Abstract
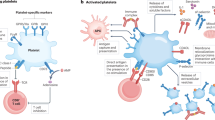
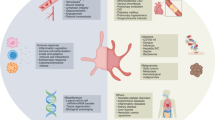
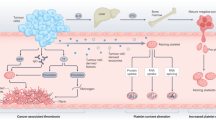
Rheumatoid arthritis (RA) is an autoimmune disease in which a variety of circulating pro-inflammatory cells and dysregulated molecules are involved in disease aetiology and progression. Platelets are an important cellular element in the circulation that can bind several dysregulated molecules (such as collagen, thrombin and fibrinogen) that are present both in the synovium and the circulation of patients with RA. Platelets not only respond to dysregulated molecules in their environment but also transport and express their own inflammatory mediators, and serve as regulators at the boundary between haemostasis and immunity. Activated platelets also produce microparticles, which further convey signalling molecules and receptors to the synovium and circulation, thereby positioning these platelet-derived particles as strategic regulators of inflammation. These diverse functions come together to make platelets facilitators of cellular crosstalk in RA. Thus, the receptor functions, ligand binding potential and dysregulated signalling pathways in platelets are becoming increasingly important for treatment in RA. This Review aims to highlight the role of platelets in RA and the need to closely examine platelets as health indicators when designing effective pharmaceutical targets in this disease.
Key points
-
Platelets have important immune effector functions in rheumatoid arthritis (RA).
-
Platelet signalling pathways are dysregulated in the presence of pro-inflammatory molecules such as collagen, thrombin, fibrinogen and cytokines.
-
Increased (pro-inflammatory) platelet signalling causes platelets to develop antigen-presenting functions and results in dysregulated intercellular aggregation and pathological clot formation.
-
Activated platelets and their thrombo-inflammatory function in RA represent an important risk factor for thrombosis and cardiovascular comorbidities.
-
Activated platelets produce pro-inflammatory microparticles that are present both in the circulation and in the synovium; these microparticles function in systemic inflammatory processes in RA.
-
In an individualized medicine approach, platelet structure and biochemical markers of inflammation would be used during diagnosis and platelet activity monitored during treatment to track immune responses in RA.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Ma, V. Y., Chan, L. & Carruthers, K. J. Incidence, prevalence, costs, and impact on disability of common conditions requiring rehabilitation in the United States: stroke, spinal cord injury, traumatic brain injury, multiple sclerosis, osteoarthritis, rheumatoid arthritis, limb loss, and back pain. Arch. Phys. Med. Rehabil. 95, 986–995 (2014).
Cross, M. et al. The global burden of rheumatoid arthritis: estimates from the global burden of disease 2010 study. Ann. Rheum. Dis. 73, 1316–1322 (2014).
Rudan, I. et al. Prevalence of rheumatoid arthritis in low- and middle-income countries: a systematic review and analysis. J. Glob. Health 5, 010409 (2015).
Usenbo, A., Kramer, V., Young, T. & Musekiwa, A. Prevalence of arthritis in Africa: a systematic review and meta-analysis. PLoS ONE 10, e0133858 (2015).
Panagopoulos, P. K. & Lambrou, G. I. Bone erosions in rheumatoid arthritis: recent developments in pathogenesis and therapeutic implications. J. Musculoskelet. Neuronal Interact. 18, 304–319 (2018).
Pietraforte, D. et al. Redox control of platelet functions in physiology and pathophysiology. Antioxid. Redox Signal. 21, 177–193 (2014).
Balogh, E. et al. Oxidative stress impairs energy metabolism in primary cells and synovial tissue of patients with rheumatoid arthritis. Arthritis Res. Ther. 20, 95 (2018).
Pretorius, E., Akeredolu, O. O., Soma, P. & Kell, D. B. Major involvement of bacterial components in rheumatoid arthritis and its accompanying oxidative stress, systemic inflammation and hypercoagulability. Exp. Biol. Med. 242, 355–373 (2017).
Ferrante, E. et al. Determinants of thromboxane biosynthesis in rheumatoid arthritis: Role of RAGE and oxidant stress. Free Radic. Biol. Med. 49, 857–864 (2010).
Staron, A., Makosa, G. & Koter-Michalak, M. Oxidative stress in erythrocytes from patients with rheumatoid arthritis. Rheumatol. Int. 32, 331–334 (2012).
Olumuyiwa-Akeredolu, O. O. & Pretorius, E. Platelet and red blood cell interactions and their role in rheumatoid arthritis. Rheumatol. Int. 35, 1955–1964 (2015).
Olumuyiwa-Akeredolu, O. O., Soma, P., Buys, A. V., Debusho, L. K. & Pretorius, E. Characterizing pathology in erythrocytes using morphological and biophysical membrane properties: Relation to impaired hemorheology and cardiovascular function in rheumatoid arthritis. Biochim. Biophys. Acta Biomembr. 1859, 2381–2391 (2017).
van Breukelen-van der Stoep, D. F., Klop, B., van Zeben, D., Hazes, J. M. & Castro Cabezas, M. Cardiovascular risk in rheumatoid arthritis: how to lower the risk? Atherosclerosis 231, 163–172 (2013).
Mantel, A. et al. Risk factors for the rapid increase in risk of acute coronary events in patients with new-onset rheumatoid arthritis: a nested case-control study. Arthritis Rheumatol. 67, 2845–2854 (2015).
Meyer, P. W. et al. Circulating cytokine profiles and their relationships with autoantibodies, acute phase reactants, and disease activity in patients with rheumatoid arthritis. Mediators Inflamm. 2010, 158514 (2010).
Wruck, C. J. et al. Role of oxidative stress in rheumatoid arthritis: insights from the Nrf2-knockout mice. Ann. Rheum. Dis. 70, 844–850 (2011).
Veselinovic, M. et al. Oxidative stress in rheumatoid arthritis patients: relationship to diseases activity. Mol. Cell Biochem. 391, 225–232 (2014).
Vasanthi, P., Nalini, G. & Rajasekhar, G. Status of oxidative stress in rheumatoid arthritis. Int. J. Rheum. Dis. 12, 29–33 (2009).
