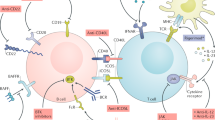
Abstract
The treat-to-target (T2T) concept has improved outcomes for patients with diabetes, hypertension and rheumatoid arthritis. This therapeutic strategy involves choosing a well-defined, relevant target, taking therapeutic steps, evaluating whether the target has been achieved, and taking action if it has not. The T2T principle has been embraced by systemic lupus erythematosus (SLE) experts, but measurable and achievable outcomes, and therapeutic options, are needed to make this approach possible in practice. Considerable evidence has been generated regarding meaningful ‘state’ outcomes for SLE. Low disease activity has been defined and studied, and the most aspirational goal, remission, has been defined by the Definition of Remission in SLE task force. By contrast, current therapeutic options in SLE are limited, and more effective and safer therapies are urgently needed. Fortunately, clinical trial activity in SLE has been unprecedented, and encouraging results have been seen for novel therapies, including biologic and small-molecule agents. Thus, with the expected advent of such treatments, it is likely that sufficiently diverse therapies for SLE will be available in the foreseeable future, allowing the routine implementation of T2T approaches in the care of patients with SLE.
Key points
-
The treat-to-target (T2T) therapeutic strategy consists of four key steps: establish a relevant individualized target, take steps to achieve it, monitor the target achievement, and adjust the therapy if the target is not achieved.
-
Validation of the Lupus Low Disease Activity State (LLDAS) definition of low disease activity and recent consensus on the final DORIS definition of remission in systemic lupus erythematosus (SLE) have provided feasible treatment targets for the adoption of a T2T strategy in SLE.
-
With the advent of novel therapeutics for SLE, including biologics and small molecules, T2T for SLE will become a clinical reality in the coming years.
-
The use of LLDAS and DORIS definitions in clinical trials for novel therapeutics could provide robust, discriminatory outcome measures.
-
Trials comparing the active attainment of LLDAS or DORIS remission as end points in a T2T approach with a conventional management approach are still needed.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Change history
11 February 2022
The formatting of Agner R. Parra Sánchez’s name in the xml was incorrect, resulting in Parra being wrongly classed as a middle name. This has now been corrected online.
References
Atar, D., Birkeland, K. I. & Uhlig, T. ‘Treat to target’: moving targets from hypertension, hyperlipidaemia and diabetes to rheumatoid arthritis. Ann. Rheum. Dis. 69, 629–630 (2010).
Wangnoo, S. K. et al. Treat-to-target trials in diabetes. Indian J. Endocrinol. Metab. 18, 166–174 (2014).
Castrejon, I. & Pincus, T. Differences in treat-to-target in patients with rheumatoid arthritis versus hypertension and diabetes — consequences for clinical care. Bull. NYU Hosp. Jt. Dis. 69, 104–110 (2011).
Drosos, A. A., Pelechas, E. & Voulgari, P. V. Treatment strategies are more important than drugs in the management of rheumatoid arthritis. Clin. Rheumatol. 39, 1363–1368 (2020).
Smolen, J. S. Treat to target in rheumatology: a historical account on occasion of the 10th anniversary. Rheum. Dis. Clin. North Am. 45, 477–485 (2019).
Grigor, C. et al. Effect of a treatment strategy of tight control for rheumatoid arthritis (the TICORA study): a single-blind randomised controlled trial. Lancet 364, 263–269 (2004).
Aletaha, D., Alasti, F. & Smolen, J. S. Optimisation of a treat-to-target approach in rheumatoid arthritis: strategies for the 3-month time point. Ann. Rheum. Dis. 75, 1479–1485 (2016).
Mueller, R. B. et al. Superiority of a treat-to-target strategy over conventional treatment with fixed csDMARD and corticosteroids: a multi-center randomized controlled trial in RA patients with an inadequate response to conventional synthetic DMARDs, and new therapy with certolizumab pegol. J. Clin. Med. 8, 302 (2019).
