K. E., Etomi, O., Denton, C. P., Ong, V. H. & Murray, C. D. Intravenous immunoglobulin therapy for severe gastrointestinal involvement in systemic sclerosis. Clin. Exp. Rheumatol. 33, S168–S170 (2015).
Kumar, S. et al. Role of muscarinic-3 receptor antibody in systemic sclerosis: correlation with disease duration and effects of IVIG. Am. J. Physiol. Gastrointest. Liver Physiol. 310, G1052–G1060 (2016).
Volkmann, E. R. & Hoffmann-Vold, A. M. Gastrointestinal tract microbiota modifications in systemic sclerosis. Eur. J. Rheumatol. 7, S228–S236 (2020).
Fretheim, H. et al. Fecal microbiota transplantation in systemic sclerosis: a double-blind, placebo-controlled randomized pilot trial. PLoS One 15, e0232739 (2020).
de Groot, P. et al. Faecal microbiota transplantation halts progression of human new-onset type 1 diabetes in a randomised controlled trial. Gut 70, 92–105 (2021).
Smillie, C. S. et al. Strain tracking reveals the determinants of bacterial engraftment in the human gut following fecal microbiota transplantation. Cell Host Microbe 23, 229–240.e225 (2018).
Kong, L. et al. Linking strain engraftment in fecal microbiota transplantation with maintenance of remission in Crohn’s disease. Gastroenterology 159, 2193–2202.e2195 (2020).
Henke, M. T. et al. Ruminococcus gnavus, a member of the human gut microbiome associated with Crohn’s disease, produces an inflammatory polysaccharide. Proc. Natl Acad. Sci. USA 116, 12672–12677 (2019).
Henke, M. T. et al. Capsular polysaccharide correlates with immune response to the human gut microbe. Proc. Natl Acad. Sci. USA 118, e2007595118 (2021).
May-Zhang, A. A. et al. Combinatorial transcriptional profiling of mouse and human enteric neurons identifies shared and disparate subtypes in situ. Gastroenterology 160, 755–770.e26 (2021).
Jarret, A. et al. Enteric nervous system-derived IL-18 orchestrates mucosal barrier immunity. Cell 180, 50–63.e12 (2020).
Baker, C. V. & Bronner-Fraser, M. Establishing neuronal identity in vertebrate neurogenic placodes. Development 127, 3045–3056 (2000).
Barki, N. et al. Chemogenetics defines a short-chain fatty acid receptor gut–brain axis. Elife 11, e73777 (2022).
Abdullah, N., Defaye, M. & Altier, C. Neural control of gut homeostasis. Am. J. Physiol. Gastrointest. Liver Physiol. 319, G718–G732 (2020).
Brookes, S. J., Spencer, N. J., Costa, M. & Zagorodnyuk, V. P. Extrinsic primary afferent signalling in the gut. Nat. Rev. Gastroenterol. Hepatol. 10, 286–296 (2013).
Serlin, H. K. & Fox, E. A. Neurotrophin-4 is essential for survival of the majority of vagal afferents to the mucosa of the small intestine, but not the stomach. Auton. Neurosci. 233, 102811 (2021).
Powley, T. L. et al. Vagal innervation of the stomach reassessed: brain-gut connectome uses smart terminals. Ann. N. Y. Acad. Sci. 1454, 14–30 (2019).
Chen, B. N., Olsson, C., Sharrad, D. F. & Brookes, S. J. Sensory innervation of the guinea pig colon and rectum compared using retrograde tracing and immunohistochemistry. Neurogastroenterol. Motil. 28, 1306–1316 (2016).
Kamitakahara, A., Wu, H. H. & Levitt, P. Distinct projection targets define subpopulations of mouse brainstem vagal neurons that express the autism-associated MET receptor tyrosine kinase. J. Comp. Neurol. 525, 3787–3808 (2017).
Megat, S. et al. Differences between dorsal root and trigeminal ganglion nociceptors in mice revealed by translational profiling. J. Neurosci. 39, 6829–6847 (2019).
