Abstract
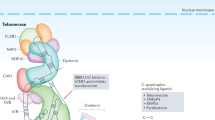
Telomerase is a key enzyme for cell survival that prevents telomere shortening and the subsequent cellular senescence that is observed after many rounds of cell division. In contrast, inactivation of telomerase is observed in most cells of the adult liver. Absence of telomerase activity and shortening of telomeres has been implicated in hepatocyte senescence and the development of cirrhosis, a chronic liver disease that can lead to hepatocellular carcinoma (HCC) development. During hepatocarcinogenesis, telomerase reactivation is required to enable the uncontrolled cell proliferation that leads to malignant transformation and HCC development. Part of the telomerase complex, telomerase reverse transcriptase, is encoded by TERT, and several mechanisms of telomerase reactivation have been described in HCC that include somatic TERT promoter mutations, TERT amplification, TERT translocation and viral insertion into the TERT gene. An understanding of the role of telomeres and telomerase in HCC development is important to develop future targeted therapies and improve survival of this disease. In this Review, the roles of telomeres and telomerase in liver carcinogenesis are discussed, in addition to their potential translation to clinical practice as biomarkers and therapeutic targets.
Key points
-
Telomerase is a key enzyme for cell survival that prevents telomere shortening after cell divisions.
-
Cirrhosis is characterized by replicative senescence owing to short telomeres and no telomerase expression (TERT gene) in mature hepatocytes.
-
A return of telomerase expression is observed in 90% of hepatocellular carcinomas (HCC).
-
Mechanisms of telomerase reactivation in HCC are related to TERT promoter mutations, TERT amplification, chromosome translocations and HBV or adeno-associated virus type 2 viral insertion into the TERT promoter.
-
Several biomarkers and therapies directed against telomerase are in development, but none of them is currently validated for use in HCC.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
El-Serag, H. B. Hepatocellular carcinoma. N. Engl. J. Med. 365, 1118–1127 (2011).
Forner, A., Llovet, J. M. & Bruix, J. Hepatocellular carcinoma. Lancet 379, 1245–1255 (2012).
Llovet, J. M. et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 359, 378–390 (2008).
Kudo, M. et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: a randomised phase 3 non-inferiority trial. Lancet 391, 1163–1173 (2018).
Bruix, J. et al. Efficacy and safety of sorafenib in patients with advanced hepatocellular carcinoma: subanalyses of a phase III trial. J. Hepatol. 57, 821–829 (2012).
Abou-Alfa, G. K. et al. Cabozantinib (C) versus placebo (P) in patients (pts) with advanced hepatocellular carcinoma (HCC) who have received prior sorafenib: results from the randomized phase III CELESTIAL trial [abstract]. J. Clin. Oncol. 36 (Suppl. 4), 207 (2018).
Bruix, J. et al. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 389, 56–66 (2017).
Llovet, J. M. & Hernandez-Gea, V. Hepatocellular carcinoma: reasons for phase III failure and novel perspectives on trial design. Clin. Cancer Res. 20, 2072–2079 (2014).
Pinyol, R., Nault, J. C., Quetglas, I. M., Zucman-Rossi, J. & Llovet, J. M. Molecular profiling of liver tumors: classification and clinical translation for decision making. Semin. Liver Dis. 34, 363–375 (2014).
Nault, J. C. Pathogenesis of hepatocellular carcinoma according to aetiology. Best Pract. Res. Clin. Gastroenterol. 28, 937–947 (2014).
Zucman-Rossi, J., Villanueva, A., Nault, J. C. & Llovet, J. M. Genetic landscape and biomarkers of hepatocellular carcinoma. Gastroenterology 149, 1226–1239 (2015).
Nault, J. C. & Zucman-Rossi, J. Genetics of hepatobiliary carcinogenesis. Semin. Liver Dis. 31, 173–187 (2011).
Guichard, C. et al. Integrated analysis of somatic mutations and focal copy-number changes identifies key genes and pathways in hepatocellular carcinoma. Nat. Genet. 44, 694–698 (2012).
Schulze, K. et al. Exome sequencing of hepatocellular carcinomas identifies new mutational signatures and potential therapeutic targets. Nat. Genet. 47, 505–511 (2015).
Schulze, K., Nault, J. C. & Villanueva, A. Genetic profiling of hepatocellular carcinoma using next-generation sequencing. J. Hepatol. 65, 1031–1042 (2016).
Stratton, M. R., Campbell, P. J. & Futreal, P. A. The cancer genome. Nature 458, 719–724 (2009).