Solomon, D. H. et al. Benefits and sustainability of a learning collaborative for implementation of treat-to-target in rheumatoid arthritis: results of a cluster-randomized controlled phase II clinical trial. Arthritis Care Res. 70, 1551–1556 (2018).
Goekoop-Ruiterman, Y. P. et al. Comparison of treatment strategies in early rheumatoid arthritis: a randomized trial. Ann. Intern. Med. 146, 406–415 (2007).
Goekoop-Ruiterman, Y. P. et al. Clinical and radiographic outcomes of four different treatment strategies in patients with early rheumatoid arthritis (the BeSt study): a randomized, controlled trial. Arthritis Rheum. 52, 3381–339 (2005).
Smolen, J. S. et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2016 update. Ann. Rheum. Dis. 76, 960–977 (2017).
Stoffer, M. A. et al. Evidence for treating rheumatoid arthritis to target: results of a systematic literature search update. Ann. Rheum. Dis. 75, 16–22 (2016).
Dures, E. et al. Treat-to-target in PsA: methods and necessity. RMD Open 6, e001083 (2020).
Kiltz, U. et al. Treat-to-target (T2T) recommendations for gout. Ann. Rheum. Dis. 76, 632–638 (2017).
Franklyn, K., Hoi, A., Nikpour, M. & Morand, E. F. The need to define treatment goals for systemic lupus erythematosus. Nat. Rev. Rheumatol. 10, 567–571 (2014).
van Vollenhoven, R. F. et al. Treat-to-target in systemic lupus erythematosus: recommendations from an international task force. Ann. Rheum. Dis. 73, 958–967 (2014).
Franklyn, K. et al. Definition and initial validation of a Lupus Low Disease Activity State (LLDAS). Ann. Rheum. Dis. 75, 1615–1621 (2016).
van Vollenhoven, R. et al. A framework for remission in SLE: consensus findings from a large international task force on definitions of remission in SLE (DORIS). Ann. Rheum. Dis. 76, 554–561 (2017).
Fanouriakis, A. et al. 2019 update of the EULAR recommendations for the management of systemic lupus erythematosus. Ann. Rheum. Dis. 78, 736–745 (2019).
Piga, M. et al. Failure to achieve lupus low disease activity state (LLDAS) six months after diagnosis is associated with early damage accrual in Caucasian patients with systemic lupus erythematosus. Arthritis Res. Ther. 19, 247 (2017).
Hao, Y. et al. Determinants and protective associations of the lupus low disease activity state in a prospective Chinese cohort. Clin. Rheumatol. https://doi.org/10.1007/s10067-021-05940-z (2021).
Tsang, A. S. M. W. & Bultink, I. E. Systemic lupus erythematosus: review of synthetic drugs. Expert Opin. Pharmacother. 16, 2793–2806 (2015).
Basta, F., Fasola, F., Triantafyllias, K. & Schwarting, A. Systemic lupus erythematosus (SLE) therapy: the old and the new. Rheumatol. Ther. 7, 433–446 (2020).
Bruce, I. N. et al. Factors associated with damage accrual in patients with systemic lupus erythematosus: results from the Systemic Lupus International Collaborating Clinics (SLICC) Inception Cohort. Ann. Rheum. Dis. 74, 1706–1713 (2015).
Antonis Fanouriakis, G. B. Treat-to-target in lupus: what does the future hold? Int. J. Clin. Rheumatol. 10, 461–477 (2015).
Gatto, M., Zen, M., Iaccarino, L. & Doria, A. New therapeutic strategies in systemic lupus erythematosus management. Nat. Rev. Rheumatol. 15, 30–48 (2019).
Aringer, M., Leuchten, N. & Schneider, M. Treat to target in systemic lupus erythematosus. Rheum. Dis. Clin. North Am. 45, 537–548 (2019).
Golder, V. et al. Does expert opinion match the operational definition of the Lupus Low Disease Activity State (LLDAS)? A case-based construct validity study. Semin. Arthritis Rheum. 46, 798–803 (2017).