Bunescu, A., Seideman, P., Lenkei, R., Levin, K. & Egberg, N. Enhanced Fcgamma receptor I, alphaMbeta2 integrin receptor expression by monocytes and neutrophils in rheumatoid arthritis: interaction with platelets. J. Rheumatol. 31, 2347–2355 (2004).
Tamura, N. et al. Soluble CD154 in rheumatoid arthritis: elevated plasma levels in cases with vasculitis. J. Rheumatol. 28, 2583–2590 (2001).
Is¸ık, M., Sahin, H. & Huseyin, E. New platelet indices as inflammatory parameters for patients with rheumatoid arthritis. Eur. J. Rheumatol. 1, 144–146 (2014).
Yazici, S. et al. The platelet indices in patients with rheumatoid arthritis: mean platelet volume reflects disease activity. Platelets 21, 122–125 (2010).
Milovanovic, M., Nilsson, E. & Jaremo, P. Relationships between platelets and inflammatory markers in rheumatoid arthritis. Clin. Chim. Acta 343, 237–240 (2004).
Gasparyan, A. Y., Stavropoulos-Kalinoglou, A., Mikhailidis, D. P., Douglas, K. M. & Kitas, G. D. Platelet function in rheumatoid arthritis: arthritic and cardiovascular implications. Rheumatol. Int. 31, 153–164 (2011).
Boilard, E. et al. Platelets amplify inflammation in arthritis via collagen-dependent microparticle production. Science 327, 580–583 (2010).
Lindemann, S. et al. Activated platelets mediate inflammatory signaling by regulated interleukin 1β synthesis. J. Cell Biol. 154, 485–490 (2001).
Rosengren, S., Corr, M. & Boyle, D. L. Platelet-derived growth factor and transforming growth factor beta synergistically potentiate inflammatory mediator synthesis by fibroblast-like synoviocytes. Arthritis Res. Ther. 12, R65 (2010).
Ahamed, J. et al. In vitro and in vivo evidence for shear-induced activation of latent transforming growth factor-β1. Blood 112, 3650–3660 (2008).
Lefevre, S. et al. Disease-specific effects of matrix and growth factors on adhesion and migration of rheumatoid synovial fibroblasts. J. Immunol. 198, 4588–4595 (2017).
Brown, A. J., Sepuru, K. M., Sawant, K. V. & Rajarathnam, K. Platelet-derived chemokine CXCL7 dimer preferentially exists in the glycosaminoglycan-bound form: implications for neutrophil-platelet crosstalk. Front. Immunol. 8, 1248 (2017).
Fox, J. M. et al. CXCL4/platelet factor 4 is an agonist of CCR1 and drives human monocyte migration. Sci. Rep. 8, 9466 (2018).
Ohyama, K. et al. Immune complexome analysis of serum and its application in screening for immune complex antigens in rheumatoid arthritis. Clin. Chem. 57, 905–909 (2011).
Dyer, K. D. et al. Mouse and human eosinophils degranulate in response to platelet-activating factor (PAF) and lysoPAF via a PAF-receptor-independent mechanism: evidence for a novel receptor. J. Immunol. 184, 6327–6334 (2010).
Undas, A. et al. Thrombin generation in rheumatoid arthritis: dependence on plasma factor composition. Thromb. Haemost. 104, 224–230 (2010).
Wang, M. J. et al. Determination of role of thromboxane A2 in rheumatoid arthritis. Discov. Med. 19, 23–32 (2015).
Eastgate, J. A., Symons, J. A., Wood, N. C., Capper, S. J. & Duff, G. W. Plasma levels of interleukin-1-alpha in rheumatoid arthritis. Br. J. Rheumatol. 30, 295–297 (1991).
Nishimura, S. et al. IL-1α induces thrombopoiesis through megakaryocyte rupture in response to acute platelet needs. J. Cell Biol. 209, 453–466 (2015).
Lukasik, Z. M., Makowski, M. & Makowska, J. S. From blood coagulation to innate and adaptive immunity: the role of platelets in the physiology and pathology of autoimmune disorders. Rheumatol. Int. 38, 959–974 (2018).
Flad, H. D. & Brandt, E. Platelet-derived chemokines: pathophysiology and therapeutic aspects. Cell. Mol. Life Sci. 67, 2363–2386 (2010).
Habets, K. L., Huizinga, T. W. & Toes, R. E. Platelets and autoimmunity. Eur. J. Clin. Invest. 43, 746–757 (2013).
Henn, V. et al. CD40 ligand on activated platelets triggers an inflammatory reaction of endothelial cells. Nature 391, 591–594 (1998).
Boilard, E., Blanco, P. & Nigrovic, P. A. Platelets: active players in the pathogenesis of arthritis and SLE. Nat. Rev. Rheumatol. 8, 534–542 (2012).
Habets, K. L. et al. Anti-citrullinated protein antibodies contribute to platelet activation in rheumatoid arthritis. Arthritis Res. Ther. 17, 209 (2015).
Hsu, J. et al. Bruton’s tyrosine kinase mediates platelet receptor-induced generation of microparticles: a potential mechanism for amplification of inflammatory responses in rheumatoid arthritis synovial joints. Immunol. Lett. 150, 97–104 (2013).
Schmitt-Sody, M. et al. In vivo interactions of platelets and leucocytes with the endothelium in murine antigen-induced arthritis: the role of P-selectin. Scand. J. Rheumatol. 36, 311–319 (2007).
Manfredi, A. A. et al. Anti-TNFα agents curb platelet activation in patients with rheumatoid arthritis. Ann. Rheum. Dis. 75, 1511–1520 (2016).
Del Rey, M. J. et al. Clinicopathological correlations of podoplanin (gp38) expression in rheumatoid synovium and its potential contribution to fibroblast platelet crosstalk. PLoS ONE 9, e99607 (2014).
Kehrel, B. Platelet-collagen interactions. Semin. Thromb. Hemost. 21, 123–129 (1995).
Haywood, L. & Walsh, D. A. Vasculature of the normal and arthritic synovial joint. Histol. Histopathol. 16, 277–284 (2001).