Fujimoto, A. et al. Whole-genome sequencing of liver cancers identifies etiological influences on mutation patterns and recurrent mutations in chromatin regulators. Nat. Genet. 44, 760–764 (2012).
Totoki, Y. et al. Trans-ancestry mutational landscape of hepatocellular carcinoma genomes. Nat. Genet. 46, 1267–1273 (2014).
Hoare, M., Das, T. & Alexander, G. Ageing, telomeres, senescence, and liver injury. J. Hepatol. 53, 950–961 (2010).
Rudolph, K. L., Hartmann, D. & Opitz, O. G. Telomere dysfunction and DNA damage checkpoints in diseases and cancer of the gastrointestinal tract. Gastroenterology 137, 754–762 (2009).
Satyanarayana, A., Manns, M. P. & Rudolph, K. L. Telomeres and telomerase: a dual role in hepatocarcinogenesis. Hepatology 40, 276–283 (2004).
Olovnikov, A. M. Principle of marginotomy in template synthesis of polynucleotides [Russian]. Dokl. Akad. Nauk SSSR 201, 1496–1499 (1971).
Blackburn, E. H. Switching and signaling at the telomere. Cell 106, 661–673 (2001).
Varela, E. & Blasco, M. A. 2009 Nobel Prize in Physiology or Medicine: telomeres and telomerase. Oncogene 29, 1561–1565 (2010).
Shampay, J., Szostak, J. W. & Blackburn, E. H. DNA sequences of telomeres maintained in yeast. Nature 310, 154–157 (1984).
Calado, R. T. & Young, N. S. Telomere diseases. N. Engl. J. Med. 361, 2353–2365 (2009).
Smith, F. W. & Feigon, J. Quadruplex structure of Oxytricha telomeric DNA oligonucleotides. Nature 356, 164–168 (1992).
Brown, J. P., Wei, W. & Sedivy, J. M. Bypass of senescence after disruption of p21CIP1/WAF1 gene in normal diploid human fibroblasts. Science 277, 831–834 (1997).
Stein, G. H., Drullinger, L. F., Soulard, A. & Dulic, V. Differential roles for cyclin-dependent kinase inhibitors p21 and p16 in the mechanisms of senescence and differentiation in human fibroblasts. Mol. Cell. Biol. 19, 2109–2117 (1999).
Counter, C. M. et al. Telomere shortening associated with chromosome instability is arrested in immortal cells which express telomerase activity. EMBO J. 11, 1921–1929 (1992).
Wright, W. E. & Shay, J. W. The two-stage mechanism controlling cellular senescence and immortalization. Exp. Gerontol. 27, 383–389 (1992).
d’Adda di Fagagna, F. et al. A DNA damage checkpoint response in telomere-initiated senescence. Nature 426, 194–198 (2003).
Artandi, S. E. & DePinho, R. A. Telomeres and telomerase in cancer. Carcinogenesis 31, 9–18 (2010).
Gilson, E. & Geli, V. How telomeres are replicated. Nat. Rev. Mol. Cell Biol. 8, 825–838 (2007).
Gunes, C. & Rudolph, K. L. The role of telomeres in stem cells and cancer. Cell 152, 390–393 (2013).
Munoz-Espin, D. & Serrano, M. Cellular senescence: from physiology to pathology. Nat. Rev. Mol. Cell Biol. 15, 482–496 (2014).
Urabe, Y. et al. Telomere length in human liver diseases. Liver 16, 293–297 (1996).
Wiemann, S. U. et al. Hepatocyte telomere shortening and senescence are general markers of human liver cirrhosis. FASEB J. 16, 935–942 (2002).
Paradis, V. et al. Replicative senescence in normal liver, chronic hepatitis C, and hepatocellular carcinomas. Hum. Pathol. 32, 327–332 (2001).
Aravinthan, A. et al. Hepatocyte senescence predicts progression in non-alcohol-related fatty liver disease. J. Hepatol. 58, 549–556 (2013).
Brunt, E. M., Walsh, S. N., Hayashi, P. H., Labundy, J. & Di Bisceglie, A. M. Hepatocyte senescence in end-stage chronic liver disease: a study of cyclin-dependent kinase inhibitor p21 in liver biopsies as a marker for progression to hepatocellular carcinoma. Liver Int. 27, 662–671 (2007).
Aravinthan, A. et al. Hepatocyte expression of the senescence marker p21 is linked to fibrosis and an adverse liver-related outcome in alcohol-related liver disease. PLOS One 8, e72904 (2013).
Rudolph, K. L., Chang, S., Millard, M., Schreiber-Agus, N. & DePinho, R. A. Inhibition of experimental liver cirrhosis in mice by telomerase gene delivery. Science 287, 1253–1258 (2000).