Golder, V. E. A. Lupus low disease activity state as a treatment endpoint for systemic lupus erythematosus: a prospective validation study. Lancet Rheumatol. 1, e95–e102 (2019).
Golder, V. et al. Association of the lupus low disease activity state (LLDAS) with health-related quality of life in a multinational prospective study. Arthritis Res. Ther. 19, 62 (2017).
Golder, V. et al. Frequency and predictors of the lupus low disease activity state in a multi-national and multi-ethnic cohort. Arthritis Res. Ther. 18, 260 (2016).
Babaoglu, H., Li, J., Goldman, D., Magder, L. S. & Petri, M. Time to lupus low disease activity state in the Hopkins Lupus Cohort: role of African American ethnicity. Arthritis Care Res. 72, 225–232 (2020).
van Vollenhoven, R. F. et al. 2021 DORIS definition of remission in SLE: final recommendations from an international task force. Lupus Sci. Med. 8, e000538 (2021).
Mucke, J., Dusing, C., Klose, N., Schneider, M. & Chehab, G. Remission in SLE — do DORIS criteria match the treating physician’s judgment? A cross sectional study to assess reasons for discordance. Rheumatology 60, 4298–4305 (2021).
Petri, M. & Magder, L. S. Comparison of remission and lupus low disease activity state in damage prevention in a United States systemic lupus erythematosus cohort. Arthritis Rheumatol. 70, 1790–1795 (2018).
Zen, M. et al. The effect of different durations of remission on damage accrual: results from a prospective monocentric cohort of Caucasian patients. Ann. Rheum. Dis. 76, 562–565 (2017).
Zen, M. et al. Prolonged remission in Caucasian patients with SLE: prevalence and outcomes. Ann. Rheum. Dis. 74, 2117–2122 (2015).
Ugarte-Gil, M. F. et al. Remission and Low Disease Activity Status (LDAS) protect lupus patients from damage occurrence: data from a multiethnic, multinational Latin American Lupus Cohort (GLADEL). Ann. Rheum. Dis. 76, 2071–2074 (2017).
Pons-Estel, G. et al. Impact of remission and low disease activity status on hospitalizations among SLE patients from the GLADEL Latin American Cohort [abstract]. Arthritis Rheumatol. 72, 1272 (2020).
Reategui-Sokolova, C. et al. Remission and low disease activity state prevent hospitalizations in systemic lupus erythematosus patients. Lupus 28, 1344–1349 (2019).
Ugarte-Gil, M. et al. LLDAS (Low Lupus Disease Activity State) and remission prevent damage accrual in systemic lupus erythematosus (SLE) patients in a primarily Mestizo cohort [abstract]. Arthritis Rheumatol. 72, 0254 (2020).
Ugarte-Gil, M. F. et al. Better health-related quality of life in systemic lupus erythematosus predicted by low disease activity state/remission: data from the Peruvian Almenara Lupus Cohort. Arthritis Care Res. 72, 1159–1162 (2020).
Wilhelm, T. R., Magder, L. S. & Petri, M. Remission in systemic lupus erythematosus: durable remission is rare. Ann. Rheum. Dis. 76, 547–553 (2017).
Tsang, A. S. M. W., Bultink, I. E., Heslinga, M. & Voskuyl, A. E. Both prolonged remission and Lupus Low Disease Activity State are associated with reduced damage accrual in systemic lupus erythematosus. Rheumatology 56, 121–128 (2017).
Tsang, A. S. M. W. P. et al. The relationship between remission and health-related quality of life in a cohort of SLE patients. Rheumatology 58, 628–635 (2019).
Golder, V. et al. Evaluation of remission definitions for systemic lupus erythematosus: a prospective cohort study. Lancet Rheumatol. 1, e103–e110 (2019).
Zirkzee, E., Bonte-Mineur, F. & Kok, M. Implementation of treat-to-target principles in the management of systemic lupus erythematosus. Lupus 27, 1218–1219 (2018).