Kular, J. K., Basu, S. & Sharma, R. I. The extracellular matrix: structure, composition, age-related differences, tools for analysis and applications for tissue engineering. J. Tissue Eng. 5, 2041731414557112 (2014).
Hakala, M., Risteli, L., Manelius, J., Nieminen, P. & Risteli, J. Increased type I collagen degradation correlates with disease severity in rheumatoid arthritis. Ann. Rheum. Dis. 52, 866–869 (1993).
Kwasny-Krochin, B., Gluszko, P. & Undas, A. Unfavorably altered fibrin clot properties in patients with active rheumatoid arthritis. Thromb. Res. 126, 11–16 (2010).
Scinocca, M. et al. Antihomocitrullinated fibrinogen antibodies are specific to rheumatoid arthritis and frequently bind citrullinated proteins/peptides. J. Rheumatol. 41, 270–279 (2014).
Vadseth, C. et al. Pro-thrombotic state induced by post-translational modification of fibrinogen by reactive nitrogen species. J. Biol. Chem. 279, 8820–8826 (2004).
Türk, S. M. et al. Can we predict thrombotic tendency in rheumatoid arthritis? A thromboelastographic analysis (with ROTEM). Clin. Rheumatol. 37, 2341–2349 (2018).
Di Franco, M. et al. Possible implication of red blood cells in the prothrombotic risk in early rheumatoid arthritis. J. Rheumatol. 42, 1352–1354 (2015).
Andia, I. Rheumatoid arthritis: the ins and outs of platelets in RA. Nat. Rev. Rheumatol. 13, 262–264 (2017).
Uslu, A. U. et al. Two new inflammatory markers associated with Disease Activity Score-28 in patients with rheumatoid arthritis: neutrophil-lymphocyte ratio and platelet-lymphocyte ratio. Int. J. Rheum. Dis. 18, 731–735 (2015).
Zengin, O. et al. New inflammatory markers in early rheumatoid arthritis. Z. Rheumatol. 77, 144–150 (2018).
Rodriguez-Carrio, J. et al. Red cell distribution width is associated with cardiovascular risk and disease parameters in rheumatoid arthritis. Rheumatology 54, 641–646 (2015).
Beinsberger, J., Heemskerk, J. W. & Cosemans, J. M. Chronic arthritis and cardiovascular disease: altered blood parameters give rise to a prothrombotic propensity. Semin. Arthritis Rheum. 44, 345–352 (2014).
Olumuyiwa-Akeredolu, O. O. & Pretorius, E. Rheumatoid arthritis: notable biomarkers linking to chronic systemic conditions and cancer. Curr. Pharm. Des. 22, 918–924 (2016).
Kim, K. W., Kim, B. M., Moon, H. W., Lee, S. H. & Kim, H. R. Role of C-reactive protein in osteoclastogenesis in rheumatoid arthritis. Arthritis Res. Ther. 17, 41 (2015).
Chang, M. K., Binder, C. J., Torzewski, M. & Witztum, J. L. C-Reactive protein binds to both oxidized LDL and apoptotic cells through recognition of a common ligand: phosphorylcholine of oxidized phospholipids. PNAS 99, 13043–13048 (2002).
Sato, A., Oe, K., Yamanaka, H., Yokoyama, I. & Ebina, K. C-Reactive protein specifically enhances platelet-activating factor-induced inflammatory activity in vivo. Eur. J. Pharmacol. 745, 46–51 (2014).
Biro, E. et al. Activated complement components and complement activator molecules on the surface of cell-derived microparticles in patients with rheumatoid arthritis and healthy individuals. Ann. Rheum. Dis. 66, 1085–1092 (2007).
Martinez, M. C., Tesse, A., Zobairi, F. & Andriantsitohaina, R. Shed membrane microparticles from circulating and vascular cells in regulating vascular function. Am. J. Physiol. Heart Circ. Physiol. 288, H1004–H1009 (2005).
Tesse, A. et al. Origin and biological significance of shed-membrane microparticles. Endocr. Metab. Immune Disord. Drug Targets 6, 287–294 (2006).
Connor, D. E., Exner, T., Ma, D. D. & Joseph, J. E. The majority of circulating platelet-derived microparticles fail to bind annexin V, lack phospholipid-dependent procoagulant activity and demonstrate greater expression of glycoprotein Ib. Thromb. Haemost. 103, 1044–1052 (2010).
Ataullakhanov, F. et al. Platelet microparticle membranes have 50- to 100-fold higher specific procoagulant activity than activated platelets. Thromb. Haemost. 97, 425–434 (2017).
Walker, B. et al. Dynamic adhesion of eryptotic erythrocytes to immobilized platelets via platelet phosphatidylserine receptors. Am. J. Physiol. Cell Physiol. 306, C291–C297 (2014).
Knijff-Dutmer, E. A., Koerts, J., Nieuwland, R., Kalsbeek-Batenburg, E. M. & van de Laar, M. A. Elevated levels of platelet microparticles are associated with disease activity in rheumatoid arthritis. Arthritis Rheum. 46, 1498–1503 (2002).
Vinuela-Berni, V. et al. Proportions of several types of plasma and urine microparticles are increased in patients with rheumatoid arthritis with active disease. Clin. Exp. Immunol. 180, 442–451 (2015).
Cloutier, N. et al. The exposure of autoantigens by microparticles underlies the formation of potent inflammatory components: the microparticle-associated immune complexes. EMBO Mol. Med. 5, 235–249 (2013).
Boilard, E. et al. Platelets participate in synovitis via Cox-1-dependent synthesis of prostacyclin independently of microparticle generation. J. Immunol. 186, 4361–4366 (2011).
van Eijk, I. C. et al. Circulating microparticles remain associated with complement activation despite intensive anti-inflammatory therapy in early rheumatoid arthritis. Ann. Rheum. Dis. 69, 1378–1382 (2010).
Jenne, C. N., Urrutia, R. & Kubes, P. Platelets: bridging hemostasis, inflammation, and immunity. Int. J. Lab. Hematol. 35, 254–261 (2013).