Hartmann, D. et al. Telomerase gene mutations are associated with cirrhosis formation. Hepatology 53, 1608–1617 (2011).
Calado, R. T. et al. Constitutional telomerase mutations are genetic risk factors for cirrhosis. Hepatology 53, 1600–1607 (2011).
Deng, Y., Chan, S. S. & Chang, S. Telomere dysfunction and tumour suppression: the senescence connection. Nat. Rev. Cancer 8, 450–458 (2008).
Patnaik, M. M., Kamath, P. S. & Simonetto, D. A. Hepatic manifestations of telomere biology disorders. J. Hepatol. 69, 736–743 (2018).
Carulli, L., Dei Cas, A. & Nascimbeni, F. Synchronous cryptogenic liver cirrhosis and idiopathic pulmonary fibrosis: a clue to telomere involvement. Hepatology 56, 2001–2003 (2012).
Gorgy, A. I. et al. Hepatopulmonary syndrome is a frequent cause of dyspnea in the short telomere disorders. Chest 148, 1019–1026 (2015).
Ganne-Carrie, N. et al. Predictive score for the development of hepatocellular carcinoma and additional value of liver large cell dysplasia in Western patients with cirrhosis. Hepatology 23, 1112–1118 (1996).
Trinchet, J. C. et al. Complications and competing risks of death in compensated viral cirrhosis (ANRS CO12 CirVir prospective cohort). Hepatology 62, 737–750 (2015).
Lechel, A. et al. Telomerase deletion limits progression of p53-mutant hepatocellular carcinoma with short telomeres in chronic liver disease. Gastroenterology 132, 1465–1475 (2007).
Blasco, M. A. et al. Telomere shortening and tumor formation by mouse cells lacking telomerase RNA. Cell 91, 25–34 (1997).
Rudolph, K. L. et al. Longevity, stress response, and cancer in aging telomerase-deficient mice. Cell 96, 701–712 (1999).
Farazi, P. A. et al. Differential impact of telomere dysfunction on initiation and progression of hepatocellular carcinoma. Cancer Res. 63, 5021–5027 (2003).
Wiemann, S. U. et al. Contrasting effects of telomere shortening on organ homeostasis, tumor suppression, and survival during chronic liver damage. Oncogene 24, 1501–1509 (2005).
Farazi, P. A., Glickman, J., Horner, J. & Depinho, R. A. Cooperative interactions of p53 mutation, telomere dysfunction, and chronic liver damage in hepatocellular carcinoma progression. Cancer Res. 66, 4766–4773 (2006).
Sahin, E. et al. Telomere dysfunction induces metabolic and mitochondrial compromise. Nature 470, 359–365 (2011).
Sahin, E. & Depinho, R. A. Linking functional decline of telomeres, mitochondria and stem cells during ageing. Nature 464, 520–528 (2010).
Farazi, P. A. & DePinho, R. A. Hepatocellular carcinoma pathogenesis: from genes to environment. Nat. Rev. Cancer 6, 674–687 (2006).
Plentz, R. R. et al. Hepatocellular telomere shortening correlates with chromosomal instability and the development of human hepatoma. Hepatology 40, 80–86 (2004).
Meena, J. K. et al. Telomerase abrogates aneuploidy-induced telomere replication stress, senescence and cell depletion. EMBO J. 34, 1371–1384 (2015).
Begus-Nahrmann, Y. et al. Transient telomere dysfunction induces chromosomal instability and promotes carcinogenesis. J. Clin. Invest. 122, 2283–2288 (2012).
Ohashi, K. et al. Telomere changes in human hepatocellular carcinomas and hepatitis virus infected noncancerous livers. Cancer 77, 1747–1751 (1996).
Ferlicot, S., Paradis, V., Dargere, D., Monges, G. & Bedossa, P. Detection of telomerase in hepatocellular carcinomas using a PCR ELISA assay: comparison with hTR expression. J. Clin. Pathol. 52, 725–729 (1999).
Nakayama, J. et al. Telomerase activation by hTRT in human normal fibroblasts and hepatocellular carcinomas. Nat. Genet. 18, 65–68 (1998).
Shimada, M. et al. The role of telomerase activity in hepatocellular carcinoma. Am. J. Gastroenterol. 95, 748–752 (2000).
Youssef, N., Paradis, V., Ferlicot, S. & Bedossa, P. In situ detection of telomerase enzymatic activity in human hepatocellular carcinogenesis. J. Pathol. 194, 459–465 (2001).
Horn, S. et al. TERT promoter mutations in familial and sporadic melanoma. Science 339, 959–961 (2013).
