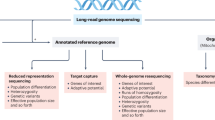
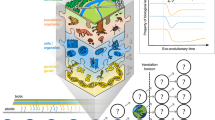
Abstract
Billions of hectares of natural ecosystems have been degraded through human actions. The global community has agreed on targets to halt and reverse these declines, and the restoration sector faces the important but arduous task of implementing programmes to meet these objectives. Existing and emerging genomics tools offer the potential to improve the odds of achieving these targets. These tools include population genomics that can improve seed sourcing, meta-omics that can improve assessment and monitoring of restoration outcomes, and genome editing that can generate novel genotypes for restoring challenging environments. We identify barriers to adopting these tools in a restoration context and emphasize that regulatory and ethical frameworks are required to guide their use.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Ceballos, G. et al. Accelerated modern human-induced species losses: entering the sixth mass extinction. Sci. Adv. 1, e1400253 (2015).
Gibbs, H. K. & Salmon, J. M. Mapping the world’s degraded lands. Appl. Geogr. 57, 12–21 (2015).
Suding, K. et al. Committing to ecological restoration. Science 348, 638–640 (2015).
Iftekhar, M. S. et al. How economics can further the success of ecological restoration. Conserv. Biol. 31, 261–268 (2017).
Crouzeilles, R. et al. A global meta-analysis on the ecological drivers of forest restoration success. Nat. Commun. 7, 11666 (2016).
Wortley, L., Hero, J. M. & Howes, M. Evaluating ecological restoration success: a review of the literature. Restor. Ecol. 21, 537–543 (2013).
Broadhurst, L. M. et al. Maximizing seed resources for restoration in an uncertain future. Bioscience 66, 73–79 (2016).
Nevill, P. G., Cross, A. T. & Dixon, K. W. Ethical seed sourcing is a key issue in meeting global restoration targets. Curr. Biol. 28, R1378–R1379 (2018).
Collen, B. & Nicholson, E. Taking the measure of change. Science 346, 166–167 (2014).
Gellie, N. et al. Networked and embedded scientific experiments in restoration will improve outcomes. Front. Ecol. Environ. 16, 288–294 (2018).
Perring, M. P. et al. Advances in restoration ecology: rising to the challenges of the coming decades. Ecosphere 6, 1–25 (2015).
Wetterstrand, K. A. The cost of sequencing a human genome. NHGRI http://www.genome.gov/sequencingcosts (2016).
Shafer, A. B. et al. Genomics and the challenging translation into conservation practice. Trends Ecol. Evol. 30, 78–87 (2015).
Luikart, G. et al. The power and promise of population genomics: from genotyping to genome typing. Nat. Rev. Genet. 4, 981 (2003).
Ellegren, H. Genome sequencing and population genomics in non-model organisms. Trends Ecol. Evol. 29, 51–63 (2014).
Funk, W. C. et al. Harnessing genomics for delineating conservation units. Trends Ecol. Evol. 27, 489–496 (2012).
Ji, Y. et al. Reliable, verifiable and efficient monitoring of biodiversity via metabarcoding. Ecol. Lett. 16, 1245–1257 (2013).
Bohmann, K. et al. Environmental DNA for wildlife biology and biodiversity monitoring. Trends Ecol. Evol. 29, 358–367 (2014).
Prowse, T. A. A. et al. Dodging silver bullets: good CRISPR gene-drive design is critical for eradicating exotic vertebrates. Proc. R. Soc. B 284, 20170799 (2017).
Webber, B. L., Raghu, S. & Edwards, O. R. Opinion: is CRISPR-based gene drive a biocontrol silver bullet or global conservation threat? Proc. Natl Acad. Sci. USA 112, 10565–10567 (2015).
Moro, D. et al. Identifying knowledge gaps for gene drive research to control invasive animal species: the next CRISPR step. Glob. Ecol. Conserv. 13, e00363 (2018).
Williams, A. V., Nevill, P. G. & Krauss, S. L. Next generation restoration genetics: applications and opportunities. Trends Plant Sci. 19, 529–537 (2014).
Mijangos, J. L. et al. Contribution of genetics to ecological restoration. Mol. Ecol. 24, 22–37 (2015).
Vranckx, G. et al. Meta-analysis of susceptibility of woody plants to loss of genetic diversity through habitat fragmentation. Conserv. Biol. 26, 228–237 (2011).