Bombardier, C., Gladman, D. D., Urowitz, M. B., Caron, D. & Chang, C. H. Derivation of the SLEDAI. A disease activity index for lupus patients. The Committee on Prognosis Studies in SLE. Arthritis Rheum. 35, 630–640 (1992).
Gladman, D. et al. The development and initial validation of the Systemic Lupus International Collaborating Clinics/American College of Rheumatology damage index for systemic lupus erythematosus. Arthritis Rheum. 39, 363–369 (1996).
Zandbelt, M. M., Welsing, P. M., van Gestel, A. M. & van Riel, P. L. Health Assessment Questionnaire modifications: is standardisation needed? Ann. Rheum. Dis. 60, 841–845 (2001).
Mucke, J., Kuss, O., Brinks, R., Schanze, S. & Schneider, M. LUPUS-BEST — treat-to-target in systemic lupus erythematosus: study protocol for a three-armed cluster-randomised trial. Lupus Sci. Med. 8, e000516 (2021).
Navarra, S. V. et al. Efficacy and safety of belimumab in patients with active systemic lupus erythematosus: a randomised, placebo-controlled, phase 3 trial. Lancet 377, 721–731 (2011).
Furie, R. et al. A phase III, randomized, placebo-controlled study of belimumab, a monoclonal antibody that inhibits B lymphocyte stimulator, in patients with systemic lupus erythematosus. Arthritis Rheum. 63, 3918–3930 (2011).
Oon, S. et al. Lupus Low Disease Activity State (LLDAS) discriminates responders in the BLISS-52 and BLISS-76 phase III trials of belimumab in systemic lupus erythematosus. Ann. Rheum. Dis. 78, 629–633 (2019).
Parodis, I. et al. Predictors of low disease activity and clinical remission following belimumab treatment in systemic lupus erythematosus. Rheumatology 58, 2170–2176 (2019).
Parodis, I. et al. Definitions of remission in systemic lupus erythematosus: a post-hoc analysis of two randomised clinical trials. Lancet Rheumatol. 1, e163–e173 (2019).
Morand, E. F. et al. Attainment of treat-to-target endpoints in SLE patients with high disease activity in the atacicept phase 2b ADDRESS II study. Rheumatology 59, 2930–2938 (2020).
Morand, E. F., Trasieva, T., Berglind, A., Illei, G. G. & Tummala, R. Lupus Low Disease Activity State (LLDAS) attainment discriminates responders in a systemic lupus erythematosus trial: post-hoc analysis of the Phase IIb MUSE trial of anifrolumab. Ann. Rheum. Dis. 77, 706–713 (2018).
Wallace, D. J. et al. Baricitinib for systemic lupus erythematosus: a double-blind, randomised, placebo-controlled, phase 2 trial. Lancet 392, 222–231 (2018).
Doria, A. et al. Efficacy and safety of subcutaneous belimumab in anti-double-stranded DNA-positive, hypocomplementemic patients with systemic lupus erythematosus. Arthritis Rheumatol. 70, 1256–1264 (2018).
Stohl, W. et al. Efficacy and safety of subcutaneous belimumab in systemic lupus erythematosus: a fifty-two-week randomized, double-blind, placebo-controlled study. Arthritis Rheumatol. 69, 1016–1027 (2017).
Furie, R. et al. Op0164 BLISS-LN: a randomised, double-blind, placebo-controlled phase 3 trial of intravenous belimumab in patients with active lupus nephritis. Ann. Rheum. Dis. 79, 103–103 (2020).
Merrill, J. T. et al. Efficacy and safety of rituximab in moderately-to-severely active systemic lupus erythematosus: the randomized, double-blind, phase II/III systemic lupus erythematosus evaluation of rituximab trial. Arthritis Rheum. 62, 222–233 (2010).
Rovin, B. H. et al. Efficacy and safety of rituximab in patients with active proliferative lupus nephritis: the Lupus Nephritis Assessment with Rituximab study. Arthritis Rheum. 64, 1215–1226 (2012).