Huang, F. W. et al. Highly recurrent TERT promoter mutations in human melanoma. Science 339, 957–959 (2013).
Bell, R. J. et al. The transcription factor GABP selectively binds and activates the mutant TERT promoter in cancer. Science 348, 1036–1039 (2015).
Ko, E., Seo, H. W., Jung, E. S., Kim, B. H. & Jung, G. The TERT promoter SNP rs2853669 decreases E2F1 transcription factor binding and increases mortality and recurrence risks in liver cancer. Oncotarget 7, 684–699 (2016).
Heidenreich, B., Rachakonda, P. S., Hemminki, K. & Kumar, R. TERT promoter mutations in cancer development. Curr. Opin. Genet. Dev. 24, 30–37 (2014).
Chiba, K. et al. Cancer-associated TERT promoter mutations abrogate telomerase silencing. eLife 4, e07918 (2015).
Borah, S. et al. TERT promoter mutations and telomerase reactivation in urothelial cancer. Science 347, 1006–1010 (2015).
Chiba, K. et al. Mutations in the promoter of the telomerase gene TERT contribute to tumorigenesis by a two-step mechanism. Science 357, 1416–1420 (2017).
Killela, P. J. et al. TERT promoter mutations occur frequently in gliomas and a subset of tumors derived from cells with low rates of self-renewal. Proc. Natl Acad. Sci. U. S. A. 110, 6021–6026 (2013).
Allory, Y. et al. Telomerase reverse transcriptase promoter mutations in bladder cancer: high frequency across stages, detection in urine, and lack of association with outcome. Eur. Urol. 65, 360–366 (2014).
Vinagre, J. et al. Frequency of TERT promoter mutations in human cancers. Nat. Commun. 4, 2185 (2013).
Griewank, K. G. et al. TERT promoter mutations are frequent in cutaneous basal cell carcinoma and squamous cell carcinoma. PLoS One 8, e80354 (2013).
Griewank, K. G. et al. TERT promoter mutations in ocular melanoma distinguish between conjunctival and uveal tumours. Br. J. Cancer 109, 497–501 (2013).
Griewank, K. G. et al. TERT promoter mutations are frequent in atypical fibroxanthomas and pleomorphic dermal sarcomas. Mod. Pathol. 27, 502–508 (2014).
Tallet, A. et al. Overexpression and promoter mutation of the TERT gene in malignant pleural mesothelioma. Oncogene 33, 3748–3752 (2014).
Barthel, F. P. et al. Systematic analysis of telomere length and somatic alterations in 31 cancer types. Nat. Genet. 49, 349–357 (2017).
Valentijn, L. J. et al. TERT rearrangements are frequent in neuroblastoma and identify aggressive tumors. Nat. Genet. 47, 1411–1414 (2015).
Peifer, M. et al. Telomerase activation by genomic rearrangements in high-risk neuroblastoma. Nature 526, 700–704 (2015).
Karlsson, J. et al. Activation of human telomerase reverse transcriptase through gene fusion in clear cell sarcoma of the kidney. Cancer Lett. 357, 498–501 (2015).
Nault, J. C. et al. High frequency of telomerase reverse-transcriptase promoter somatic mutations in hepatocellular carcinoma and preneoplastic lesions. Nat. Commun. 4, 2218 (2013).
The Cancer Genome Atlas Research Network. Comprehensive and integrative genomic characterization of hepatocellular carcinoma. Cell 169, 1327–1341 (2017).
Park, J. I. et al. Telomerase modulates Wnt signalling by association with target gene chromatin. Nature 460, 66–72 (2009).
Chen, Y. L. et al. TERT promoter mutation in resectable hepatocellular carcinomas: a strong association with hepatitis C infection and absence of hepatitis B infection. Int. J. Surg. 12, 659–665 (2014).
Quaas, A. et al. Frequency of TERT promoter mutations in primary tumors of the liver. Virchows Arch. 465, 673–677 (2014).
Cevik, D., Yildiz, G. & Ozturk, M. Common telomerase reverse transcriptase promoter mutations in hepatocellular carcinomas from different geographical locations. World J. Gastroenterol. 21, 311–317 (2015).
Huang, D. S. et al. Recurrent TERT promoter mutations identified in a large-scale study of multiple tumour types are associated with increased TERT expression and telomerase activation. Eur. J. Cancer 51, 969–976 (2015).
Yang, X. et al. Telomerase reverse transcriptase promoter mutations in hepatitis B virus-associated hepatocellular carcinoma. Oncotarget 7, 27838–27847 (2016).