Breed, M. et al. Mating patterns and pollinator mobility are critical traits in forest fragmentation genetics. Heredity 105, 108–114 (2015).
Pecl, G. T. et al. Biodiversity redistribution under climate change: impacts on ecosystems and human well-being. Science 355, eaai9214 (2017).
Aitken, S. N. & Whitlock, M. C. Assisted gene flow to facilitate local adaptation to climate change. Annu. Rev. Ecol. Evol. Syst. 44, 367 (2013).
Harrison, P. A. et al. Integrating climate change and habitat fragmentation to identify candidate seed sources for ecological restoration. Restor. Ecol. 25, 524–531 (2017).
Breed, M. et al. Priority actions to improve provenance decision-making. Bioscience 68, 510–516 (2018).
Kawecki, T. J. & Ebert, D. Conceptual issues in local adaptation. Ecol. Lett. 7, 1225–1241 (2004).
Yang, J. et al. The development of universal response functions to facilitate climate-smart regeneration of black spruce and white pine in Ontario, Canada. For. Ecol. Manage. 339, 34–43 (2015).
Wang, T., O’Neill, G. A. & Aitken, S. N. Integrating environmental and genetic effects to predict responses of tree populations to climate. Ecol. Appl. 20, 153–163 (2010).
Wang, S. et al. 2b-RAD: a simple and flexible method for genome-wide genotyping. Nat. Methods 9, 808 (2012).
Christmas, M. et al. Finding needles in a genomic haystack: targeted capture identifies clear signatures of selection in a non-model plant species. Mol. Ecol. 25, 4216–4233 (2016).
Deleury, E. et al. An evaluation of pool-sequencing transcriptome-based exon capture for population genomics of non-model species. Preprint at bioRxiv https://www.biorxiv.org/content/10.1101/583534v2 (2019).
Nuzhdin, S. V., Friesen, M. L. & McIntyre, L. M. Genotype–phenotype mapping in a post-GWAS world. Trends Genet. 28, 421–426 (2012).
Supple, M. A. et al. Landscape genomic prediction for restoration of a Eucalyptus foundation species under climate change. eLife 7, e31835 (2018).
Dick, C. W. et al. Spatial scales of pollen and seed-mediated gene flow in tropical rain forest trees. Trop. Plant Biol. 1, 20–33 (2008).
Hardy, O. J. et al. Fine-scale genetic structure and gene dispersal inferences in 10 neotropical tree species. Mol. Ecol. 15, 559–571 (2006).
Broadhurst, L. M. et al. Seed supply for broadscale restoration: maximizing evolutionary potential. Evol. Appl. 1, 587–597 (2008).
Leimu, R. & Fischer, M. A meta-analysis of local adaptation in plants. PLOS ONE 3, e4010 (2008).
Yeaman, S. et al. Convergent local adaptation to climate in distantly related conifers. Science 353, 1431–1433 (2016).
Steane, D. A. et al. Genome-wide scans reveal cryptic population structure in a dry-adapted eucalypt. Tree Genet. Genomes 11, 33 (2015).
Fitzpatrick, M. C. & Keller, S. R. Ecological genomics meets community-level modelling of biodiversity: mapping the genomic landscape of current and future environmental adaptation. Ecol. Lett. 18, 1–16 (2015).
Manel, S. et al. Predicting genotype environmental range from genome–environment associations. Mol. Ecol. 27, 2823–2833 (2018).
Bay, R. A. et al. Genomic signals of selection predict climate-driven population declines in a migratory bird. Science 359, 83–86 (2018).
Rossetto, M. et al. Restore and Renew: a genomics-era framework for species provenance delimitation. Restor. Ecol. 27, 538–548 (2018).
Steane, D. A. et al. Genome-wide scans detect adaptation to aridity in a widespread forest tree species. Mol. Ecol. 23, 2500–2513 (2014).
Rockman, M. V. The QTN program and the alleles that matter for evolution: all that’s gold does not glitter. Evolution 66, 1–17 (2012).
Price, N. et al. Combining population genomics and fitness QTLs to identify the genetics of local adaptation in Arabidopsis thaliana. Proc. Natl Acad. Sci. USA 115, 5028–5033 (2018).
Kettle, C. J. et al. Cryptic genetic bottlenecks during restoration of an endangered tropical conifer. Biol. Conserv. 141, 1953–1961 (2008).