Smith, T. L. & Weyrich, A. S. Platelets as central mediators of systemic inflammatory responses. Thromb. Res. 127, 391–394 (2011).
von Hundelshausen, P. & Weber, C. Platelets as immune cells: bridging inflammation and cardiovascular disease. Circ. Res. 100, 27–40 (2007).
Aslam, R. et al. Platelet Toll-like receptor expression modulates lipopolysaccharide-induced thrombocytopenia and tumor necrosis factor-alpha production in vivo. Blood 107, 637–641 (2006).
Andonegui, G. et al. Platelets express functional Toll-like receptor-4. Blood 106, 2417–2423 (2005).
Semple, J. W., Italiano, J. E. Jr & Freedman, J. Platelets and the immune continuum. Nat. Rev. Immunol. 11, 264–274 (2011).
Weyrich, A. S., Lindemann, S. & Zimmerman, G. A. The evolving role of platelets in inflammation. J. Thromb. Haemost. 1, 1897–1905 (2003).
Speth, C. et al. Complement and platelets: mutual interference in the immune network. Mol. Immunol. 67, 108–118 (2015).
Thomas, M. R. & Storey, R. F. The role of platelets in inflammation. Thromb. Haemost. 114, 449–458 (2015).
Elzey, B. D., Ratliff, T. L., Sowa, J. M. & Crist, S. A. Platelet CD40L at the interface of adaptive immunity. Thromb. Res. 127, 180–183 (2011).
Iannacone, M. Platelet-mediated modulation of adaptive immunity. Semin. Immunol. 28, 555–560 (2016).
Morrell, C. N., Aggrey, A. A., Chapman, L. M. & Modjeski, K. L. Emerging roles for platelets as immune and inflammatory cells. Blood 123, 2759–2767 (2014).
Farr, M., Wainwright, A., Salmon, M., Hollywell, C. A. & Bacon, P. A. Platelets in the synovial fluid of patients with rheumatoid arthritis. Rheumatol. Int. 4, 13–17 (1984).
Duchez, A. C. et al. Platelet microparticles are internalized in neutrophils via the concerted activity of 12-lipoxygenase and secreted phospholipase A2-IIA. PNAS 112, E3564–E3573 (2015).
Tamagawa-Mineoka, R. Important roles of platelets as immune cells in the skin. J. Dermatol. Sci. 77, 93–101 (2015).
Meyer, A. et al. Platelet TGF-β1 contributions to plasma TGF-β1, cardiac fibrosis, and systolic dysfunction in a mouse model of pressure overload. Blood 119, 1064–1074 (2012).
Fu, S. et al. TGF-β induces Foxp3 + T-regulatory cells from CD4 + CD25 - precursors. Am. J. Transplant 4, 1614–1627 (2004).
Lichtman, M. K., Otero-Vinas, M. & Falanga, V. Transforming growth factor beta (TGF-β) isoforms in wound healing and fibrosis. Wound Repair Regen. 24, 215–222 (2016).
Gerdes, N. et al. Platelets regulate CD4+ T cell differentiation via multiple chemokines in humans. Thromb. Haemost. 106, 353–362 (2011).
Zhu, L., Huang, Z., Stalesen, R., Hansson, G. K. & Li, N. Platelets provoke distinct dynamics of immune responses by differentially regulating CD4+ T cell proliferation. J. Thromb. Haemost. 12, 1156–1165 (2014).
Kim, S. J. & Jenne, C. N. Role of platelets in neutrophil extracellular trap (NET) production and tissue injury. Semin. Immunol. 28, 546–554 (2016).
Clark, S. R. et al. Platelet TLR4 activates neutrophil extracellular traps to ensnare bacteria in septic blood. Nat. Med. 13, 463–469 (2007).
Page, C. & Pitchford, S. Neutrophil and platelet complexes and their relevance to neutrophil recruitment and activation. Int. Immunopharmacol. 17, 1176–1184 (2013).
Rong, M. Y. et al. Platelets induce a proinflammatory phenotype in monocytes via the CD147 pathway in rheumatoid arthritis. Arthritis Res. Ther. 16, 478 (2014).
Schmidt, R. et al. Extracellular matrix metalloproteinase inducer (CD147) is a novel receptor on platelets, activates platelets, and augments nuclear factor kappaB-dependent inflammation in monocytes. Circ. Res. 102, 302–309 (2008).
Zamora, C. et al. Functional consequences of platelet binding to T lymphocytes in inflammation. J. Leukoc. Biol. 94, 521–529 (2013).
Nurden, A. T. Platelets, inflammation and tissue regeneration. Thromb. Haemost. 105 (Suppl. 1), 13–33 (2011).
Larsen, E. et al. PADGEM protein: a receptor that mediates the interaction of activated platelets with neutrophils and monocytes. Cell 59, 305–312 (1989).
Diacovo, T. G., Puri, K. D., Warnock, R. A., Springer, T. A. & von Andrian, U. H. Platelet-mediated lymphocyte delivery to high endothelial venules. Science 273, 252–255 (1996).
van Gils, J. M., Zwaginga, J. J. & Hordijk, P. L. Molecular and functional interactions among monocytes, platelets, and endothelial cells and their relevance for cardiovascular diseases. J. Leukoc. Biol. 85, 195–204 (2009).
McEver, R. P. Adhesive interactions of leukocytes, platelets, and the vessel wall during hemostasis and inflammation. Thromb. Haemost. 86, 746–756 (2001).
Sreeramkumar, V. et al. Neutrophils scan for activated platelets to initiate inflammation. Science 346, 1234–1238 (2014).
Dinkla, S. et al. Platelet microparticles inhibit IL-17 production by regulatory T cells through P-selectin. Blood 127, 1976–1986 (2016).
Wang, F., Wang, N. S., Yan, C. G., Li, J. H. & Tang, L. Q. The significance of platelet activation in rheumatoid arthritis. Clin. Rheumatol. 26, 768–771 (2007).
Chapman, L. M. et al. Platelets present antigen in the context of MHC class I. J. Immunol. 189, 916–923 (2012).