Kawai-Kitahata, F. et al. Comprehensive analyses of mutations and hepatitis B virus integration in hepatocellular carcinoma with clinicopathological features. J. Gastroenterol. 51, 473–486 (2016).
Chianchiano, P. et al. Distinction of intrahepatic metastasis from multicentric carcinogenesis in multifocal hepatocellular carcinoma using molecular alterations. Hum. Pathol. 72, 127–134 (2018).
Torrecilla, S. et al. Trunk mutational events present minimal intra- and inter-tumoral heterogeneity in hepatocellular carcinoma. J. Hepatol. 67, 1222–1231 (2017).
Lee, S. E. et al. Frequent somatic TERT promoter mutations and CTNNB1 mutations in hepatocellular carcinoma. Oncotarget 7, 69267–69275 (2016).
Pezzuto, F. et al. Tumor specific mutations in TERT promoter and CTNNB1 gene in hepatitis B and hepatitis C related hepatocellular carcinoma. Oncotarget 7, 54253–54262 (2016).
Letouze, E. et al. Mutational signatures reveal the dynamic interplay of risk factors and cellular processes during liver tumorigenesis. Nat. Commun. 8, 1315 (2017).
Bayard, Q. et al. Cyclin A2/E1 activation defines a hepatocellular carcinoma subclass with a rearrangement signature of replication stress. Nat. Commun. 9, 5235 (2018).
Neuveut, C., Wei, Y. & Buendia, M. A. Mechanisms of HBV-related hepatocarcinogenesis. J. Hepatol. 52, 594–604 (2010).
Levrero, M. & Zucman-Rossi, J. Mechanisms of HBV-induced hepatocellular carcinoma. J. Hepatol. 64, S84–101 (2016).
Brechot, C., Pourcel, C., Louise, A., Rain, B. & Tiollais, P. Presence of integrated hepatitis B virus DNA sequences in cellular DNA of human hepatocellular carcinoma. Nature 286, 533–535 (1980).
Dejean, A., Bougueleret, L., Grzeschik, K. H. & Tiollais, P. Hepatitis B virus DNA integration in a sequence homologous to v-erb-A and steroid receptor genes in a hepatocellular carcinoma. Nature 322, 70–72 (1986).
Sung, W. K. et al. Genome-wide survey of recurrent HBV integration in hepatocellular carcinoma. Nat. Genet. 44, 765–769 (2012).
Xue, R. et al. Variable intra-tumor genomic heterogeneity of multiple lesions in patients with hepatocellular carcinoma. Gastroenterology 150, 998–1008 (2016).
Zhao, L. H. et al. Genomic and oncogenic preference of HBV integration in hepatocellular carcinoma. Nat. Commun. 7, 12992 (2016).
Nault, J. C. et al. Recurrent AAV2-related insertional mutagenesis in human hepatocellular carcinomas. Nat. Genet. 47, 1187–1193 (2015).
Logan, G. J. et al. Identification of liver-specific enhancer-promoter activity in the 3′ untranslated region of the wild-type AAV2 genome. Nat. Genet. 49, 1267–1273 (2017).
Cesare, A. J. & Reddel, R. R. Alternative lengthening of telomeres: models, mechanisms and implications. Nat. Rev. Genet. 11, 319–330 (2010).
Heaphy, C. M. et al. Prevalence of the alternative lengthening of telomeres telomere maintenance mechanism in human cancer subtypes. Am. J. Pathol. 179, 1608–1615 (2011).
Bechter, O. E., Zou, Y., Walker, W., Wright, W. E. & Shay, J. W. Telomeric recombination in mismatch repair deficient human colon cancer cells after telomerase inhibition. Cancer Res. 64, 3444–3451 (2004).
Heaphy, C. M. et al. Altered telomeres in tumors with ATRX and DAXX mutations. Science 333, 425 (2011).
Lovejoy, C. A. et al. Loss of ATRX, genome instability, and an altered DNA damage response are hallmarks of the alternative lengthening of telomeres pathway. PLOS Genet. 8, e1002772 (2012).
Wood, L. D. et al. Chromophobe hepatocellular carcinoma with abrupt anaplasia: a proposal for a new subtype of hepatocellular carcinoma with unique morphological and molecular features. Mod. Pathol. 26, 1586–1593 (2013).
Di Tommaso, L. et al. Advanced precancerous lesions in the liver. Best Pract. Res. Clin. Gastroenterol. 27, 269–284 (2013).
Borzio, M. et al. Impact of large regenerative, low grade and high grade dysplastic nodules in hepatocellular carcinoma development. J. Hepatol. 39, 208–214 (2003).