Leberg, P. L. Effects of population bottlenecks on genetic diversity as measured by allozyme electrophoresis. Evolution 46, 477–494 (1992).
García, C., Guichoux, E. & Hampe, A. A comparative analysis between SNPs and SSRs to investigate genetic variation in a juniper species (Juniperus phoenicea ssp. turbinata). Tree Genet. Genomes 14, 87 (2018).
Fischer, M. C. et al. Estimating genomic diversity and population differentiation—an empirical comparison of microsatellite and SNP variation in Arabidopsis halleri. BMC Genomics 18, 69 (2017).
Jordan, R. et al. Landscape genomics reveals altered genome wide diversity within revegetated stands of Eucalyptus microcarpa (Grey Box). New Phytol. 212, 992–1006 (2016).
Yan, D. et al. High-throughput eDNA monitoring of fungi to track functional recovery in ecological restoration. Biol. Conserv. 217, 113–120 (2018).
Cristescu, M. E. & Hebert, P. D. Uses and misuses of environmental DNA in biodiversity science and conservation. Annu. Rev. Ecol. Evol. Syst. 49, 209–230 (2018).
Christine, M. Microbial diversity unbound: what DNA-based techniques are revealing about the planet’s hidden biodiversity. AIBS Bull. 54, 1064–1068 (2004).
Pace, N. R. A molecular view of microbial diversity and the biosphere. Science 276, 734–740 (1997).
Ciais, P. et al. in Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change (eds Stocker, T. F. et al.) 465–570 (Cambridge Univ. Press, 2014).
Livne-Luzon, S. et al. Small-scale spatial variability in the distribution of ectomycorrhizal fungi affects plant performance and fungal diversity. Ecol. Lett. 20, 1192–1202 (2017).
Clemmensen, K. E. et al. Roots and associated fungi drive long-term carbon sequestration in boreal forest. Science 339, 1615–1618 (2013).
Mills, J. G. et al. Relating urban biodiversity to human health with the ‘holobiont’ concept. Front. Microbiol. 10, 550 (2019).
Mills, J. et al. Urban habitat restoration provides a human health benefit through microbiome rewilding: the Microbiome Rewilding Hypothesis. Restor. Ecol. 25, 866–872 (2017).
Robinson, J., Mills, J. & Breed, M. F. Walking ecosystems in microbiome-inspired green infrastructure: an ecological perspective on enhancing personal and planetary health. Challenges 9, 40 (2018).
Liddicoat, C. et al. Can bacterial indicators of a grassy woodland restoration inform ecosystem assessment and microbiota-mediated human health? Environ. Int. 129, 105–117 (2019).
Gellie, N. J. et al. Revegetation rewilds the soil bacterial microbiome of an old field. Mol. Ecol. 26, 2895–2904 (2017).
Fierer, N. et al. Comparative metagenomic, phylogenetic and physiological analyses of soil microbial communities across nitrogen gradients. ISME J. 6, 1007–1017 (2012).
Hua, Z.-S. et al. Ecological roles of dominant and rare prokaryotes in acid mine drainage revealed by metagenomics and metatranscriptomics. ISME J. 9, 1280 (2015).
Jacoby, R. et al. The role of soil microorganisms in plant mineral nutrition—current knowledge and future directions. Front. Plant Sci. 8, 1617 (2017).
Larsen, P. E. et al. Predicted Relative Metabolomic Turnover (PRMT): determining metabolic turnover from a coastal marine metagenomic dataset. Microb. Inform. Exp. 1, 4 (2011).
Faust, K. & Raes, J. Microbial interactions: from networks to models. Nat. Rev. Microbiol. 10, 538 (2012).
Cerco, C. F. & Noel, M. R. Can oyster restoration reverse cultural eutrophication in Chesapeake Bay? Estuaries Coast 30, 331–343 (2007).
Higgins, C. B. et al. Effect of aquacultured oyster biodeposition on sediment N2 production in Chesapeake Bay. Mar. Ecol. Prog. Ser. 473, 7–27 (2013).
Dandie, C. E. et al. Abundance, diversity and functional gene expression of denitrifier communities in adjacent riparian and agricultural zones. FEMS Microbiol. Ecol. 77, 69–82 (2011).
Klein, A. M. et al. Importance of pollinators in changing landscapes for world crops. Proc. R. Soc. B 274, 303–313 (2007).