Ryden-Aulin, M. et al. Off-label use of rituximab for systemic lupus erythematosus in Europe. Lupus Sci. Med. 3, e000163 (2016).
Furie R. et al. Two-year results from a randomized, controlled study of obinutuzumab for proliferative lupus nephritis [abstract]. Arthritis Rheumatol. https://acrabstracts.org/abstract/two-year-results-from-a-randomized-controlled-study-of-obinutuzumab-for-proliferative-lupus-nephritis/ (2020).
US National library of Medicine. ClinicalTrials.gov https://ClinicalTrials.gov/show/NCT04221477 (2021).
Lavie, F. et al. Increase of B cell-activating factor of the TNF family (BAFF) after rituximab treatment: insights into a new regulating system of BAFF production. Ann. Rheum. Dis. 66, 700–703 (2007).
Kraaij, T. et al. Long-term effects of combined B-cell immunomodulation with rituximab and belimumab in severe, refractory systemic lupus erythematosus: 2-year results. Nephrol. Dial. Transpl. 36, 1474–1483 (2020).
Shipa, M. et al. OP0129 Belimumab after rituximab significantly reduced IgG anti-dsDNA antibody levels and prolonged time to severe flare in patients with systemic lupus erythematosus [abstract]. Ann. Rheum. Dis. 80, 74–74 (2021).
Teng, Y. K. O. et al. Phase III, multicentre, randomised, double-blind, placebo-controlled, 104-week study of subcutaneous belimumab administered in combination with rituximab in adults with systemic lupus erythematosus (SLE): BLISS-BELIEVE study protocol. BMJ Open 9, e025687 (2019).
Carreira, P. L. & Isenberg, D. A. Recent developments in biologic therapies for the treatment of patients with systemic lupus erythematosus. Rheumatology 58, 382–387 (2019).
Furie, R. et al. Efficacy and safety of dapirolizumab pegol in patients with moderately to severely active systemic lupus erythematosus: a randomized, placebo-controlled study [abstract]. Arthritis Rheumatol. 71, 944 (2019).
US National library of Medicine. ClinicalTrials.gov https://ClinicalTrials.gov/show/NCT04294667 (2021).
Reynolds, J. A. et al. Cytokine profiling in active and quiescent SLE reveals distinct patient subpopulations. Arthritis Res. Ther. 20, 173 (2018).
Rovin, B. H. et al. A multicenter, randomized, double-blind, placebo-controlled study to evaluate the efficacy and safety of treatment with sirukumab (CNTO 136) in patients with active lupus nephritis. Arthritis Rheumatol. 68, 2174–2183 (2016).
Illei, G. G. et al. Tocilizumab in systemic lupus erythematosus: data on safety, preliminary efficacy, and impact on circulating plasma cells from an open-label phase I dosage-escalation study. Arthritis Rheum. 62, 542–552 (2010).
van Vollenhoven, R. F. et al. Efficacy and safety of ustekinumab, an IL-12 and IL-23 inhibitor, in patients with active systemic lupus erythematosus: results of a multicentre, double-blind, phase 2, randomised, controlled study. Lancet 392, 1330–1339 (2018).
US National library of Medicine. ClinicalTrials.gov https://ClinicalTrials.gov/show/NCT03517722 (2021).
Mullard, A. FDA approves AstraZeneca’s anifrolumab for lupus. Nat. Rev. Drug Discov. 20, 658 (2021).
Furie, R. et al. Anifrolumab, an anti-interferon-alpha receptor monoclonal antibody, in moderate-to-severe systemic lupus erythematosus. Arthritis Rheumatol. 69, 376–386 (2017).
Furie, R. A. et al. Type I interferon inhibitor anifrolumab in active systemic lupus erythematosus (TULIP-1): a randomised, controlled, phase 3 trial. Lancet Rheumatol. 1, e208–e219 (2019).
Morand, E. F. et al. Trial of anifrolumab in active systemic lupus erythematosus. N. Engl. J. Med. 382, 211–221 (2020).