Nault, J. C. et al. Telomerase reverse transcriptase promoter mutation is an early somatic genetic alteration in the transformation of premalignant nodules in hepatocellular carcinoma on cirrhosis. Hepatology 60, 1983–1992 (2014).
Nault, J. C. et al. Molecular classification of hepatocellular adenoma associates with risk factors, bleeding, and malignant transformation. Gastroenterology 152, 880–894 (2017).
Bioulac-Sage, P., Cubel, G., Balabaud, C. & Zucman-Rossi, J. Revisiting the pathology of resected benign hepatocellular nodules using new immunohistochemical markers. Semin. Liver Dis. 31, 91–103 (2011).
Bluteau, O. et al. Bi-allelic inactivation of TCF1 in hepatic adenomas. Nat. Genet. 32, 312–315 (2002).
Nault, J. C. et al. GNAS-activating mutations define a rare subgroup of inflammatory liver tumors characterized by STAT3 activation. J. Hepatol. 56, 184–191 (2012).
Pilati, C. et al. Genomic profiling of hepatocellular adenomas reveals recurrent FRK-activating mutations and the mechanisms of malignant transformation. Cancer Cell 25, 428–441 (2014).
Rebouissou, S. et al. Frequent in-frame somatic deletions activate gp130 in inflammatory hepatocellular tumours. Nature 457, 200–204 (2009).
Pilati, C. et al. Somatic mutations activating STAT3 in human inflammatory hepatocellular adenomas. J. Exp. Med. 208, 1359–1366 (2011).
Nault, J. C. et al. ASS1 and peri-portal gene expression in sonic hedgehog hepatocellular adenomas. Hepatology 68, 964–976 (2018).
Calderaro, J. et al. Systemic AA amyloidosis caused by inflammatory hepatocellular adenoma. N. Engl. J. Med. 379, 1178–1180 (2018).
Goutagny, S. et al. High incidence of activating TERT promoter mutations in meningiomas undergoing malignant progression. Brain Pathol. 24, 184–189 (2014).
Pestana, A., Vinagre, J., Sobrinho-Simoes, M. & Soares, P. TERT biology and function in cancer: beyond immortalisation. J. Mol. Endocrinol. 58, R129–R146 (2017).
Jung, S. W. et al. Prognostic impact of telomere maintenance gene polymorphisms on hepatocellular carcinoma patients with chronic hepatitis B. Hepatology 59, 1912–1920 (2014).
Bao, D. et al. Alterations of telomere length and mtDNA copy number are associated with overall survival in hepatocellular carcinoma patients treated with transarterial chemoembolization. Cancer Chemother. Pharmacol. 78, 791–799 (2016).
Liu, H. Q. et al. Leukocyte telomere length predicts overall survival in hepatocellular carcinoma treated with transarterial chemoembolization. Carcinogenesis 33, 1040–1045 (2012).
Kobayashi, T., Kubota, K., Takayama, T. & Makuuchi, M. Telomerase activity as a predictive marker for recurrence of hepatocellular carcinoma after hepatectomy. Am. J. Surg. 181, 284–288 (2001).
Kobayashi, T., Sugawara, Y., Shi, Y. Z. & Makuuchi, M. Telomerase expression and p53 status in hepatocellular carcinoma. Am. J. Gastroenterol. 97, 3166–3171 (2002).
Oh, B. K. et al. High telomerase activity and long telomeres in advanced hepatocellular carcinomas with poor prognosis. Lab. Invest. 88, 144–152 (2008).
Yu, J. I. et al. Clinical importance of TERT overexpression in hepatocellular carcinoma treated with curative surgical resection in HBV endemic area. Sci. Rep. 7, 12258 (2017).
Nakashio, R. et al. Alteration of telomeric repeat length in hepatocellular carcinoma is independent of telomerase activity. Int. J. Oncol. 11, 139–143 (1997).
Kim, H. et al. Telomere length, TERT and shelterin complex proteins in hepatocellular carcinomas expressing “stemness”-related markers. J. Hepatol. 59, 746–752 (2013).
Ma, L. J. et al. Telomere length variation in tumor cells and cancer-associated fibroblasts: potential biomarker for hepatocellular carcinoma. J. Pathol. 243, 407–417 (2017).
Ko, E., Seo, H. W. & Jung, G. Telomere length and reactive oxygen species levels are positively associated with a high risk of mortality and recurrence in hepatocellular carcinoma. Hepatology 67, 1378–1391 (2018).
Ohta, K. et al. Telomerase activity in hepatocellular carcinoma as a predictor of postoperative recurrence. J. Gastroenterol. 32, 791–796 (1997).