Ollerton, J., Winfree, R. & Tarrant, S. How many flowering plants are pollinated by animals? Oikos 120, 321–326 (2011).
Kaiser-Bunbury, C. N. et al. Determinants of the microstructure of plant–pollinator networks. Ecology 95, 3314–3324 (2014).
Lebuhn, G. et al. Detecting insect pollinator declines on regional and global scales. Conserv. Biol. 27, 113–120 (2013).
Griffin, S. R. et al. Wild bee community change over a 26-year chronosequence of restored tallgrass prairie. Restor. Ecol. 25, 650–660 (2017).
Yu, D. W. et al. Biodiversity soup: metabarcoding of arthropods for rapid biodiversity assessment and biomonitoring. Methods Ecol. Evol. 3, 613–623 (2012).
Richardson, R. T. et al. Application of ITS2 metabarcoding to determine the provenance of pollen collected by honey bees in an agroecosystem. Appl. Plant Sci. 3, 1400066 (2015).
de Vere, N. et al. Using DNA metabarcoding to investigate honey bee foraging reveals limited flower use despite high floral availability. Sci. Rep. 7, 42838 (2017).
Bell, K. L. et al. Applying pollen DNA metabarcoding to the study of plant–pollinator interactions. Appl. Plant Sci. 5, 1600124 (2017).
Pornon, A. et al. DNA metabarcoding data unveils invisible pollination networks. Sci. Rep. 7, 16828 (2017).
Corby-Harris, V., Maes, P. & Anderson, K. E. The bacterial communities associated with honey bee (Apis mellifera) foragers. PLOS ONE 9, e95056 (2014).
Tang, M. et al. High-throughput monitoring of wild bee diversity and abundance via mitogenomics. Methods Ecol. Evol. 6, 1034–1043 (2015).
Zhou, X. et al. Ultra-deep sequencing enables high-fidelity recovery of biodiversity for bulk arthropod samples without PCR amplification. Gigascience 2, 4 (2013).
Liu, S. et al. Mitochondrial capture enriches mito-DNA 100 fold, enabling PCR-free mitogenomics biodiversity analysis. Mol. Ecol. Res. 16, 470–479 (2016).
Bot, A., Nachtergaele, F. & Young, A. Land Resource Potential and Constraints at Regional and Country Levels (Food and Agriculture Organization of the United Nations, 2000).
Oldeman, L. R., Hakkeling, R. U. & Sombroek, W. G. World Map of The Status of Human-Induced Soil Degradation: An Explanatory Note (ISRIC, 1991).
Breed, M. F., Mortimer, P. E. & Lowe, A. J. Restoration: ‘garden of eden’ unrealistic. Nature 533, 469 (2016).
Hobbs, R. J. et al. Novel ecosystems: theoretical and management aspects of the new ecological world order. Glob. Ecol. Biogeogr. Lett. 15, 1–7 (2006).
Gupta, R. M. & Musunuru, K. Expanding the genetic editing tool kit: ZFNs, TALENs, and CRISPR-Cas9. J. Clin. Invest. 124, 4154–4161 (2014).
Pourcel, C., Salvignol, G. & Vergnaud, G. CRISPR elements in Yersinia pestis acquire new repeats by preferential uptake of bacteriophage DNA, and provide additional tools for evolutionary studies. Microbiology 151, 653–663 (2005).
Nishimasu, H. et al. Crystal structure of Cas9 in complex with guide RNA and target DNA. Cell 156, 935–949 (2014).
Hsu, P. D., Lander, E. S. & Zhang, F. Development and applications of CRISPR-Cas9 for genome engineering. Cell 157, 1262–1278 (2014).
Piaggio, A. J. et al. Is it time for synthetic biodiversity conservation? Trends Ecol. Evol. 32, 97–107 (2017).
McFarlane, G. R., Whitelaw, C. B. A. & Lillico, S. G. CRISPR-based gene drives for pest control. Trends Biotechnol. 36, 130–133 (2018).
Chiu, H. et al. Transgene-free genome editing in Caenorhabditis elegans using CRISPR-Cas. Genetics 195, 1167–1171 (2013).
Gratz, S. J. et al. in CRISPR 335–348 (Springer, 2015).
Popkin, G. Can a transgenic chestnut restore a forest icon? Science 361, 830–831 (2018).
Mei, Y. et al. Recent progress in CRISPR/Cas9 technology. J. Genet. Genomics 43, 63–75 (2016).