Agence Nationale de Sécurité du Médicament et des Produits de Santé (ANSM). Protocole d’utilisation therapeutique et de recueil d’informations Anifrolumab 300 mg, solution à diluer pour perfusion. ATU de Cohorte. https://ansm.sante.fr/uploads/2021/07/23/20210723-atuc-anifrolumab-put.pdf (2021).
Ishii, T. et al. Multicenter double-blind randomized controlled trial to evaluate the effectiveness and safety of bortezomib as a treatment for refractory systemic lupus erythematosus. Mod. Rheumatol. 28, 986–992 (2018).
Walhelm, T. et al. Clinical experience of proteasome inhibitor bortezomib regarding efficacy and safety in severe systemic lupus erythematosus: a nationwide study. Front. Immunol. 12, 756941 (2021).
Aringer, M. Janus kinase inhibitors clear to land. Rheumatology 57, 1131–1132 (2018).
US National library of Medicine. ClinicalTrials.gov https://ClinicalTrials.gov/show/NCT03843125 (2021).
US National library of Medicine. ClinicalTrials.gov https://ClinicalTrials.gov/show/NCT03616964 (2021).
US National library of Medicine. ClinicalTrials.gov https://ClinicalTrials.gov/show/NCT03616912 (2021).
Singhal, S. et al. 5 Oral selective tyrosine kinase 2 (TYK2) inhibition with BMS-986165 in patients with systemic lupus erythematosus: a phase 2, randomized, double-blind, placebo-controlled study (PAISLEY) [abstract]. Lupus Sci. Med. 6, A5–A6 (2019).
Shao, W. H. & Cohen, P. L. The role of tyrosine kinases in systemic lupus erythematosus and their potential as therapeutic targets. Expert Rev. Clin. Immunol. 10, 573–582 (2014).
US National library of Medicine. ClinicalTrials.gov https://ClinicalTrials.gov/show/NCT04305197 (2021).
Rovin, B. H. et al. A randomized, controlled double-blind study comparing the efficacy and safety of dose-ranging voclosporin with placebo in achieving remission in patients with active lupus nephritis. Kidney Int. 95, 219–231 (2019).
Onno Teng, Y. K., Parikh, S. V., Saxena, A., Solomons, N. & Huizinga, R. B. AURORA phase 3 study demonstrates voclosporin statistical superiority over standard of care in lupus nephritis (LN) [abstract]. Lupus Sci. Med. 7, 011 (2020).
US National library of Medicine. ClinicalTrials.gov https://ClinicalTrials.gov/show/NCT03021499 (2021).
Merrill, J. T. et al. Efficacy and safety of atacicept in patients with systemic lupus erythematosus: results of a twenty-four-week, multicenter, randomized, double-blind, placebo-controlled, parallel-arm, phase IIb Study. Arthritis Rheumatol. 70, 266–276 (2018).
Merrill, J. T. et al. Phase III trial results with blisibimod, a selective inhibitor of B-cell activating factor, in subjects with systemic lupus erythematosus (SLE): results from a randomised, double-blind, placebo-controlled trial. Ann. Rheum. Dis. 77, 883–889 (2018).
Merrill, J. T. et al. Efficacy and safety of subcutaneous tabalumab, a monoclonal antibody to B-cell activating factor, in patients with systemic lupus erythematosus: results from ILLUMINATE-2, a 52-week, phase III, multicentre, randomised, double-blind, placebo-controlled study. Ann. Rheum. Dis. 75, 332–340 (2016).
Cogollo, E., Silva, M. A. & Isenberg, D. Profile of atacicept and its potential in the treatment of systemic lupus erythematosus. Drug Des. Devel. Ther. 9, 1331–1339 (2015).
Ginzler, E. M. et al. Atacicept in combination with MMF and corticosteroids in lupus nephritis: results of a prematurely terminated trial. Arthritis Res. Ther. 14, R33 (2012).
US National library of Medicine. ClinicalTrials.gov https://ClinicalTrials.gov/show/NCT02885610 (2020).