Kanamaru, T. et al. Clinical implications of telomerase activity in resected hepatocellular carcinoma. Int. J. Mol. Med. 4, 267–271 (1999).
Yang, B. et al. Telomere length and survival of patients with hepatocellular carcinoma in the United States. PLOS One 11, e0166828 (2016).
Labgaa, I. et al. A pilot study of ultra-deep targeted sequencing of plasma DNA identifies driver mutations in hepatocellular carcinoma. Oncogene 37, 3740–3752 (2018).
Ruden, M. & Puri, N. Novel anticancer therapeutics targeting telomerase. Cancer Treat. Rev. 39, 444–456 (2013).
Harley, C. B. Telomerase and cancer therapeutics. Nat. Rev. Cancer 8, 167–179 (2008).
Djojosubroto, M. W. et al. Telomerase antagonists GRN163 and GRN163L inhibit tumor growth and increase chemosensitivity of human hepatoma. Hepatology 42, 1127–1136 (2005).
Tefferi, A. et al. A pilot study of the telomerase inhibitor imetelstat for myelofibrosis. N. Engl. J. Med. 373, 908–919 (2015).
Baerlocher, G. M. et al. Telomerase inhibitor imetelstat in patients with essential thrombocythemia. N. Engl. J. Med. 373, 920–928 (2015).
Buseman, C. M., Wright, W. E. & Shay, J. W. Is telomerase a viable target in cancer? Mutat. Res. 730, 90–97 (2012).
Geron. Press release: Geron provides update on imetelstat clinical development program. geron http://ir.geron.com/news-releases/news-release-details/geron-provides-update-imetelstat-clinical-development-program (2012).
Chiappori, A. A. et al. A randomized phase II study of the telomerase inhibitor imetelstat as maintenance therapy for advanced non-small-cell lung cancer. Ann. Oncol. 26, 354–362 (2015).
Salloum, R. et al. A molecular biology and phase II study of imetelstat (GRN163L) in children with recurrent or refractory central nervous system malignancies: a pediatric brain tumor consortium study. J. Neurooncol. 129, 443–451 (2016).
Sundquist, W. I. & Klug, A. Telomeric DNA dimerizes by formation of guanine tetrads between hairpin loops. Nature 342, 825–829 (1989).
Burger, A. M. et al. The G-quadruplex-interactive molecule BRACO-19 inhibits tumor growth, consistent with telomere targeting and interference with telomerase function. Cancer Res. 65, 1489–1496 (2005).
Salvati, E. et al. Telomere damage induced by the G-quadruplex ligand RHPS4 has an antitumor effect. J. Clin. Invest. 117, 3236–3247 (2007).
Tauchi, T. et al. Activity of a novel G-quadruplex-interactive telomerase inhibitor, telomestatin (SOT-095), against human leukemia cells: involvement of ATM-dependent DNA damage response pathways. Oncogene 22, 5338–5347 (2003).
Tahtouh, R. et al. Telomerase inhibition decreases alpha-fetoprotein expression and secretion by hepatocellular carcinoma cell lines: in vitro and in vivo study. PLOS One 10, e0119512 (2015).
Kotsakis, A. et al. Clinical outcome of patients with various advanced cancer types vaccinated with an optimized cryptic human telomerase reversetranscriptase (TERT) peptide: results of an expanded phase II study. Ann. Oncol. 23, 442–449 (2012).
Brunsvig, P. F. et al. Telomerase peptide vaccination in NSCLC: a phase II trial in stage III patients vaccinated after chemoradiotherapy and an 8-year update on a phase I/II trial. Clin. Cancer Res. 17, 6847–6857 (2011).
Anguille, S., Smits, E. L., Lion, E., van Tendeloo, V. F. & Berneman, Z. N. Clinical use of dendritic cells for cancer therapy. Lancet Oncol. 15, e257–e267 (2014).
Greten, T. F. et al. A phase II open label trial evaluating safety and efficacy of a telomerase peptide vaccination in patients with advanced hepatocellular carcinoma. BMC Cancer 10, 209 (2010).
Middleton, G. et al. Gemcitabine and capecitabine with or without telomerase peptide vaccine GV1001 in patients with locally advanced or metastatic pancreatic cancer (TeloVac): an open-label, randomised, phase 3 trial. Lancet Oncol. 15, 829–840 (2014).
Gilboa, E. & Vieweg, J. Cancer immunotherapy with mRNA-transfected dendritic cells. Immunol. Rev. 199, 251–263 (2004).
Su, Z. et al. Telomerase mRNA-transfected dendritic cells stimulate antigen-specific CD8+ and CD4+ T cell responses in patients with metastatic prostate cancer. J. Immunol. 174, 3798–3807 (2005).