Zufferey, A. et al. Characterization of the platelet granule proteome: evidence of the presence of MHC1 in alpha-granules. J. Proteomics 101, 130–140 (2014).
Zufferey, A. et al. Mature murine megakaryocytes present antigen-MHC class I molecules to T cells and transfer them to platelets. Blood Adv. 1, 1773–1785 (2017).
Cunin, P. et al. Megakaryocytes compensate for Kit insufficiency in murine arthritis. J. Clin. Invest. 127, 1714–1724 (2017).
Niu, G. & Chen, X. Why integrin as a primary target for imaging and therapy. Theranostics 1, 30–47 (2011).
Jones, J. L. & Walker, R. A. Integrins: a role as cell signalling molecules. Mol. Pathol. 52, 208–213 (1999).
Coller, B. S. αIIbβ3: structure and function. J. Thromb. Haemost. 13 (Suppl. 1), 17–25 (2015).
Bennett, J. S. Structure and function of the platelet integrin αIIbβ3. J. Clin. Invest. 115, 3363–3369 (2005).
Durrant, T. N., van den Bosch, M. T. & Hers, I. Integrin αIIbβ3 outside-in signaling. Blood 130, 1607–1619 (2017).
Li, Z., Delaney, M. K., O’Brien, K. A. & Du, X. Signaling during platelet adhesion and activation. Arterioscler. Thromb. Vasc. Biol. 30, 2341–2349 (2010).
Qiao, J. et al. NLRP3 regulates platelet integrin αIIbβ3 outside-in signaling, hemostasis and arterial thrombosis. Haematologica 103, 1568–1576 (2018).
Boylan, B. et al. Identification of FcγRIIa as the ITAM-bearing receptor mediating αIIbβ3 outside-in integrin signaling in human platelets. Blood 112, 2780–2786 (2008).
Zhi, H. et al. Platelet activation and thrombus formation over IgG immune complexes requires integrin αIIbβ3 and Lyn kinase. PLoS ONE 10, e0135738 (2015).
Lu, W. J. et al. Role of a Janus kinase 2-dependent signaling pathway in platelet activation. Thromb. Res. 133, 1088–1096 (2014).
Arman, M. & Krauel, K. Human platelet IgG Fc receptor FcγRIIA in immunity and thrombosis. J. Thromb. Haemost. 13, 893–908 (2015).
Cloutier, N. et al. Platelets release pathogenic serotonin and return to circulation after immune complex-mediated sequestration. PNAS 115, E1550–E1559 (2018).
Lowin, T. & Straub, R. H. Integrins and their ligands in rheumatoid arthritis. Arthritis Res. Ther. 13, 244 (2011).
Wollheim, F. A. Predictors of joint damage in rheumatoid arthritis. APMIS 104, 81–93 (1996).
Bledzka, K., Smyth, S. S. & Plow, E. F. Integrin αIIbβ3: from discovery to efficacious therapeutic target. Circ. Res. 112, 1189–1200 (2013).
Tung, C. H., Lu, M. C., Lai, N. S. & Wu, S. F. Tumor necrosis factor-alpha blockade treatment decreased CD154 (CD40-ligand) expression in rheumatoid arthritis. PLoS ONE 12, e0183726 (2017).
Guo, Y. et al. CD40L-dependent pathway is active at various stages of rheumatoid arthritis disease progression. J. Immunol. 198, 4490–4501 (2017).
Charafeddine, A. H. et al. Platelet-derived CD154: ultrastructural localization and clinical correlation in organ transplantation. Am. J. Transplant 12, 3143–3151 (2012).
Kyburz, D. et al. Human rheumatoid factor production is dependent on CD40 signaling and autoantigen. J. Immunol. 163, 3116–3122 (1999).
May, F. et al. CLEC-2 is an essential platelet-activating receptor in hemostasis and thrombosis. Blood 114, 3464–3472 (2009).
Navarro-Nunez, L., Langan, S. A., Nash, G. B. & Watson, S. P. The physiological and pathophysiological roles of platelet CLEC-2. Thromb. Haemost. 109, 991–998 (2013).
Gitz, E. et al. CLEC-2 expression is maintained on activated platelets and on platelet microparticles. Blood 124, 2262–2270 (2014).
Mullazehi, M., Mathsson, L., Lampa, J. & Ronnelid, J. High anti-collagen type-II antibody levels and induction of proinflammatory cytokines by anti-collagen antibody-containing immune complexes in vitro characterise a distinct rheumatoid arthritis phenotype associated with acute inflammation at the time of disease onset. Ann. Rheum. Dis. 66, 537–541 (2007).
Peters, M. A. et al. The loss of α2β1 integrin suppresses joint inflammation and cartilage destruction in mouse models of rheumatoid arthritis. Arthritis Rheum. 64, 1359–1368 (2012).
Jung, S. M. et al. Collagen-type specificity of glycoprotein VI as a determinant of platelet adhesion. Platelets 19, 32–42 (2008).
Nieswandt, B. & Watson, S. P. Platelet-collagen interaction: is GPVI the central receptor? Blood 102, 449–461 (2003).
Koivula, M. K. et al. Antibodies binding to citrullinated telopeptides of type I and type II collagens and to mutated citrullinated vimentin synergistically predict the development of seropositive rheumatoid arthritis. Ann. Rheum. Dis. 71, 1666–1670 (2012).
Manivel, V. A. et al. Anti-type II collagen immune complex-induced granulocyte reactivity is associated with joint erosions in RA patients with anti-collagen antibodies. Arthritis Res. Ther. 17, 8 (2015).
Cook, A. D., Rowley, M. J., Mackay, I. R., Gough, A. & Emery, P. Antibodies to type II collagen in early rheumatoid arthritis. Correlation with disease progression. Arthritis Rheum. 39, 1720–1727 (1996).
Schulz, C. et al. Collagen can selectively trigger a platelet secretory phenotype via glycoprotein VI. PLoS ONE 9, e104712 (2014).
Inoue, O. et al. Laminin stimulates spreading of platelets through integrin α6β1-dependent activation of GPVI. Blood 107, 1405–1412 (2006).