US National library of Medicine. ClinicalTrials.gov https://ClinicalTrials.gov/show/NCT04082416 (2021).
US National library of Medicine. ClinicalTrials.gov https://ClinicalTrials.gov/show/NCT03312907 (2021).
US National library of Medicine. ClinicalTrials.gov https://ClinicalTrials.gov/show/NCT00539838 (2020).
Mysler, E. F. et al. Efficacy and safety of ocrelizumab in active proliferative lupus nephritis: results from a randomized, double-blind, phase III study. Arthritis Rheum. 65, 2368–2379 (2013).
Clowse, M. E. et al. Efficacy and safety of epratuzumab in moderately to severely active systemic lupus erythematosus: results from two phase III randomized, double-blind, placebo-controlled trials. Arthritis Rheumatol. 69, 362–375 (2017).
Merrill, J. T. et al. The efficacy and safety of abatacept in patients with non-life-threatening manifestations of systemic lupus erythematosus: results of a twelve-month, multicenter, exploratory, phase IIb, randomized, double-blind, placebo-controlled trial. Arthritis Rheum. 62, 3077–3087 (2010).
Furie, R. et al. Efficacy and safety of abatacept in lupus nephritis: a twelve-month, randomized, double-blind study. Arthritis Rheumatol. 66, 379–389 (2014).
Pimentel-Quiroz, V. R., Ugarte-Gil, M. F. & Alarcon, G. S. Abatacept for the treatment of systemic lupus erythematosus. Expert Opin. Investig. Drugs 25, 493–499 (2016).
US National library of Medicine. ClinicalTrials.gov https://ClinicalTrials.gov/show/NCT04181762 (2021).
US National library of Medicine. ClinicalTrials.gov https://ClinicalTrials.gov/show/NCT04376827 (2021).
Jayne, D. et al. POS0690 Randomized, controlled, phase 2 trial of type 1 IFN inhibitor anifrolumab in patients with active proliferative lupus nephritis. Ann. Rheum. Dis. 80, 592–592 (2021).
Werth, V. et al. OP0193 BIIB059, a humanized monoclonal antibody targeting BDCA2 on plasmacytoid dendritic cells (pDC), shows dose-related efficacy in the phase 2 LILAC study in patients (pts) with active cutaneous lupus erythematosus (CLE). Ann. Rheum. Dis. 79, 120–121 (2020).
US National library of Medicine. ClinicalTrials.gov https://ClinicalTrials.gov/show/NCT02847598 (2020).
US National library of Medicine. ClinicalTrials.gov https://ClinicalTrials.gov/show/NCT04895241 (2021).
Hasni, S. et al. 183 A phase 1B/2A trial of tofacitinib, an oral Janus kinase inhibitor, in systemic lupus erythematosus [abstract]. Lupus Sci. Med. 6, A139–A140 (2019).
US National library of Medicine. ClinicalTrials.gov https://ClinicalTrials.gov/show/NCT03159936 (2020).
US National library of Medicine. ClinicalTrials.gov https://ClinicalTrials.gov/show/NCT03288324 (2021).
US National library of Medicine. ClinicalTrials.gov https://ClinicalTrials.gov/show/NCT02908100 (2020).
Hanly, J. G. et al. The frequency and outcome of lupus nephritis: results from an international inception cohort study. Rheumatology 55, 252–262 (2016).
Wang, H., Ren, Y. L., Chang, J., Gu, L. & Sun, L. Y. A systematic review and meta-analysis of prevalence of biopsy-proven lupus nephritis. Arch. Rheumatol. 33, 17–25 (2018).
Fanouriakis, A. et al. 2019 Update of the Joint European League Against Rheumatism and European Renal Association-European Dialysis and Transplant Association (EULAR/ERA-EDTA) recommendations for the management of lupus nephritis. Ann. Rheum. Dis. 79, 713–723 (2020).
Kostopoulou, M., Adamichou, C. & Bertsias, G. An update on the diagnosis and management of lupus nephritis. Curr. Rheumatol. Rep. 22, 30 (2020).