Fenoglio, D. et al. A multi-peptide, dual-adjuvant telomerase vaccine (GX301) is highly immunogenic in patients with prostate and renal cancer. Cancer Immunol. Immunother. 62, 1041–1052 (2013).
Weiss, J. M., Subleski, J. J., Wigginton, J. M. & Wiltrout, R. H. Immunotherapy of cancer by IL-12-based cytokine combinations. Expert Opin. Biol. Ther. 7, 1705–1721 (2007).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02960594 (2018).
Zhang, Y. et al. AAV-mediated TRAIL gene expression driven by hTERT promoter suppressed human hepatocellular carcinoma growth in mice. Life Sci. 82, 1154–1161 (2008).
Lanson, N. A. et al. Replication of an adenoviral vector controlled by the human telomerase reverse transcriptase promoter causes tumor-selective tumor lysis. Cancer Res. 63, 7936–7941 (2003).
Nemunaitis, J. et al. A phase I study of telomerase-specific replication competent oncolytic adenovirus (telomelysin) for various solid tumors. Mol. Ther. 18, 429–434 (2010).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03190824 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03213054 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03172819 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02293850 (2018).
Chaklader, M. et al. 17-AAG mediated targeting of Hsp90 limits tert activity in peritoneal sarcoma related malignant ascites by downregulating cyclin D1 during cell cycle entry. Exp. Oncol. 34, 90–96 (2012).
Seimiya, H., Muramatsu, Y., Ohishi, T. & Tsuruo, T. Tankyrase 1 as a target for telomere-directed molecular cancer therapeutics. Cancer Cell 7, 25–37 (2005).
Nakamura, H. et al. Genomic spectra of biliary tract cancer. Nat. Genet. 47, 1003–1010 (2015).
Wang, A. et al. Whole-exome sequencing reveals the origin and evolution of hepato-cholangiocarcinoma. Nat. Commun. 9, 894 (2018).
Lee, H. W. et al. Clinicopathological characteristics of TERT promoter mutation and telomere length in hepatocellular carcinoma. Medicine (Baltimore) 96, e5766 (2017).
Oya, H. et al. Comparison between human-telomerase reverse transcriptase mRNA and alpha-fetoprotein mRNA as a predictive value for recurrence of hepatocellular carcinoma in living donor liver transplantation. Transplant. Proc. 38, 3636–3639 (2006).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02598661 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01731951 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02426086 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02301754 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02293707 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01197625 (2018).
Khoury, H. J. et al. Immune responses and long-term disease recurrence status after telomerase-based dendritic cell immunotherapy in patients with acute myeloid leukemia. Cancer 123, 3061–3072 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01660529 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT00834665 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03491683 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03502785 (2019).
Acknowledgements
This work was supported by Institut National du Cancer (INCa) with the International Cancer Genome Consortium (ICGC LICA-FR project) and NoFLIC projects (PAIR HCC, INCa and ARC), INSERM with the “Cancer et Environnement” (plan Cancer), MUTHEC projects (INCa), TELOTHEP project (PRTK2017 INCA) and HETCOLI projects (Tumor Heterogeneity and Ecosystem Program). The group is supported by the Ligue Nationale contre le Cancer (Equipe Labellisée), Labex OncoImmunology (investissement d’avenir), Coup d’Elan de la Fondation Bettencourt-Shueller, the SIRIC CARPEM, Fondation Mérieux, Prix Duquesne (Ligue Contre le Cancer, Comité de Paris) and Prix Raymond Rosen (Fondation pour la Recherche Médicale).
Author information
Authors and Affiliations
Contributions
All authors contributed equally to all aspects of the writing of this Review.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nault, JC., Ningarhari, M., Rebouissou, S. et al. The role of telomeres and telomerase in cirrhosis and liver cancer. Nat Rev Gastroenterol Hepatol 16, 544–558 (2019). https://doi.org/10.1038/s41575-019-0165-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41575-019-0165-3
This article is cited by
-
Understanding, diagnosing, and treating pancreatic cancer from the perspective of telomeres and telomerase
Cancer Gene Therapy (2024)
-
Genetics of human telomere biology disorders
Nature Reviews Genetics (2023)
-
Major genomic mutations driving hepatocellular carcinoma
Genome Instability & Disease (2023)
-
Integrated genomic analyses of hepatocellular carcinoma
Hepatology International (2023)
-
Acetyl-CoA metabolism drives epigenome change and contributes to carcinogenesis risk in fatty liver disease
Genome Medicine (2022)