Stack, J. R. et al. Soluble glycoprotein VI, a specific marker of platelet activation is increased in the plasma of subjects with seropositive rheumatoid arthritis. PLoS ONE 12, e0188027 (2017).
Alshehri, O. M. et al. Fibrin activates GPVI in human and mouse platelets. Blood 126, 1601–1608 (2015).
Kell, D. B. & Pretorius, E. The simultaneous occurrence of both hypercoagulability and hypofibrinolysis in blood and serum during systemic inflammation, and the roles of iron and fibrin(ogen). Integr. Biol. 7, 24–52 (2015).
So, A. K. et al. Arthritis is linked to local and systemic activation of coagulation and fibrinolysis pathways. J. Thromb. Haemost. 1, 2510–2515 (2003).
Rooney, T. et al. Levels of plasma fibrinogen are elevated in well-controlled rheumatoid arthritis. Rheumatology 50, 1458–1465 (2011).
Induruwa, I. et al. Platelet collagen receptor glycoprotein VI-dimer recognizes fibrinogen and fibrin through their D-domains, contributing to platelet adhesion and activation during thrombus formation. J. Thromb. Haemost. 16, 389–404 (2018).
Seizer, P. et al. EMMPRIN (CD147) is a novel receptor for platelet GPVI and mediates platelet rolling via GPVI-EMMPRIN interaction. Thromb. Haemost. 101, 682–686 (2009).
Wang, C. H. et al. CD147 induces angiogenesis through a vascular endothelial growth factor and hypoxia-inducible transcription factor 1α–mediated pathway in rheumatoid arthritis. Arthritis Rheum. 64, 1818–1827 (2012).
Cloutier, N. et al. Platelets can enhance vascular permeability. Blood 120, 1334–1343 (2012).
Oliver, K. H., Duvernay, M. T., Hamm, H. E. & Carneiro, A. M. Loss of serotonin transporter function alters ADP-mediated glycoprotein αIIbβ3 activation through dysregulation of the 5-HT2A receptor. J. Biol. Chem. 291, 20210–20219 (2016).
Carneiro, A. M., Cook, E. H., Murphy, D. L. & Blakely, R. D. Interactions between integrin αIIbβ3 and the serotonin transporter regulate serotonin transport and platelet aggregation in mice and humans. J. Clin. Invest. 118, 1544–1552 (2008).
Duerschmied, D. et al. Platelet serotonin promotes the recruitment of neutrophils to sites of acute inflammation in mice. Blood 121, 1008–1015 (2013).
Leon-Ponte, M., Ahern, G. P. & O’Connell, P. J. Serotonin provides an accessory signal to enhance T cell activation by signaling through the 5-HT7 receptor. Blood 109, 3139–3146 (2007).
Bernardes, M. et al. Serum serotonin levels and bone in rheumatoid arthritis patients. Rheumatol. Int. 37, 1891–1898 (2017).
Lopez-Vilchez, I., Diaz-Ricart, M., White, J. G., Escolar, G. & Galan, A. M. Serotonin enhances platelet procoagulant properties and their activation induced during platelet tissue factor uptake. Cardiovasc. Res. 84, 309–316 (2009).
Okamoto, K. et al. The role of peripheral 5HT2A and 5HT1A receptors on the orofacial formalin test in rats with persistent temporomandibular joint inflammation. Neuroscience 130, 465–474 (2005).
Kling, A., Rantapaa-Dahlqvist, S., Stenlund, H. & Mjorndal, T. Decreased density of serotonin 5-HT2A receptors in rheumatoid arthritis. Ann. Rheum. Dis. 65, 816–819 (2006).
Snir, O. et al. Genetic variation in the serotonin receptor gene affects immune responses in rheumatoid arthritis. Genes Immun. 14, 83–89 (2013).
D’Atri, L. P. & Schattner, M. Platelet Toll-like receptors in thromboinflammation. Front. Biosci. 22, 1867–1883 (2017).
Panigrahi, S. et al. Engagement of platelet Toll-like receptor 9 by novel endogenous ligands promotes platelet hyperreactivity and thrombosis. Circ. Res. 112, 103–112 (2013).
Koupenova, M. et al. Platelet-TLR7 mediates host survival and platelet count during viral infection in the absence of platelet-dependent thrombosis. Blood 124, 791–802 (2014).
Anabel, A. S. et al. Human platelets express Toll-like receptor 3 and respond to poly I:C. Hum. Immunol. 75, 1244–1251 (2014).
Cognasse, F. et al. Evidence of Toll-like receptor molecules on human platelets. Immunol. Cell Biol. 83, 196–198 (2005).
Shiraki, R. et al. Expression of Toll-like receptors on human platelets. Thromb. Res. 113, 379–385 (2004).
Koupenova, M. et al. Sex differences in platelet toll-like receptors and their association with cardiovascular risk factors. Arterioscler. Thromb. Vasc. Biol. 35, 1030–1037 (2015).
Molteni, M., Gemma, S. & Rossetti, C. The role of Toll-Like Receptor 4 in infectious and noninfectious inflammation. Mediators Inflamm. 2016, 6978936 (2016).
Akira, S. & Takeda, K. Toll-like receptor signalling. Nat. Rev. Immunol. 4, 499–511 (2004).
Zhang, G. et al. Lipopolysaccharide stimulates platelet secretion and potentiates platelet aggregation via TLR4/MyD88 and the cGMP-dependent protein kinase pathway. J. Immunol. 182, 7997–8004 (2009).
Vogel, S. et al. Platelet-derived HMGB1 is a critical mediator of thrombosis. J. Clin. Invest. 125, 4638–4654 (2015).
Rivadeneyra, L. et al. Regulation of platelet responses triggered by Toll-like receptor 2 and 4 ligands is another non-genomic role of nuclear factor-kappaB. Thromb. Res. 133, 235–243 (2014).
Vieira-de-Abreu, A., Campbell, R. A., Weyrich, A. S. & Zimmerman, G. A. Platelets: versatile effector cells in hemostasis, inflammation, and the immune continuum. Semin. Immunopathol. 34, 5–30 (2012).