Arora, L. & Narula, A. Gene editing and crop improvement using CRISPR-Cas9 system. Front. Plant Sci. 8, 1932 (2017).
Boyle, E. A., Li, Y. I. & Pritchard, J. K. An expanded view of complex traits: from polygenic to omnigenic. Cell 169, 1177–1186 (2017).
Newhouse, A. E. et al. Transgenic American chestnuts show enhanced blight resistance and transmit the trait to T1 progeny. Plant Sci. 228, 88–97 (2014).
Chilcoat, D., Liu, Z.-B. & Sander, J. in Progress in Molecular Biology and Translational Science Vol. 149 (eds Weeks, W. & Yang, B.) 27–46 (Academic Press, 2017).
Shi, J. et al. ARGOS8 variants generated by CRISPR-Cas9 improve maize grain yield under field drought stress conditions. Plant Biotechnol. J. 15, 207–216 (2017).
Borrelli, V. M. et al. The enhancement of plant disease resistance using CRISPR/Cas9 technology. Front. Plant Sci. 9, 1245 (2018).
Deredec, A., Burt, A. & Godfray, H. C. J. The population genetics of using homing endonuclease genes in vector and pest management. Genetics 179, 2013–2026 (2008).
Burt, A. Site-specific selfish genes as tools for the control and genetic engineering of natural populations. Proc. R. Soc. B 270, 921–928 (2003).
Morita, T. et al. Evolution of the mouse t haplotype: recent and worldwide introgression to Mus musculus. Proc. Natl Acad. Sci. USA 89, 6851–6855 (1992).
Gallardo, T. et al. Generation of a germ cell-specific mouse transgenic Cre line, Vasa-Cre. Genesis 45, 413–417 (2007).
Hammond, A. et al. A CRISPR-Cas9 gene drive system targeting female reproduction in the malaria mosquito vector Anopheles gambiae. Nat. Biotechnol. 34, 78–83 (2016).
Deredec, A., Godfray, H. C. J. & Burt, A. Requirements for effective malaria control with homing endonuclease genes. Proc. Natl Acad. Sci. USA 108, E874–E880 (2011).
Eckhoff, P. A. et al. Impact of mosquito gene drive on malaria elimination in a computational model with explicit spatial and temporal dynamics. Proc. Natl Acad. Sci. USA 114, E255–E264 (2016).
Kyrou, K. et al. A CRISPR–Cas9 gene drive targeting doublesex causes complete population suppression in caged Anopheles gambiae mosquitoes. Nat. Biotechnol. 36, 1062 (2018).
Hodgins, K. A., Rieseberg, L. & Otto, S. P. Genetic control of invasive plants species using selfish genetic elements. Evol. Appl. 2, 555–569 (2009).
D’antonio, C. & Meyerson, L. A. Exotic plant species as problems and solutions in ecological restoration: a synthesis. Restor. Ecol. 10, 703–713 (2002).
Drury, D. W. et al. CRISPR/Cas9 gene drives in genetically variable and nonrandomly mating wild populations. Sci. Adv. 3, e1601910 (2017).
Carvalho, D. O. et al. Suppression of a field population of Aedes aegypti in Brazil by sustained release of transgenic male mosquitoes. PLOS Negl. Trop. Dis. 9, e0003864 (2015).
Noble, C. et al. Current CRISPR gene drive systems are likely to be highly invasive in wild populations. eLife 7, e33423 (2018).
Reed, F. A. CRISPR/Cas9 gene drive: growing pains for a new technology. Genetics 205, 1037–1039 (2017).
Zentner, G. E. & Wade, M. J. The promise and peril of CRISPR gene drives: genetic variation and inbreeding may impede the propagation of gene drives based on the CRISPR genome editing technology. Bioessays 39, 1700109 (2017).
National Academies of Sciences, Engineering, and Medicine. Forest Health and Biotechnology: Possibilities and Considerations (National Academies Press, 2019).
Stirling, A., Hayes, K. & Delborne, J. Towards inclusive social appraisal: risk, participation and democracy in governance of synthetic biology. BMC Proc. 12, 15 (2018).
Newhouse, A. E. et al. Transgenic American chestnuts do not inhibit germination of native seeds or colonization of mycorrhizal fungi. Front. Plant Sci. 9, 1046 (2018).