Zeher, M. et al. Efficacy and safety of enteric-coated mycophenolate sodium in combination with two glucocorticoid regimens for the treatment of active lupus nephritis. Lupus 20, 1484–1493 (2011).
Condon, M. B. et al. Prospective observational single-centre cohort study to evaluate the effectiveness of treating lupus nephritis with rituximab and mycophenolate mofetil but no oral steroids. Ann. Rheum. Dis. 72, 1280–1286 (2013).
Davidson, J. E. et al. Renal remission status and longterm renal survival in patients with lupus nephritis: a retrospective cohort analysis. J. Rheumatol. 45, 671–677 (2018).
Dall’Era, M. et al. Predictors of long-term renal outcome in lupus nephritis trials: lessons learned from the Euro-Lupus Nephritis cohort. Arthritis Rheumatol. 67, 1305–1313 (2015).
Tamirou, F. et al. A proteinuria cut-off level of 0.7 g/day after 12 months of treatment best predicts long-term renal outcome in lupus nephritis: data from the MAINTAIN Nephritis Trial. Lupus Sci. Med. 2, e000123 (2015).
Ugolini-Lopes, M. R. et al. Early proteinuria response: a valid real-life situation predictor of long-term lupus renal outcome in an ethnically diverse group with severe biopsy-proven nephritis? Lupus Sci. Med. 4, e000213 (2017).
Gunnarsson, I. et al. Histopathologic and clinical outcome of rituximab treatment in patients with cyclophosphamide-resistant proliferative lupus nephritis. Arthritis Rheum. 56, 1263–1272 (2007).
Lim, C. C. et al. Severe infections in patients with lupus nephritis treated with immunosuppressants: a retrospective cohort study. Nephrology 22, 478–484 (2017).
Acknowledgements
The work of A.R.P.S. is supported by the European Union’s Horizon 2020 research and innovation programme support for the Amsterdam Rheumatology Center for Autoimmune Diseases (ARCAID; grant number 847551).
Author information
Authors and Affiliations
Contributions
A.R.P.S. researched data for the article. All authors contributed substantially to discussion of the content, wrote the article and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
R.F.v.V. declares that he has received research support (institutional grants) from BMS, GSK, Lilly and UCB and support for educational programmes from Pfizer and Roche. R.F.v.V. declares that he has also received consulting fees from AbbVie, AstraZeneca, Biogen, Biotest, BMS, Galapagos, Gilead, Janssen, Pfizer, Sanofi, Servier, UCB and Vielabio and personal honoraria as a speaker from AbbVie, Galapagos, GSK, Janssen, Pfizer and UCB. A.E.V. declares that he has received research support (institutional grants) from GSK and UCB, consulting fees from GSK, AstraZeneca and Roche, and personal honoraria as a speaker from GSK. A.R.P.S. declares no competing interests.
Additional information
Peer review information
Nature Reviews Rheumatology thanks C.C. Mok and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Parra Sánchez, A.R., Voskuyl, A.E. & van Vollenhoven, R.F. Treat-to-target in systemic lupus erythematosus: advancing towards its implementation. Nat Rev Rheumatol 18, 146–157 (2022). https://doi.org/10.1038/s41584-021-00739-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41584-021-00739-3
This article is cited by
-
Präzisionsmedizin in der Diagnostik und Therapie von Asthma
Die Innere Medizin (2024)
-
Characteristics of patients with initial diagnosis of systemic lupus erythematosus in emergency department and their outcomes: a retrospective single-center study
Clinical Rheumatology (2024)
-
Advances in the study of macrophage polarization in inflammatory immune skin diseases
Journal of Inflammation (2023)
-
A conformation-locking inhibitor of SLC15A4 with TASL proteostatic anti-inflammatory activity
Nature Communications (2023)
-
MicroRNA as a potential biomarker for systemic lupus erythematosus: pathogenesis and targeted therapy
Clinical and Experimental Medicine (2023)