Shashkin, P. N., Brown, G. T., Ghosh, A., Marathe, G. K. & McIntyre, T. M. Lipopolysaccharide is a direct agonist for platelet RNA splicing. J. Immunol. 181, 3495–3502 (2008).
Rondina, M. T. et al. The septic milieu triggers expression of spliced tissue factor mRNA in human platelets. J. Thromb. Haemost. 9, 748–758 (2011).
Cheng, N., He, R., Tian, J., Ye, P. P. & Ye, R. D. Cutting edge: TLR2 is a functional receptor for acute-phase serum amyloid A. J. Immunol. 181, 22–26 (2008).
Blair, P. et al. Stimulation of Toll-like receptor 2 in human platelets induces a thromboinflammatory response through activation of phosphoinositide 3-kinase. Circ. Res. 104, 346–354 (2009).
Huang, Q. Q. & Pope, R. M. The role of Toll-like receptors in rheumatoid arthritis. Curr. Rheumatol. Rep. 11, 357–364 (2009).
Iwahashi, M. et al. Expression of Toll-like receptor 2 on CD16+ blood monocytes and synovial tissue macrophages in rheumatoid arthritis. Arthritis Rheum. 50, 1457–1467 (2004).
Radstake, T. R. et al. Expression of Toll-like receptors 2 and 4 in rheumatoid synovial tissue and regulation by proinflammatory cytokines interleukin-12 and interleukin-18 via interferon-gamma. Arthritis Rheum. 50, 3856–3865 (2004).
Roelofs, M. F. et al. The expression of Toll-like receptors 3 and 7 in rheumatoid arthritis synovium is increased and costimulation of Toll-like receptors 3, 4, and 7/8 results in synergistic cytokine production by dendritic cells. Arthritis Rheum. 52, 2313–2322 (2005).
Taniguchi, N. et al. High mobility group box chromosomal protein 1 plays a role in the pathogenesis of rheumatoid arthritis as a novel cytokine. Arthritis Rheum. 48, 971–981 (2003).
Raijmakers, R. et al. Elevated levels of fibrinogen-derived endogenous citrullinated peptides in synovial fluid of rheumatoid arthritis patients. Arthritis Res. Ther. 14, R114 (2012).
Kimura, E. et al. Identification of citrullinated cellular fibronectin in synovial fluid from patients with rheumatoid arthritis. Mod. Rheumatol. 24, 766–769 (2014).
Chang, X., Yamada, R. & Yamamoto, K. Inhibition of antithrombin by hyaluronic acid may be involved in the pathogenesis of rheumatoid arthritis. Arthritis Res. Ther. 7, R268–R273 (2005).
Brentano, F., Kyburz, D. & Gay, S. Toll-like receptors and rheumatoid arthritis. Methods Mol. Biol. 517, 329–343 (2009).
Kim, K. W. et al. Human rheumatoid synovial fibroblasts promote osteoclastogenic activity by activating RANKL via TLR-2 and TLR-4 activation. Immunol. Lett. 110, 54–64 (2007).
Fischer, A. et al. The involvement of Toll-like receptor 9 in the pathogenesis of erosive autoimmune arthritis. J. Cell. Mol. Med. 22, 4399–4409 (2018).
Sacre, S. M. et al. The Toll-like receptor adaptor proteins MyD88 and Mal/TIRAP contribute to the inflammatory and destructive processes in a human model of rheumatoid arthritis. Am. J. Pathol. 170, 518–525 (2007).
Ospelt, C. et al. Overexpression of Toll-like receptors 3 and 4 in synovial tissue from patients with early rheumatoid arthritis: Toll-like receptor expression in early and longstanding arthritis. Arthritis Rheum. 58, 3684–3692 (2008).
Israels, S. J. & McMillan-Ward, E. M. CD63 modulates spreading and tyrosine phosphorylation of platelets on immobilized fibrinogen. Thromb. Haemost. 93, 311–318 (2005).
Israels, S. J., McMillan-Ward, E. M., Easton, J., Robertson, C. & McNicol, A. CD63 associates with the alphaIIb beta3 integrin-CD9 complex on the surface of activated platelets. Thromb. Haemost. 85, 134–141 (2001).
Arntz, O. J. et al. Rheumatoid arthritis patients with circulating extracellular vesicles positive for IgM rheumatoid factor have higher disease activity. Front. Immunol. 9, 2388 (2018).
Marjoram, R. J. et al. Suboptimal activation of protease-activated receptors enhances α2β1 integrin-mediated platelet adhesion to collagen. J. Biol. Chem. 284, 34640–34647 (2009).
Russell, F. A. & McDougall, J. J. Proteinase activated receptor (PAR) involvement in mediating arthritis pain and inflammation. Inflamm Res. 58, 119–126 (2009).
Duvernay, M., Young, S., Gailani, D., Schoenecker, J. & Hamm, H. E. Protease-activated receptor (PAR) 1 and PAR4 differentially regulate factor V expression from human platelets. Mol. Pharmacol. 83, 781–792 (2013).
Trumel, C. et al. A key role of adenosine diphosphate in the irreversible platelet aggregation induced by the PAR1-activating peptide through the late activation of phosphoinositide 3-kinase. Blood 94, 4156–4165 (1999).
Steinhoff, M. et al. Proteinase-activated receptors: transducers of proteinase-mediated signaling in inflammation and immune response. Endocr. Rev. 26, 1–43 (2005).
Fender, A. C., Rauch, B. H., Geisler, T. & Schror, K. Protease-activated receptor PAR-4: An inducible switch between thrombosis and vascular inflammation? Thromb. Haemost. 117, 2013–2025 (2017).
Weinberg, J. B., Pippen, A. M. M. & Greenberg, C. S. Extravascular fibrin formation and dissolution in synovial tissue of patients with osteoarthritis and rheumatoid arthritis. Arthritis Rheum. 34, 996–1005 (1991).
Stephens, G. et al. Platelet activation induces metalloproteinase-dependent GP VI cleavage to down-regulate platelet reactivity to collagen. Blood 105, 186–191 (2005).