Barnhill-Dilling, S. K. & Delborne, J. A. The genetically engineered American chestnut tree as opportunity for reciprocal restoration in Haudenosaunee communities. Biol. Conserv. 232, 1–9 (2019).
Strauss, S. H., Costanza, A. & Séguin, A. Genetically engineered trees: paralysis from good intentions. Science 349, 794–795 (2015).
Koop, A. L. et al. Development and validation of a weed screening tool for the United States. Biol. Invasions 14, 273–294 (2012).
National Academies of Sciences, Engineering, and Medicine. Genetically Engineered Crops: Experiences and Prospects (National Academies Press, 2016).
Hayes, K. et al. in Environmental Risk Assessment of Genetically Modified Organisms Vol. 3 Ch. 1 (eds Kapuscinski, A. R. et al.) 1–28 (CABI, 2007).
Sun, T. Y. et al. Dynamic probabilistic modeling of environmental emissions of engineered nanomaterials. Environ. Sci. Technol. 50, 4701–4711 (2016).
Hayes, K., Regan, H. & Burgman, M. in Environmental Risk Assessment of Genetically Modified Organisms Vol. 3 (eds Kapuscinski, A. R. et al.) 188–208 (CABI, 2007).
Häggman, H. et al. Genetically engineered trees for plantation forests: key considerations for environmental risk assessment. Plant Biotechnol. J. 11, 785–798 (2013).
National Academies of Sciences, Engineering, and Medicine. Preparing for Future Products of Biotechnology (National Academies Press, 2017).
Hur, M. et al. Effect of genetically modified poplars on soil microbial communities during the phytoremediation of waste mine tailings. Appl. Environ. Microbiol. 77, 7611–7619 (2011).
Munns, W. R. Jr et al. Ecosystem services as assessment endpoints for ecological risk assessment. Integr. Environ. Assess. Manag. 12, 522–528 (2016).
Jimenez-Sanchez, G. Genomics innovation: transforming healthcare, business, and the global economy. Genome 58, 511–517 (2015).
Brancalion, P. H. & van Melis, J. On the need for innovation in ecological restoration. Ann. Mo. Bot. Gard. 102, 227–236 (2017).
Matzek, V., Gornish, E. S. & Hulvey, K. B. Emerging approaches to successful ecological restoration: five imperatives to guide innovation. Restor. Ecol. 25, S110–S113 (2017).
Abernathy, W. J. & Utterback, J. M. Patterns of industrial innovation. Technol. Rev. 80, 40–47 (1978).
Christensen, C. The Innovator’s Dilemma: When New Technologies Cause Great Firms to Fail (Harvard Business School Press, 1997).
John, G., Weiss, A. M. & Dutta, S. Marketing in technology-intensive markets: toward a conceptual framework. J. Mark. 63, 78–91 (1999).
Rogers, E. M. Diffusion of Innovations (Simon and Schuster, 2010).
Hobbs, R. J. & Norton, D. A. Towards a conceptual framework for restoration ecology. Restor. Ecol. 4, 93–110 (1996).
Suding, K. N. Toward an era of restoration in ecology: successes, failures, and opportunities ahead. Annu. Rev. Ecol. Evol. Syst. 42, 465–487 (2011).
Higgs, E. et al. On principles and standards in ecological restoration. Restor. Ecol. 26, 399–403 (2018).
Gann, G. D. et al. The SER Standards: a globally relevant and inclusive tool for improving restoration practice—a reply to Higgs et al. Restor. Ecol. 26, 426–430 (2018).
Hobbs, R. J. Restoration Ecology’s silver jubilee: innovation, debate, and creating a future for restoration ecology. Restor. Ecol. 26, 801–805 (2018).
Massatti, R. et al. Population history provides foundational knowledge for utilizing and developing native plant restoration materials. Evol. Appl. 11, 2025–2039 (2018).
Acknowledgements
We thank J. Robinson for help preparing figure 1 and P. Cassey and B. Potts for comments on earlier versions of this manuscript. M.F.B. is funded by Australian Research Council (ARC) grants DP180100668, DE150100542 and DP150103414. P.A.H is supported by ARC grant IC150100004. V.G. is supported by a Postdoctoral Research Project awarded by Water Research Australia (1110–17).