Niu, X. et al. Mapping of extrinsic innervation of the gastrointestinal tract in the mouse embryo. J. Neurosci. 40, 6691–6708 (2020).
Gabella, G. in The Rat Nervous System 3rd edn (ed. Paxinos, G.) 77–109 (Academic Press, 2004).
Browning, K. N. & Travagli, R. A. Central nervous system control of gastrointestinal motility and secretion and modulation of gastrointestinal functions. Compr. Physiol. 4, 1339–1368 (2014).
Benarroch, E. E. in Encyclopedia of the Neurological Sciences 2nd edn (eds Aminoff, M. J. & Daroff, R. B.) 805–808 (Academic Press, 2014).
Maddern, G. J., Kiroff, G. K., Leppard, P. I. & Jamieson, G. G. Domperidone, metoclopramide, and placebo. All give symptomatic improvement in gastroesophageal reflux. J. Clin. Gastroenterol. 8, 135–140 (1986).
Chrysos, E. et al. Erythromycin enhances oesophageal motility in patients with gastro-oesophageal reflux. Anz. J. Surg. 71, 98–102 (2001).
Arts, J., Caenepeel, P., Verbeke, K. & Tack, J. Influence of erythromycin on gastric emptying and meal related symptoms in functional dyspepsia with delayed gastric emptying. Gut 54, 455–460 (2005).
Kao, C. H., Wang, S. J. & Pang, D. Y. Effects of oral erythromycin on upper gastrointestinal motility in patients with non-insulin-dependent diabetes mellitus. Nucl. Med. Commun. 16, 790–793 (1995).
Di Stefano, M. et al. Effect of buspirone, a 5-HT1A receptor agonist, on esophageal motility in healthy volunteers. Dis. Esophagus 25, 470–476 (2012).
Van Oudenhove, L., Kindt, S., Vos, R., Coulie, B. & Tack, J. Influence of buspirone on gastric sensorimotor function in man. Aliment. Pharmacol. Ther. 28, 1326–1333 (2008).
Scheerens, C., Tack, J. & Rommel, N. Buspirone, a new drug for the management of patients with ineffective esophageal motility? United European Gastroenterol. J. 3, 261–265 (2015).
Kumar, N., Barai, S., Gambhir, S. & Rastogi, N. Effect of mirtazapine on gastric emptying in patients with cancer-associated anorexia. Indian. J. Palliat. Care 23, 335–337 (2017).
Gooden, J. Y. & Takahashi, P. Y. Mirtazapine treatment of diabetic gastroparesis as a novel method to reduce tube-feed residual: a case report. J. Med. Case Rep. 7, 38 (2013).
Carbone, F., Vanuytsel, T. & Tack, J. The effect of mirtazapine on gastric accommodation, gastric sensitivity to distention, and nutrient tolerance in healthy subjects. Neurogastroenterol. Motil 29, https://doi.org/10.1111/nmo.13146 (2017).
Omari, T. I. et al. Effect of baclofen on esophagogastric motility and gastroesophageal reflux in children with gastroesophageal reflux disease: a randomized controlled trial. J. Pediatr. 149, 468–474 (2006).
Andrews, P. L. & Wood, K. L. Systemic baclofen stimulates gastric motility and secretion via a central action in the rat. Br. J. Pharmacol. 89, 461–467 (1986).
Blonski, W., Vela, M. F., Freeman, J., Sharma, N. & Castell, D. O. The effect of oral buspirone, pyridostigmine, and bethanechol on esophageal function evaluated with combined multichannel esophageal impedance-manometry in healthy volunteers. J. Clin. Gastroenterol. 43, 253–260 (2009).
Dhar, S. I. et al. Effects of pyridostigmine on esophageal and pharyngeal motility in dysphagic patients undergoing high-resolution manometry. Dysphagia 37, 4–10 (2022).
Pasha, S. F., Lunsford, T. N. & Lennon, V. A. Autoimmune gastrointestinal dysmotility treated successfully with pyridostigmine. Gastroenterology 131, 1592–1596 (2006).