Ramachandran, R., Noorbakhsh, F., Defea, K. & Hollenberg, M. D. Targeting proteinase-activated receptors: therapeutic potential and challenges. Nat. Rev. Drug Discov. 11, 69–86 (2012).
Gachet, C. ADP receptors of platelets and their inhibition. Thromb. Haemost. 86, 222–232 (2001).
Jung, S. M. & Moroi, M. Platelet collagen receptor integrin α2β1 activation involves differential participation of ADP-receptor subtypes P2Y1 and P2Y12 but not intracellular calcium change. Eur. J. Biochem. 268, 3513–3522 (2001).
Koessler, J. et al. The role of agonist-induced activation and inhibition for the regulation of purinergic receptor expression in human platelets. Thromb. Res. 168, 40–46 (2018).
Becker, L. V. et al. Activities of enzymes that hydrolyze adenine nucleotides in platelets from patients with rheumatoid arthritis. Clin. Biochem. 43, 1096–1100 (2010).
Aburima, A. et al. cAMP signaling regulates platelet myosin light chain (MLC) phosphorylation and shape change through targeting the RhoA-Rho kinase-MLC phosphatase signaling pathway. Blood 122, 3533–3545 (2013).
Aslan, J. E. & McCarty, O. J. Rho GTPases in platelet function. J. Thromb. Haemost. 11, 35–46 (2013).
Ajeganova, S. et al. Longitudinal levels of apolipoproteins and antibodies against phosphorylcholine are independently associated with carotid artery atherosclerosis 5 years after rheumatoid arthritis onset—a prospective cohort study. Rheumatology 50, 1785–1793 (2011).
Szekanecz, Z., Vegvari, A., Szabo, Z. & Koch, A. E. Chemokines and chemokine receptors in arthritis. Front. Biosci. (Schol. Ed.) 2, 153–167 (2010).
Koopman, F. A. et al. Autonomic dysfunction precedes development of rheumatoid arthritis: a prospective cohort study. EBioMedicine 6, 231–237 (2016).
Bahlas, S. et al. Rapid immunoprofiling of cytokines, chemokines and growth factors in patients with active rheumatoid arthritis using Luminex Multiple Analyte Profiling technology for precision medicine. Clin. Exp. Rheumatol. 27, 112–119 (2018).
Zhang, Y. et al. A novel circulating miRNA-based model predicts the response to tripterysium glycosides tablets: moving toward model-based precision medicine in rheumatoid arthritis. Front. Pharmacol. 9, 378 (2018).
Ragouzeos, D. et al. “Am I OK?” using human centered design to empower rheumatoid arthritis patients through patient reported outcomes. Patient Educ. Couns. https://doi.org/10.1016/j.pec.2018.10.016 (2018).
Machlus, K. R. & Italiano, J. E. Jr. The incredible journey: from megakaryocyte development to platelet formation. J. Cell Biol. 201, 785–796 (2013).
Lefrancais, E. et al. The lung is a site of platelet biogenesis and a reservoir for haematopoietic progenitors. Nature 544, 105–109 (2017).
Jin, J. & Kunapuli, S. P. Coactivation of two different G protein-coupled receptors is essential for ADP-induced platelet aggregation. PNAS 95, 8070–8074 (1998).
Shen, Y. et al. Requirement of leucine-rich repeats of glycoprotein (GP) Ibα for shear-dependent and static binding of von Willebrand factor to the platelet membrane GP Ib-IX-V complex. Blood 95, 903–910 (2000).
Bester, J. & Pretorius, E. Effects of IL-1β, IL-6 and IL-8 on erythrocytes, platelets and clot viscoelasticity. Sci. Rep. 6, 32188 (2016).
Acknowledgements
The work of the authors was funded by the South African Medical Research Council and the National Research Foundation (South Africa; grant number N00345 to E.P.).
Review criteria
A search for original articles published between 1984 and 2018 without language restriction was performed in PubMed, MEDLINE, Google Scholar and Scopus. The search terms used were “platelet rheumatoid arthritis”, “platelet ligands”, “platelet microparticle”, “platelet uptake and secretion”, “platelet receptors”, “platelet signalling pathways” and “immune functions”. All articles identified were reviewed manually for eligibility.
Reviewer information
Nature Reviews Rheumatology thanks J. Semple and the other anonymous reviewer(s), for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
O.O.-A. researched data for the article. O.O.-A. and E.P. provided substantial contributions to discussion of content. O.O.-A., M.J.P. and E.P. wrote the article and all authors reviewed and/or edited the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Plateletcrit
-
A measure of the number of platelets present in a blood fraction.
- Platelet distribution width
-
A blood parameter that measures the variability in platelet size distribution.
- Mean platelet volume
-
A measure of the average size of platelets within a blood sample.
- Platelet spreading
-
An indicator of cellular activation that involves reorganization of the actin cytoskeleton to extend filopodia and lamellipodia, thereby converting platelets from biconcave to amorphous cells.
Rights and permissions
About this article
Cite this article
Olumuyiwa-Akeredolu, Oo., Page, M.J., Soma, P. et al. Platelets: emerging facilitators of cellular crosstalk in rheumatoid arthritis. Nat Rev Rheumatol 15, 237–248 (2019). https://doi.org/10.1038/s41584-019-0187-9
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41584-019-0187-9
This article is cited by
-
Prodrug-based nanomedicines for rheumatoid arthritis
Discover Nano (2024)
-
Platelets and platelet-derived vesicles as an innovative cellular and subcellular platform for managing multiple sclerosis
Molecular Biology Reports (2023)
-
Hematological markers and ultrasound 7-joint inflammation score as add-on tools in the assessment of inflammation in rheumatoid arthritis patients
Egyptian Rheumatology and Rehabilitation (2022)
-
Deposition of platelet-derived microparticles in podocytes contributes to diabetic nephropathy
International Urology and Nephrology (2022)
-
Flavored Food Additives on the Leaves of Piper betle L.: A Human Health Perspective
Applied Biochemistry and Biotechnology (2022)