Author information
Authors and Affiliations
Contributions
M.F.B., P.A.H., R.H. and D.A.S. researched data for the article. M.F.B., P.A.H., C.B., M.B., V.G., S.V.C.G., R.H., J.G.M., T.A.A.P., D.A.S. and J.J.M. made substantial contributions to discussions of the content of the manuscript. M.F.B., P.A.H., C.B., V.G., N.J.C.G., S.V.C.G., J.G.M., T.A.A.P., D.A.S. and J.J.M. wrote the article. All authors reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
AFR100: https://afr100.org/
Earth Microbiome Project: http://www.earthmicrobiome.org/
Initiative 20×20: https://www.wri.org/our-work/project/initiative-20x20
REDD+: https://redd.unfccc.int/
The Bonn Challenge: http://www.bonnchallenge.org/
UNFCCC Paris Agreement: https://unfccc.int/process-and-meetings/the-paris-agreement/the-paris-agreement
Supplementary information
Glossary
- Adaptive variation
-
Genetic variation that increases the fitness of an organism.
- Alpha diversity
-
The species diversity within a given sample or site.
- Beta diversity
-
The turnover of species diversity across a landscape.
- CRISPR–Cas9 system
-
A targeted genome-editing tool comprising two components: the programmable Cas9 endonuclease, which introduces double-strand breaks into the DNA; and a guide RNA, which targets the Cas9 nuclease to a specific DNA sequence.
- Effective population sizes
-
The size of ideal breeding populations, which meet Hardy–Weinberg equilibrium assumptions, that would maintain the same allele frequencies as a census population.
- Environmental DNA or RNA
-
DNA or RNA present in an environmental sample, such as water, soil and air.
- Gene flow
-
The exchange of genetic material within or between populations as a result of the movement of gametes or individuals.
- Genetic drift
-
The change in allele frequencies through generations of a population due to random sampling.
- Genotype-by-environment
-
Differential trait responses (such as growth or survival) of genotypes grown in contrasting environments, resulting in a statistical genotype and environment interaction for traits.
- Guide RNA
-
(gRNA). A small sequence of synthetic RNA (about 20 bases long) located within a longer RNA scaffold, which binds to DNA and directs the Cas9 endonuclease to the targeted genomic location.
- Metabarcoding
-
A meta-omics approach that combines DNA identification and DNA sequencing, in which universal primers are used to amplify DNA barcodes from bulk samples, such as soil environmental DNA.
- Metabolomic turnover
-
The change in metabolic molecules within cells, biofluids, tissues or organisms.
- Metagenomics
-
A meta-omics approach similar to metabarcoding, but instead of using DNA barcodes it involves random sequencing of DNA from bulk samples.
- Metatranscriptomic
-
Pertaining to a meta-omics approach similar to metagenomics, but instead of randomly sequencing DNA it randomly sequences transcriptomes or expressed genes.
- Meta-omics
-
A collection of methods (including metabarcoding, metagenomics and metatranscriptomics) that use next-generation sequencing to characterize whole communities of organisms.
- Neutral variation
-
Genetic variation that is not shaped by natural selection and does not directly impact the fitness of an organism.
- Population genomics
-
The application of high-density, genome-wide molecular markers to the study of neutral and adaptive evolutionary processes occurring within species.
- Provenance
-
The geographical location of a plant population or seed source.
- Seed transfer zones
-
The geographical regions over which seeds can be transferred with minimal maladaptive responses.
- Transfer functions
-
The relationships between the performance of multiple plant populations within a test site and the environmental dissimilarity between the populations’ home site and test site.
Rights and permissions
About this article
Cite this article
Breed, M.F., Harrison, P.A., Blyth, C. et al. The potential of genomics for restoring ecosystems and biodiversity. Nat Rev Genet 20, 615–628 (2019). https://doi.org/10.1038/s41576-019-0152-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41576-019-0152-0
This article is cited by
-
Projection of current and future distribution of adaptive genetic units in an alpine ungulate
Heredity (2024)
-
Genetic assessment of the value of restoration planting within an endangered eucalypt woodland
Scientific Reports (2023)
-
Genetic consequence of widespread plantations of Cryptomeria japonica var. sinensis in Southern China: implications for afforestation strategies under climate change
Tree Genetics & Genomes (2023)
-
Digital entrepreneurship from cellular data: How omics afford the emergence of a new wave of digital ventures in health
Electronic Markets (2023)
-
Applying simple genomic workflows to optimise practical plant translocation outcomes
Plant Ecology (2023)