O’Dea, C. J., Brookes, J. H. & Wattchow, D. A. The efficacy of treatment of patients with severe constipation or recurrent pseudo-obstruction with pyridostigmine. Colorectal Dis. 12, 540–548 (2010).
Manini, M. L., Camilleri, M., Grothe, R. & Di Lorenzo, C. Application of pyridostigmine in pediatric gastrointestinal motility disorders: a case series. Paediatr. Drugs 20, 173–180 (2018).
Bharucha, A. E. et al. Pilot study of pyridostigmine in constipated patients with autonomic neuropathy. Clin. Auton. Res. 18, 194–202 (2008).
Schey, R. et al. Domperidone to treat symptoms of gastroparesis: benefits and side effects from a large single-center cohort. Dig. Dis. Sci. 61, 3545–3551 (2016).
Reddymasu, S. C., Soykan, I. & McCallum, R. W. Domperidone: review of pharmacology and clinical applications in gastroenterology. Am. J. Gastroenterol. 102, 2036–2045 (2007).
Shin, A. Patient considerations in the management of chronic constipation: focus on prucalopride. Patient Prefer. Adherence 10, 1373–1384 (2016).
Smart, C. J. & Malik, K. I. Prucalopride for the treatment of ileus. Expert. Opin. Investig. Drugs 26, 489–493 (2017).
Oustamanolakis, P. & Tack, J. Prucalopride for chronic intestinal pseudo-obstruction. Aliment. Pharmacol. Ther. 35, 398–399 (2012).
Langworthy, J., Parkman, H. P. & Schey, R. Emerging strategies for the treatment of gastroparesis. Expert Rev. Gastroenterol. Hepatol. 10, 817–825 (2016).
Khanna, D. et al. Reliability and validity of the University of California, Los Angeles scleroderma clinical trial consortium gastrointestinal tract instrument. Arthritis Rheum. 61, 1257–1263 (2009).
Spiegel, B. M. et al. Development of the NIH patient-reported outcomes measurement information system (PROMIS) gastrointestinal symptom scales. Am. J. Gastroenterol. 109, 1804–1814 (2014).
Acknowledgements
Z.H.M.’s work is supported by funding from NIH/NIAMS K23 AR071473. D.K.’s work is supported by funding from K24 ARAR063120.
Author information
Authors and Affiliations
Contributions
All authors researched data for the article, made a substantial contribution to discussion of the content, wrote the article and reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
J.C. declares that she is a consultant for Phathom Pharmaceuticals. R.J.X. declares that he is co-founder of Jnana Therapeutics and Celsius Therapeutics, a board member for Moonlake Immunotherapeutics and on the Scientific Advisory Board for Nestle. P.J.P. declares that he is co-founder of, holds equity in and is a consultant for Neurogastrx. D.K. declares that he has received consulting fees from Acceleron, Actelion, Amgen, Bayer, Boehringer Ingelheim, Chemomab, CSL Behring, Genentech/Roche, Horizon, Paracrine Cell Therapy, Mitsubishi Tanabe Pharma, Prometheus and Theraly. Z.H.M., S.K. and J.Z.C. declare no competing interests.
Peer review
Peer review information
Nature Reviews Rheumatology thanks G. Gyger, R. Naik and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
HealthMeasures: https://www.healthmeasures.net/
UCLA SCTC GIT 2.0 Questionnaire: http://bit.ly/UCLASCTCGIT
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
McMahan, Z.H., Kulkarni, S., Chen, J. et al. Systemic sclerosis gastrointestinal dysmotility: risk factors, pathophysiology, diagnosis and management. Nat Rev Rheumatol 19, 166–181 (2023). https://doi.org/10.1038/s41584-022-00900-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41584-022-00900-6
This article is cited by
-
Protein profiling in systemic sclerosis patients with different pulmonary complications using proteomic antibody microarray
Arthritis Research & Therapy (2024)
-
Treatable Traits in Systemic Sclerosis
Clinical Reviews in Allergy & Immunology (2023)